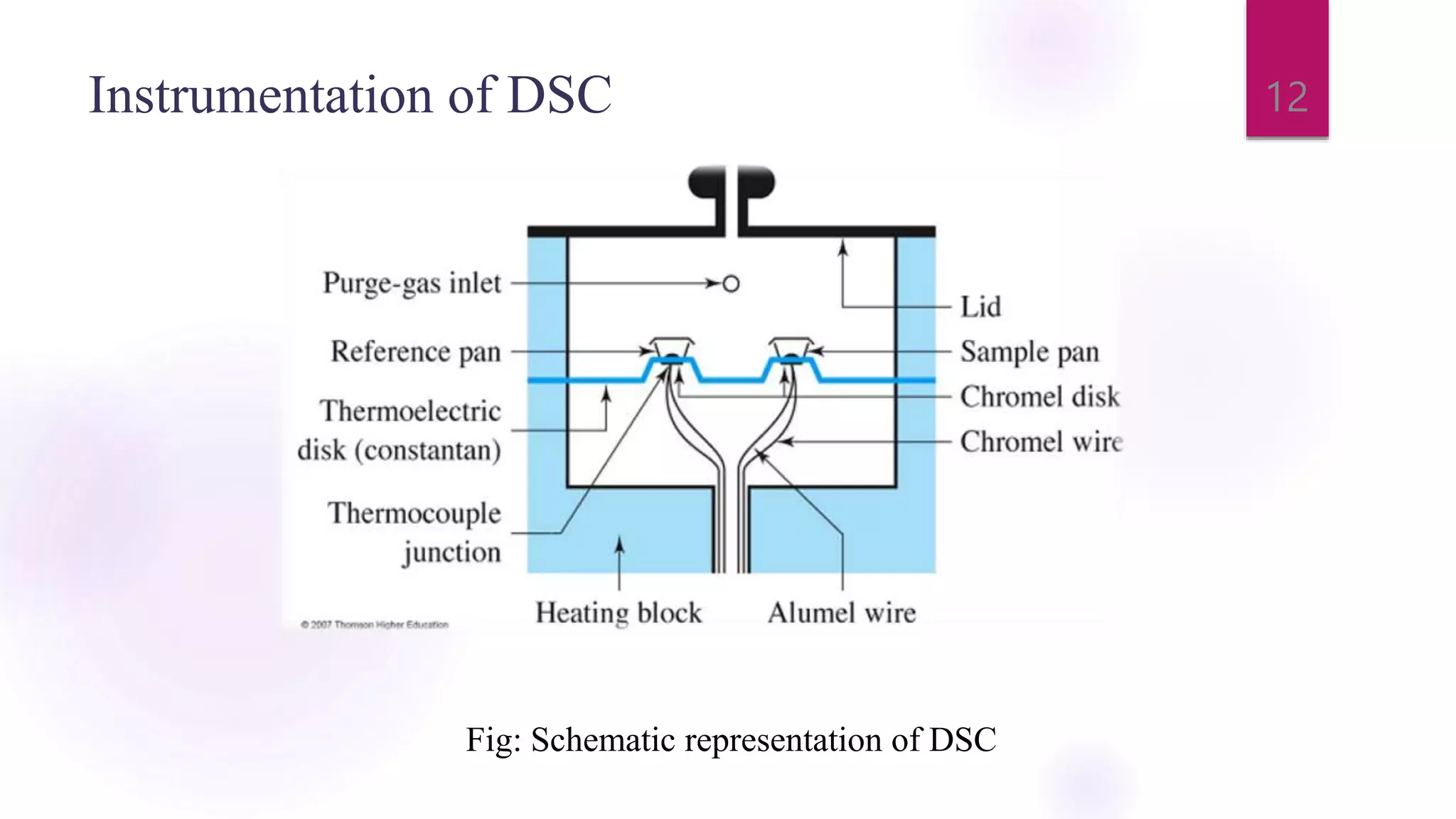
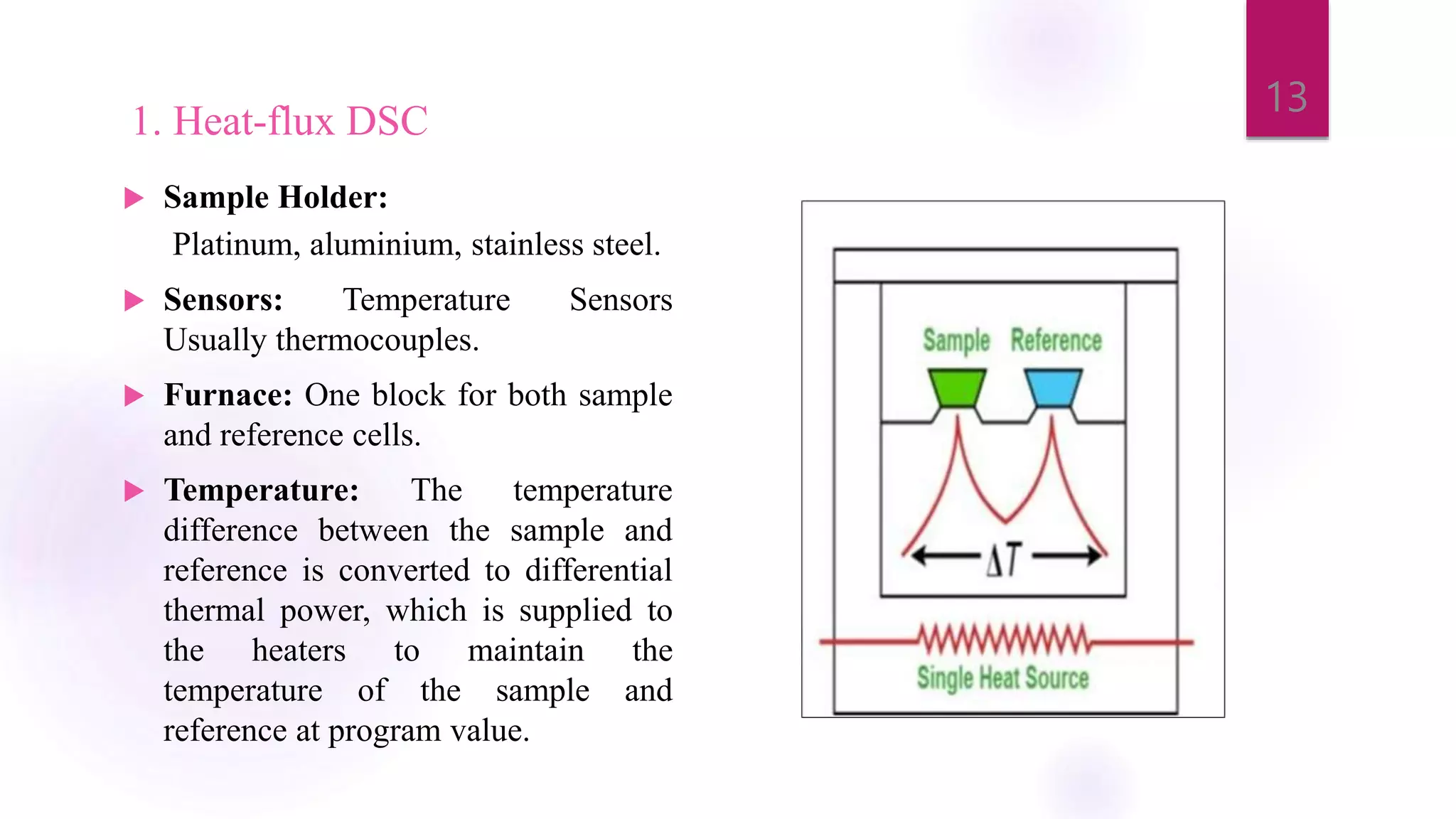
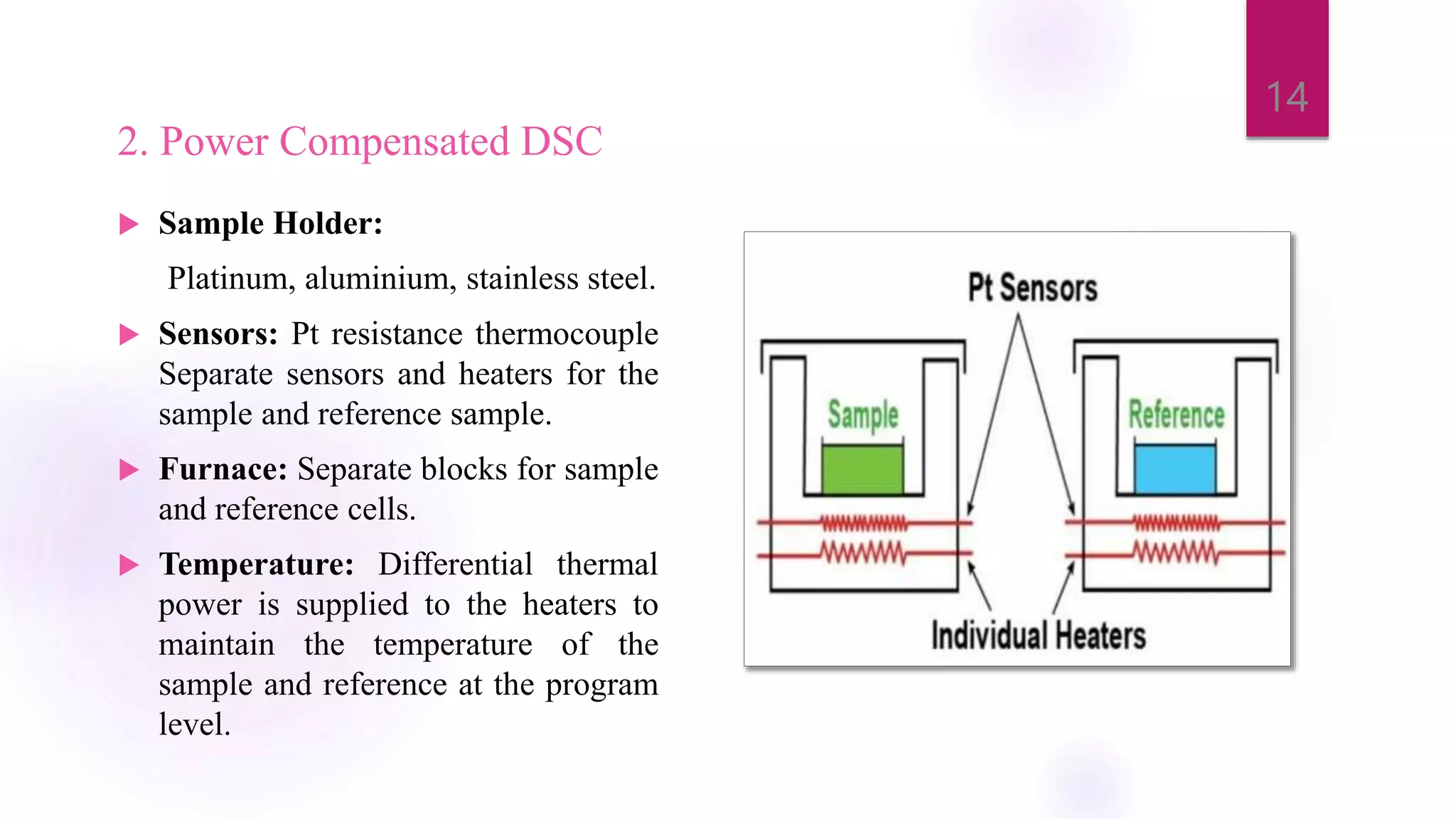
