Traditional drug discovery process design 6 powerpoint ppt slides.
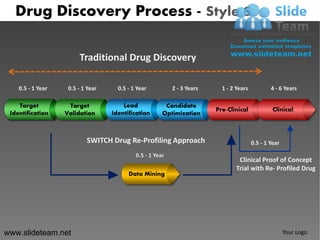
- 1. Drug Discovery Process - Style 6 Traditional Drug Discovery 0.5 - 1 Year 0.5 - 1 Year 0.5 - 1 Year 2 - 3 Years 1 - 2 Years 4 - 6 Years Target Target Lead Candidate Pre-Clinical Clinical Identification Validation Identification Optimization SWITCH Drug Re-Profiling Approach 0.5 - 1 Year 0.5 - 1 Year Clinical Proof of Concept Trial with Re- Profiled Drug Data Mining www.slideteam.net Your Logo
- 2. Drug Discovery Process - Style 6 Traditional Drug Discovery 0.5 - 1 Year 0.5 - 1 Year 0.5 - 1 Year 2 - 3 Years 1 - 2 Years 4 - 6 Years Target Target Lead Candidate Pre-Clinical Clinical Identification Validation Identification Optimization SWITCH Drug Re-Profiling Approach 0.5 - 1 Year 0.5 - 1 Year Clinical Proof of Concept Trial with Re- Profiled Drug Data Mining Put Text Here Your Text Here Put Text Here Your Text Here Put Text Here Your Text Here • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes here here here here here here • Download this • Download this • Download this • Download this • Download this • Download this awesome awesome awesome awesome awesome awesome diagram diagram diagram diagram diagram diagram • Bring your • Bring your • Bring your • Bring your • Bring your • Bring your presentation to presentation to presentation to presentation to presentation to presentation to life life life life life life www.slideteam.net Your Logo
- 3. Drug Discovery Process - Style 6 Traditional Drug Discovery 0.5 - 1 Year 0.5 - 1 Year 0.5 - 1 Year 2 - 3 Years 1 - 2 Years 4 - 6 Years Target Target Lead Candidate Pre-Clinical Clinical Identification Validation Identification Optimization SWITCH Drug Re-Profiling Approach 0.5 - 1 Year 0.5 - 1 Year Clinical Proof of Concept Trial with Re- Profiled Drug Data Mining Put Text Here Your Text Here Put Text Here Your Text Here Put Text Here Your Text Here • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes here here here here here here • Download this • Download this • Download this • Download this • Download this • Download this awesome awesome awesome awesome awesome awesome diagram diagram diagram diagram diagram diagram • Bring your • Bring your • Bring your • Bring your • Bring your • Bring your presentation to presentation to presentation to presentation to presentation to presentation to life life life life life life www.slideteam.net Your Logo
- 4. Drug Discovery Process - Style 6 Traditional Drug Discovery 0.5 - 1 Year 0.5 - 1 Year 0.5 - 1 Year 2 - 3 Years 1 - 2 Years 4 - 6 Years Target Target Lead Candidate Pre-Clinical Clinical Identification Validation Identification Optimization SWITCH Drug Re-Profiling Approach 0.5 - 1 Year 0.5 - 1 Year Clinical Proof of Concept Trial with Re- Profiled Drug Data Mining Put Text Here Your Text Here Put Text Here Your Text Here Put Text Here Your Text Here • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes here here here here here here • Download this • Download this • Download this • Download this • Download this • Download this awesome awesome awesome awesome awesome awesome diagram diagram diagram diagram diagram diagram • Bring your • Bring your • Bring your • Bring your • Bring your • Bring your presentation to presentation to presentation to presentation to presentation to presentation to life life life life life life www.slideteam.net Your Logo
- 5. Drug Discovery Process - Style 6 Traditional Drug Discovery 0.5 - 1 Year 0.5 - 1 Year 0.5 - 1 Year 2 - 3 Years 1 - 2 Years 4 - 6 Years Target Target Lead Candidate Pre-Clinical Clinical Identification Validation Identification Optimization SWITCH Drug Re-Profiling Approach 0.5 - 1 Year 0.5 - 1 Year Clinical Proof of Concept Trial with Re- Profiled Drug Data Mining Put Text Here Your Text Here Put Text Here Your Text Here Put Text Here Your Text Here • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes here here here here here here • Download this • Download this • Download this • Download this • Download this • Download this awesome awesome awesome awesome awesome awesome diagram diagram diagram diagram diagram diagram • Bring your • Bring your • Bring your • Bring your • Bring your • Bring your presentation to presentation to presentation to presentation to presentation to presentation to life life life life life life www.slideteam.net Your Logo
