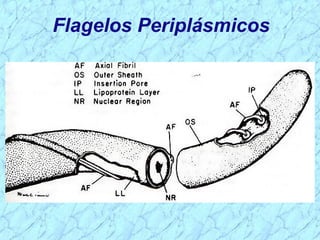
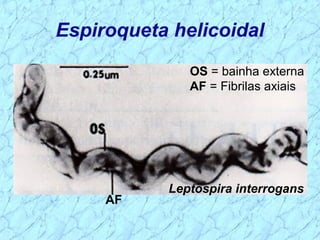
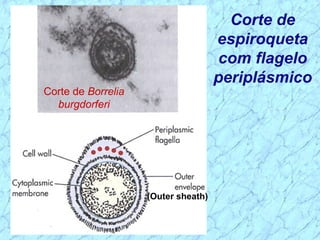
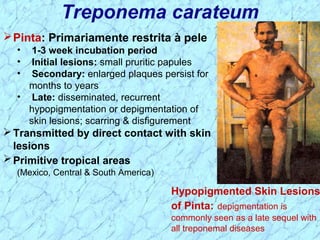
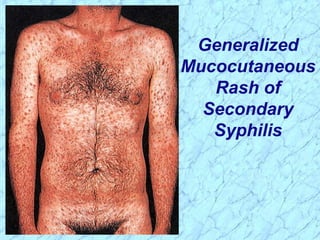
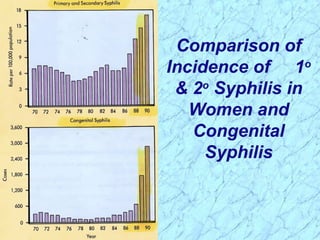
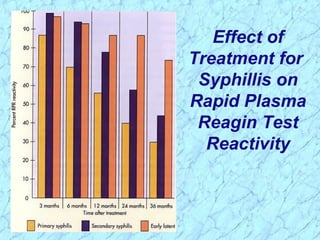
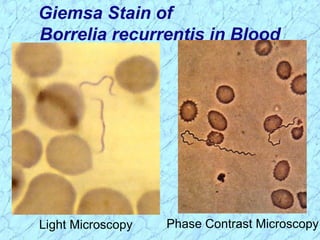
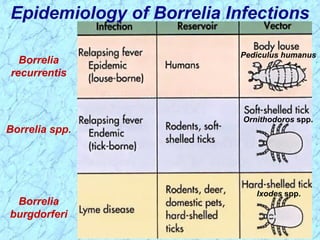
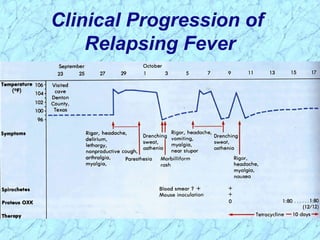
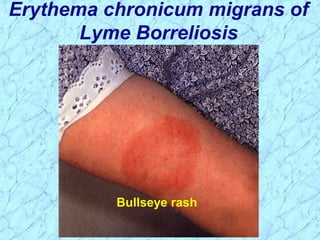
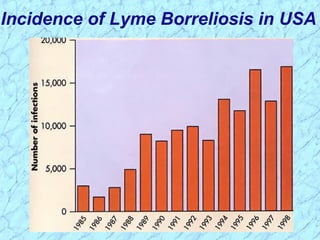
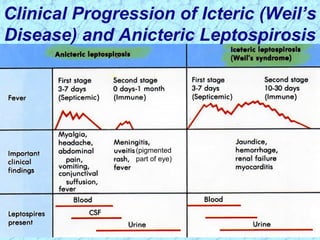
Treponema pallidum causes syphilis in humans. It has a long incubation period and can progress through primary, secondary, latent, and tertiary stages if left untreated. Borrelia recurrentis causes epidemic relapsing fever which is transmitted between humans by body lice. Borrelia burgdorferi causes Lyme disease transmitted by certain ticks and causes a characteristic bullseye rash. Leptospira interrogans causes leptospirosis, a zoonotic disease transmitted by contact with infected animal urine, which can range from mild to severe with jaundice and organ failure.