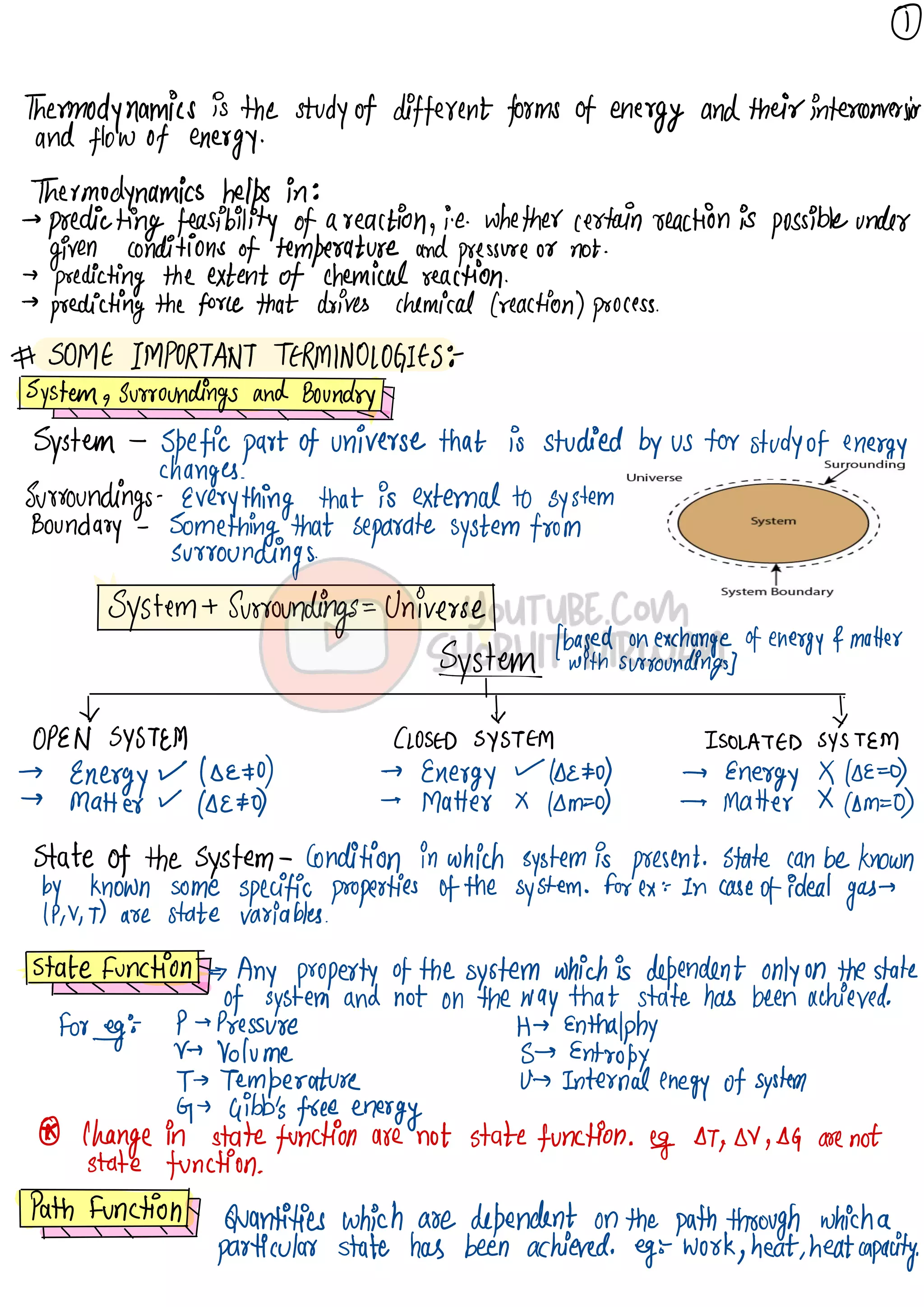
Thermodynamics is the study of energy and its transformations. It helps predict the feasibility and extent of chemical reactions based on temperature and pressure conditions. A system exchanges energy and matter with its surroundings. Thermodynamic processes include isothermal, isochoric, isobaric, and adiabatic processes. The first law of thermodynamics states that energy is conserved and the change in internal energy of a system equals heat supplied plus work done. Enthalpy is a state function that is the sum of a system's internal energy and pressure-volume work, and its change is a measure of heat absorbed or released at constant pressure.










![④
k3④ It Fs also possible to calculate enthalpy of reaction using
bond enthalpy .
Do HEEbond enthalpy
otreaastnts-Zbondenthpafopgqot.LI
:
-
calculate the enthalpy change CAH) of the following reaction .
GHzCg) t
Iz 021g) - 2 Coz Cg) t HD Cg)
Given bond enthalpies of various bonds:-
Dc-
H
HE 414 KS mot
'
Bec HE 814 KJmot
'
Do H-0=499 KT molt
A ⇐o
H-0=724 KJ mot
'
SH:-
Caegtiation tf
, elaborate af, D
Ao- HH-0=640 KImom
(H -
CE C -
H
)t
Z (0=0) → 2 (o =
⇐o
) t CH-
o-
H
)
As we know,
DrHo -
-
fsumotboenadeeannthglpiesf-
(sum of bond enthalpies
of of products ]
=
Hbc-
HH't BecHo -
IIDo H-9-
14 D⇐otto -12 Do-
H Hof
=
(2x 414 t 810T
Ex 499) -
(4×724+2×460) KT
=
(2885.5-3816) KJ
= -
930.5 KJ
←
# Enthalpy of solution , Dsoe Ho or
↳The enthalpy change when one mole of solute is dissolved completely in
specific amount of solvent or water is called enthalpy of solution.
°
It solvent Ps in excess i.e. the interactions between the ions too solute
molecules ) are
negligible then the enthalpy change is called enthalpy of solution
at infinite dilution.
°
Aoe H =
Dealtice Hot Dhyd Ho](https://image.slidesharecdn.com/thermodynamicspart1byshobhitnirwan-230228015935-e9b410d7/75/Thermodynamics-Part-1-by-Shobhit-Nirwan-pdf-13-2048.jpg)
