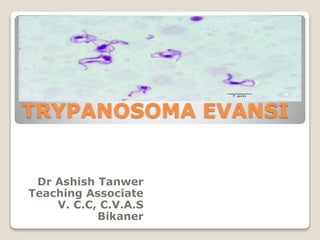
Trypanosoma evansi
- 1. TRYPANOSOMA EVANSI Dr Ashish Tanwer Teaching Associate V. C.C, C.V.A.S Bikaner
- 2. • Disease Surra in all hosts Tebersa in camels Murine in horses o 1st trypanosome shown to be pathogenic for mammals and is a intercellular parasite in blood and lymph. • Host: Camel, Horses, Donkeys, Cattle, Buffalo, Sheep Goat, Dog, Cat, elephant and other animals (tiger, fox, jackal, hyena, orangutan and mongoose), rodents, rabbits and guinea pig. • Vectors: Tabanids, Stomoxys, Haematopota, Hippobosca, Ornithodorus tick. • Location: Blood and lymph.
- 3. • Morphology T. evansi is monomorphic but polymorphism sporadically Slender, thin, indistinguishable from T. brucei and 15-34 μm length. Has sub-terminal kinetoplast, well developed undulating membrane and prominent free flagellum. • Epidemiology Host susceptibility Increased incidence is in equines and dogs than cattle and buffaloes. Buffaloes greater than cattle; Imported dogs greater than native dogs; pups greater than adults.
- 4. Transmission Mechanically biting flies- Tabanus, Stomoxys, Haematopota, Chrysops, Hippobosca and Lyperosia; Ticks-Ornithodorus spp. Interrupted feeding of flies from one host to other as parasite do not survive in the proboscis of the flies after 4-72 hours. Mechanically by non-blood sucking flies which transmit the diseases by picking the infection from infected meat or open lesions or mucous membrane of susceptible animals. Dogs get infection by ingestion of tissues from infected carcasses. During mass vaccination when sterilization procedures are inadequate. Carrier status Several wild and domestic animals carry the latent infection, e.g. cattle and buffaloes.
- 6. Stress factors Strain, malnutrition, vaccination against viral and microbial infections and intercurrent diseases may reduce the vitality of animals and increase the susceptibility to trypanosomes. Trypanotolerance Animal hosts are parasitaemic for prolonged periods of time, but generally remain in good health. Half breeds resulting from Jersey bull and N’-Dama female show good tolerance to trypanosomosis Incidence In India, more common in areas where the environment for breeding of insect vectors being the most suitable for transmission. Incidence coincides with rain, flood and inundations
- 7. PATHOGENESIS AND CLINICAL SIGNS Pathogenesis • Course of the disease depends on the factors like genetic constitution of the host, previous exposure to disease and the virulence of the infection, strain of the parasite and species of the host. • Mechanism responsible for anaemia-3 factors Haemolysis of RBCs by trypanosomes. Increased erythrophagocytosis mediated by antibody on antigen with complement Haemodilution due to increased plasma production. Others- reduced erythropoiesis, intravascular coagulation and death.
- 8. • Causes of death Progressive anaemia o Intravascular coagulation Hypoglycemia due to disturbed metabolism by malfunction of adrenals, pancreas and thyroid and due to direct utilization of glucose by the parasite. • Biochemical changes Increase in globulin and decrease in albumin globulin ratio by 14.4%. The infection is severe in dogs and horses than cattle and buffaloes.
- 10. Clinical signs • Equines Incubation Period: 4 to 9 days Severe in horses Donkeys are resistant Death within few days to few months depending on the virulence of strain of the organism. Intermittent fever (44○C) and anaemia are main symptoms. Transient local/ urticarial eruptions/ plaques on neck/ flanks; edema of legs and lower parts of the body.
- 11. Hemorrhage at the junction of the skin and mucous membrane at nostrils, eyes and anus and petechial haemorrhage on mucous membrane of eye or vulva in mares; pale and dirty yellowish mucous membrane Staggering gait, paraplegia, laboured breathing. Diarrhoea ,constipation, urine with albumin and blood and dark yellowish; Increased thirst, signs of catarrh with yellow tinged nasal discharge. Less common: Enlargement of lymph nodes, keratitis, petechiae in vaginal mucus membrane.