- 6. Drug Discovery Process - Style 6 Traditional Drug Discovery 0.5 - 1 Year 0.5 - 1 Year 0.5 - 1 Year 2 - 3 Years 1 - 2 Years 4 - 6 Years Target Target Lead Candidate Pre-Clinical Clinical Identification Validation Identification Optimization SWITCH Drug Re-Profiling Approach 0.5 - 1 Year 0.5 - 1 Year Clinical Proof of Concept Trial with Re- Profiled Drug Data Mining Put Text Here Your Text Here Put Text Here Your Text Here Put Text Here Your Text Here • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes here here here here here here • Download this • Download this • Download this • Download this • Download this • Download this awesome awesome awesome awesome awesome awesome diagram diagram diagram diagram diagram diagram • Bring your • Bring your • Bring your • Bring your • Bring your • Bring your presentation to presentation to presentation to presentation to presentation to presentation to life life life life life life www.slideteam.net Your Logo
- 7. Drug Discovery Process - Style 6 Traditional Drug Discovery 0.5 - 1 Year 0.5 - 1 Year 0.5 - 1 Year 2 - 3 Years 1 - 2 Years 4 - 6 Years Target Target Lead Candidate Pre-Clinical Clinical Identification Validation Identification Optimization SWITCH Drug Re-Profiling Approach 0.5 - 1 Year 0.5 - 1 Year Clinical Proof of Concept Trial with Re- Profiled Drug Data Mining Put Text Here Your Text Here Put Text Here Your Text Here Put Text Here Your Text Here • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes here here here here here here • Download this • Download this • Download this • Download this • Download this • Download this awesome awesome awesome awesome awesome awesome diagram diagram diagram diagram diagram diagram • Bring your • Bring your • Bring your • Bring your • Bring your • Bring your presentation to presentation to presentation to presentation to presentation to presentation to life life life life life life www.slideteam.net Your Logo
- 8. Drug Discovery Process - Style 6 Traditional Drug Discovery 0.5 - 1 Year 0.5 - 1 Year 0.5 - 1 Year 2 - 3 Years 1 - 2 Years 4 - 6 Years Target Target Lead Candidate Pre-Clinical Clinical Identification Validation Identification Optimization SWITCH Drug Re-Profiling Approach 0.5 - 1 Year 0.5 - 1 Year Clinical Proof of Concept Trial with Re- Profiled Drug Data Mining Put Text Here Your Text Here Put Text Here Your Text Here Put Text Here Your Text Here • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes • Your Text Goes here here here here here here • Download this • Download this • Download this • Download this • Download this • Download this awesome awesome awesome awesome awesome awesome diagram diagram diagram diagram diagram diagram • Bring your • Bring your • Bring your • Bring your • Bring your • Bring your presentation to presentation to presentation to presentation to presentation to presentation to life life life life life life www.slideteam.net Your Logo
- 9. All images are 100% editable in Powerpoint “Change color, size and orientation of any icon to your liking” www.slideteam.net
- 10. Ungrouping the object 2 1 3 1. Right click the object. 2. Choose Group and then Ungroup. 3. Click beside the object and drag the arrow over it. www.slideteam.net
- 11. Edit Color 2 3 1 1. Select the shape to change the color and Right click the object( click any object which you want to change color) 2. Choose Format Shape in the dialog box. 3. Choose “Fill” in the Format Shape box then “Solid” or “Gradient” depending on the appearance of the object. Change colour as shown in the picture. www.slideteam.net
