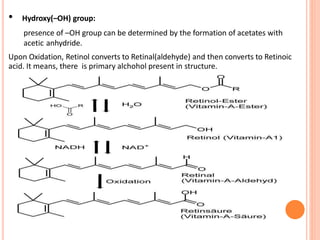
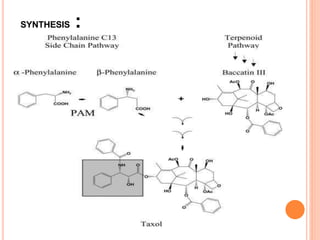
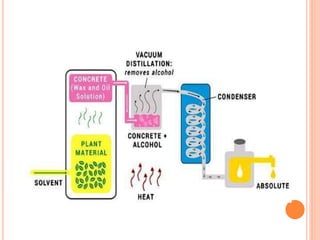
Terpenoids are a class of naturally occurring organic chemicals derived from five-carbon isoprene units. This document provides an introduction and overview of terpenoids, including their general properties, methods of isolation from plants, classification based on the number of isoprene units, and common analytical techniques used for structural elucidation such as determining functional groups, unsaturation, and the number of rings in the structure.





















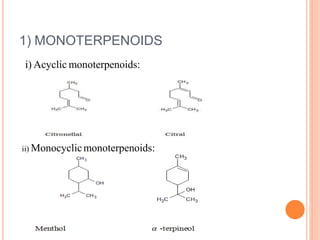
































![• Menthol was converted to p-Cymene [1-methyl-4-
isopropylbenzene], which was also obtained by dehydrogenation of
pulegone.
• Pulegone on reduction yields menthone which on further reduction
gives menthol.](https://image.slidesharecdn.com/tersli-200212113201/85/TERPENOIDS-57-320.jpg)