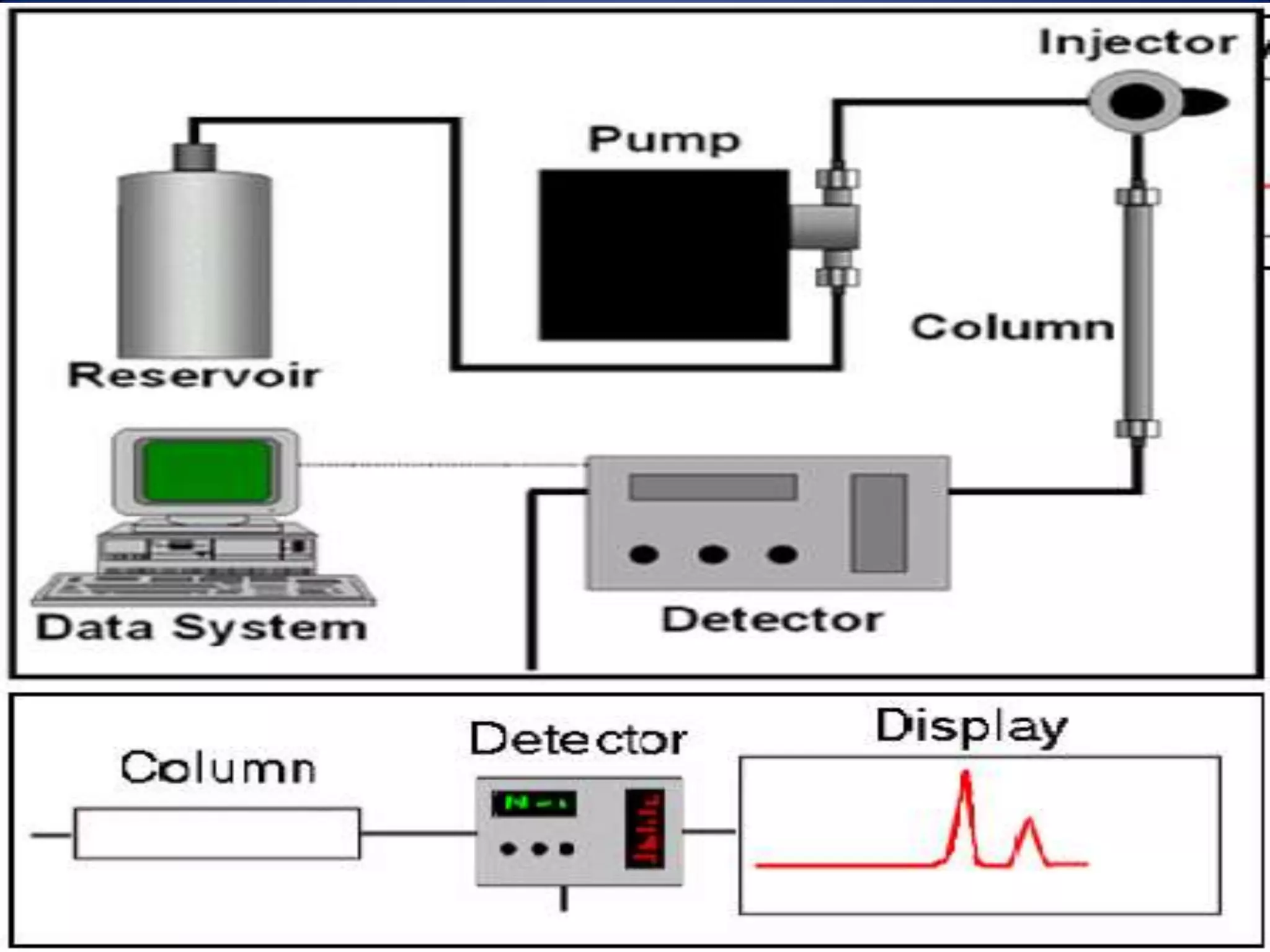