- 12. • Cattle and Buffaloes Course varies from a symptomless carrier to per acute infection. Per acute - Death within in 2-3 hour; nervous form and death in convulsion. Acute - Dull, sleepy, staggering gait, encircling movements, nervous excitement, beating head against wall /manger, apparent blindness, stamping of feet, bellowing, groaning, twitching of muscle, shivering of body, coma and death within 6-12 hours. Sub acute/ Chronic -Parasitaemia is usually low and doesn’t coincide with temperature and afebrile animal be positive for parasites, edema of legs, diarrhea, intermittent fever, rapid pulse, dullness, sleepy, bilateral lachrymation, progressive emaciation and death; abortion reported in buffaloes. PM findings: Spleenomegaly, hepatomegaly, enlargement of lymph nodes and kidney, petechial haemorrhage at the junction of skin and mucous membrane
- 14. • Dogs Incubation period: 5 to 6 days; untreated dogs die within 12 months, acute and fatal; more serious in pups and imported dogs. Fever, anorexia, edema of head and throat, corneal opacity or blindness. Laryngeal edema resulting in change of voice simulating rabies. Muscular spasm of limb, staggering gait, excitement like biting kennel bars simulating rabies. • Cats: Chronic cases are reported; at times may be fatal. •Sheep and Goats: Rare; intensity of symptoms are also moderate; emaciation and anaemia may be present. • Pigs: non-pathogenic and low parasitaemia.
- 15. DIAGNOSIS • History of prevalence of T. evansi infection and biting flies like tabanids. • Clinical signs. • Differential diagnosis Anthrax Snake bite Nervous ketosis, Organic and inorganic poisoning, Milk fever Hypovitaminosis, Milk fever, Brain tumor/ cyst
- 16. • Direct examination In acute cases-demonstration of organisms in blood smears stained freshly with Giemsa stain. Chronic cases- examination of thick and thin blood smear at the height of the temperature will be positive/ lymph node puncture smears. • Chemical tests To detect the changes in the chemical composition of the blood produced by T. evansi, like alteration of proteins Non-specific and less reliable. Mercuric chloride test, Formal gel test, thymol turbidity test are used for camels. Stilbamide test-diagnosis of latent infection in bovines in India.
- 17. • Animal inoculation tests For detection of latent infection of bovines and more reliable than microscopic examination. Albino mice/ rat are most suitable for detecting sub patent infection; guinea pigs and rabbits are less susceptible. Injection of 2 ml of blood intraperitoneally into the mice causes organisms appear after 2-3 days in the mice. • Immunodiagnostic tests IFAT, CFT, ELISA & IHA are most reliable tests. Allergic tests are doubtful- 1ml antigen is injected at the side of the neck with a 2nd dose after 48 hours. Infected cases show hot, edematous, painful swelling and healthy cases show circumscribed, hard, nodular swelling.
- 18. TREATMENT • Quinapyramines (Antryside) - Prophylactic (Triquin)- Drug of choice. Antryside methyl sulphate solution 3mg/kg as 10% aqueous solution, S/C; effective against T. evansi, T. congolense, T. brucei, T. vivax. Antrycide chloride as antrycide prosalt In combination with antrycide methyl sulphate -3 parts of A. M.S + 2 parts A. chloride A solution of 3.5 gm in 15 ml water is given @ 7.4 mg/kg; effective against T. evansi and T.brucei, for 3 months.
- 19. • Suramins (Sulphonated naphthalamines) - Curative (Naganaol, Antrypol) Effective against T. evansi, T. brucei, T. equiperdum. Cattle: 0.5 g/ 45 kg followed by half dose after 2 weeks, I/V. Horse: 4g/ 45 kg as single dose, I/ V. Dog: 3.5 ml- 10%solution followed by 2nd dose at 3-4 weeks interval, I/V. • Diamidines: Diminazene aceturate (Berenil) - Curative,@ 3.5 mg/ kg ; cattle - 10 mg /kg; S/C or deep I/M Very effective against T. congolense and less effective against T. evansi, T. brucei , in bovines, ovines and caprines. Dog and camels react to dogs; resistance may occur to the drug.
- 20. • Phenanthiridine compounds Homidium bromide and Homidium chloride- 1-2% sol @ 1gm/kg b. wt, I/M against T. congolense and T.vivax but not against T. evansi. Pyrithridium bromide - 4 % sol @ 2mg/ kg b. wt, S/C or I/M against T.congolense and T.vivax. Isometamidium chloride - 0.5 mg / kg b. wt, against T. simiae in pigs.
- 21. CONTROL • Control is difficult and the factors responsible for the wide spread existence are Latent cases of cattle and buffalo, Reservoir wild animals Drug resistant strains and Wide spread prevalence of vectors • Control program consists of Treatment of affected animals Chemoprophylaxis Disposal of manures properly o Proper drainage
- 22. Regular spraying of insecticides: Sprinkling of kerosene oil over water in ponds, ditches, streams or insecticides to kill adults and aquatic larvae. Removal of moist beddings, hay, dung from stable. Keeping animals clean by regular grooming. Segregation of sick animals from healthy during outbreak in fly -proof shed. Identification of endemic areas by regular examination of blood of animals for implementation of preventive measures.
- 23. Thanks