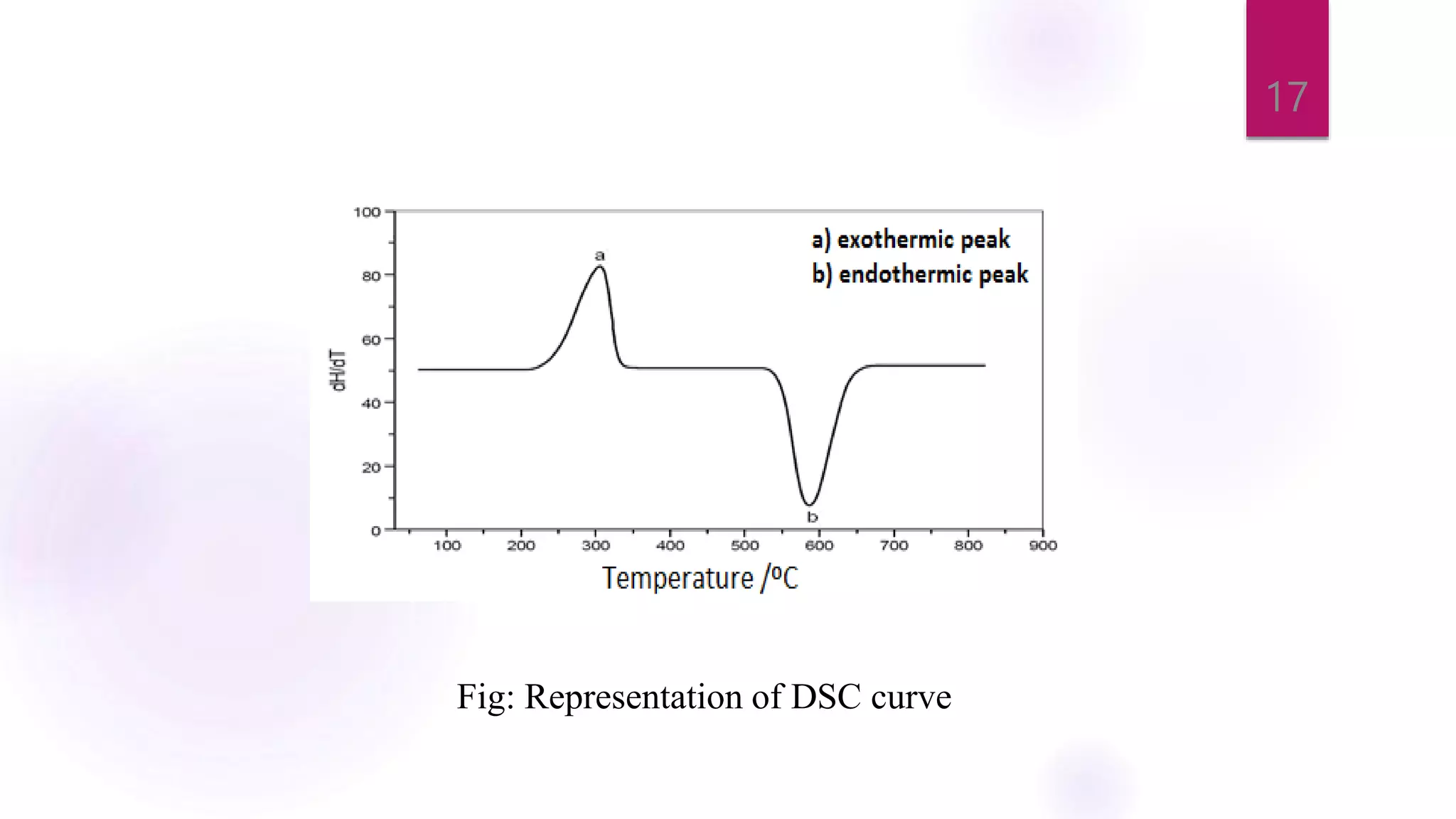
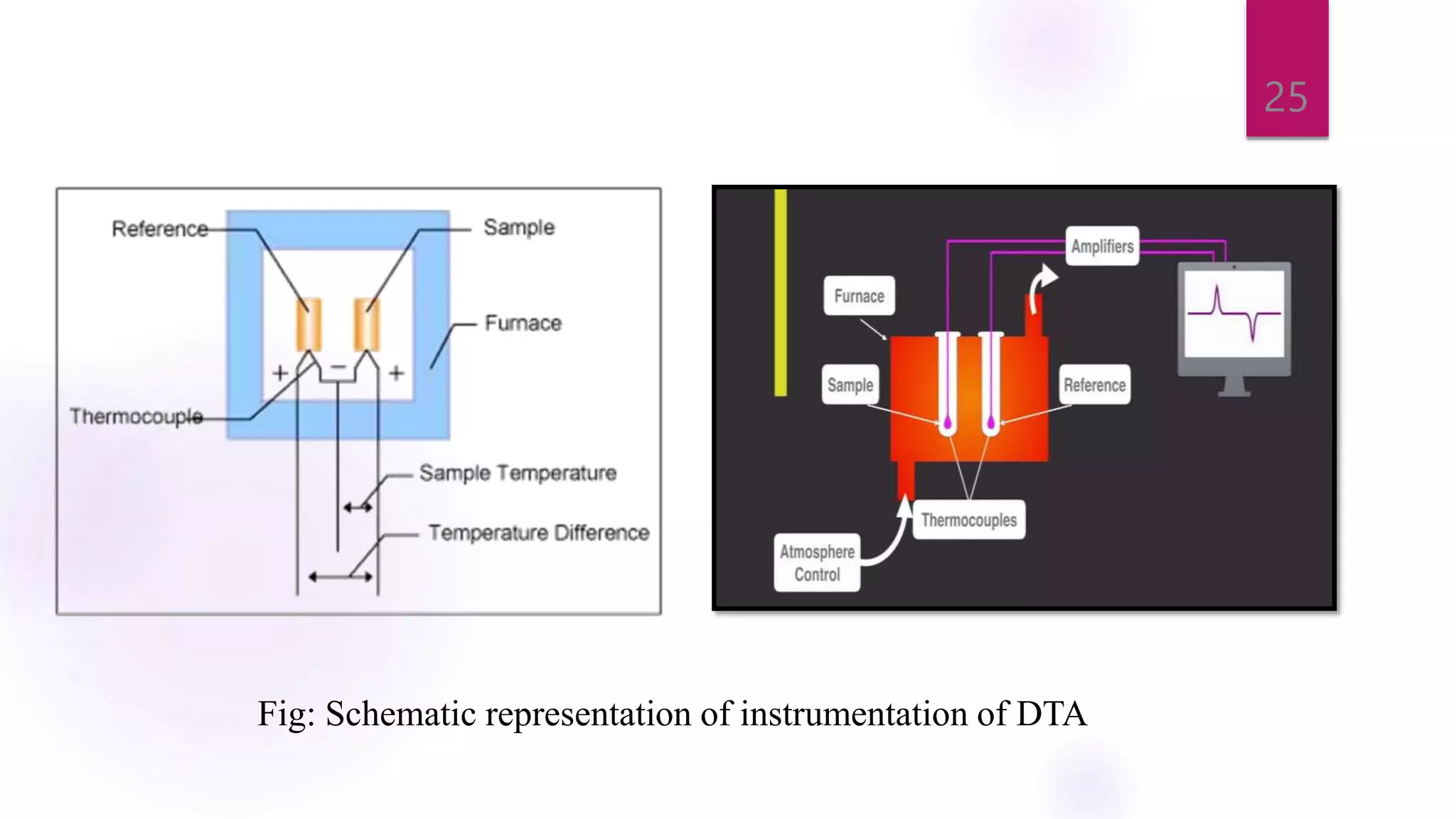
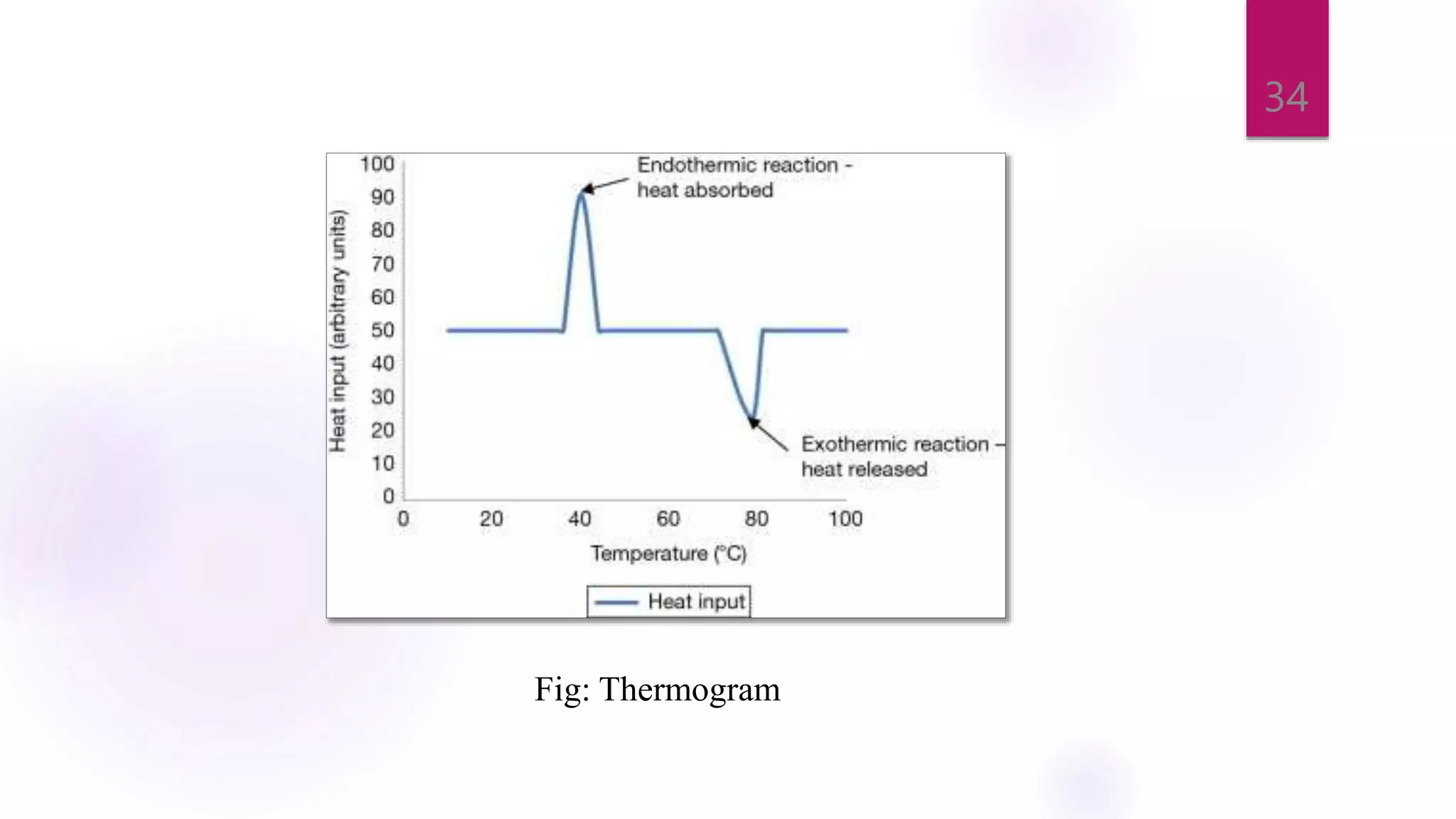
The document provides a comprehensive overview of thermal techniques, specifically focusing on Differential Scanning Calorimetry (DSC) and Differential Thermal Analysis (DTA). It discusses principles, instrumentation, advantages, disadvantages, and applications of these techniques in analyzing material properties as a function of temperature. Key methods include measuring heat differences in thermal reactions, with applications in purity determination, stability studies, and characterization of various materials.