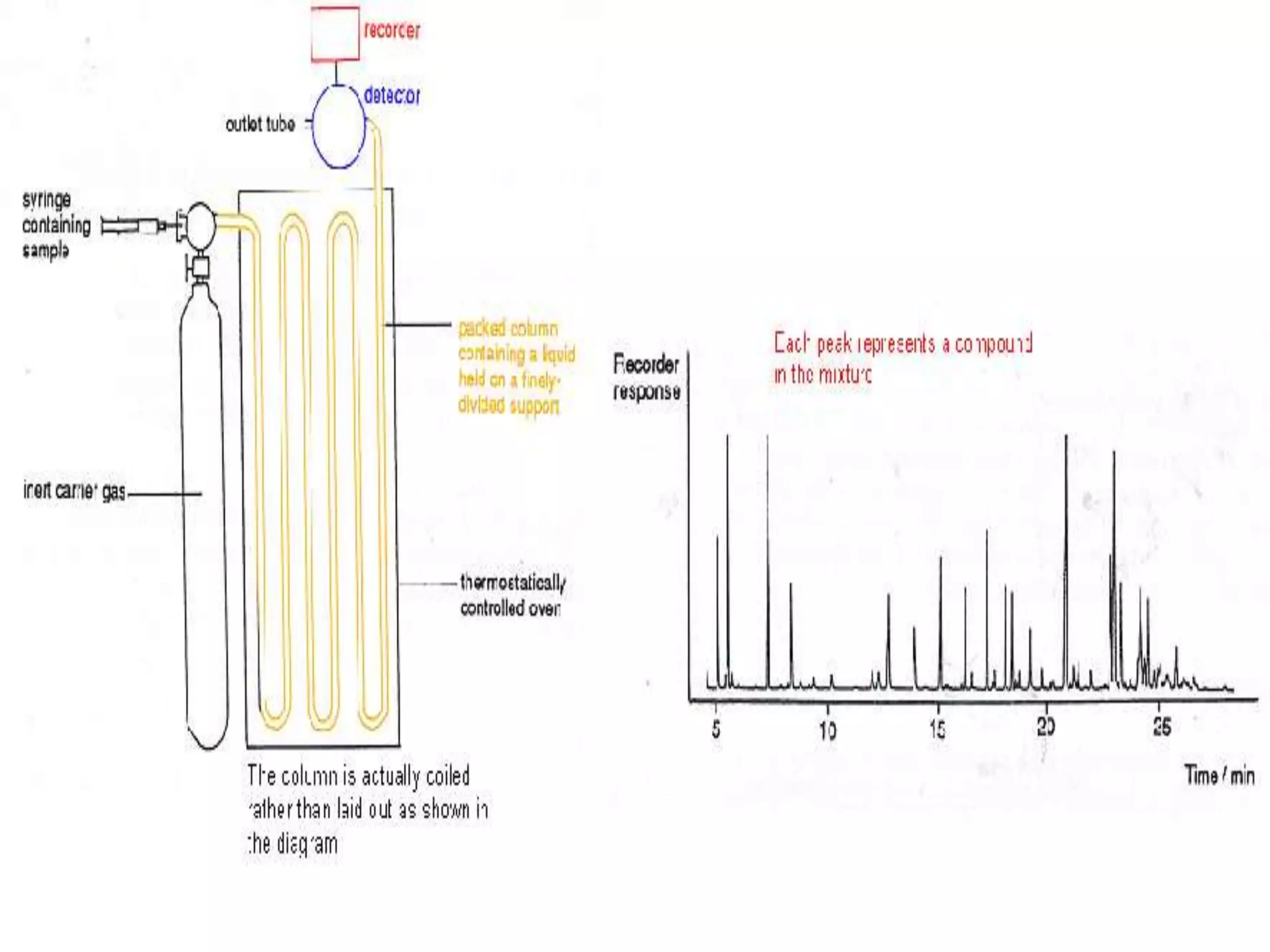
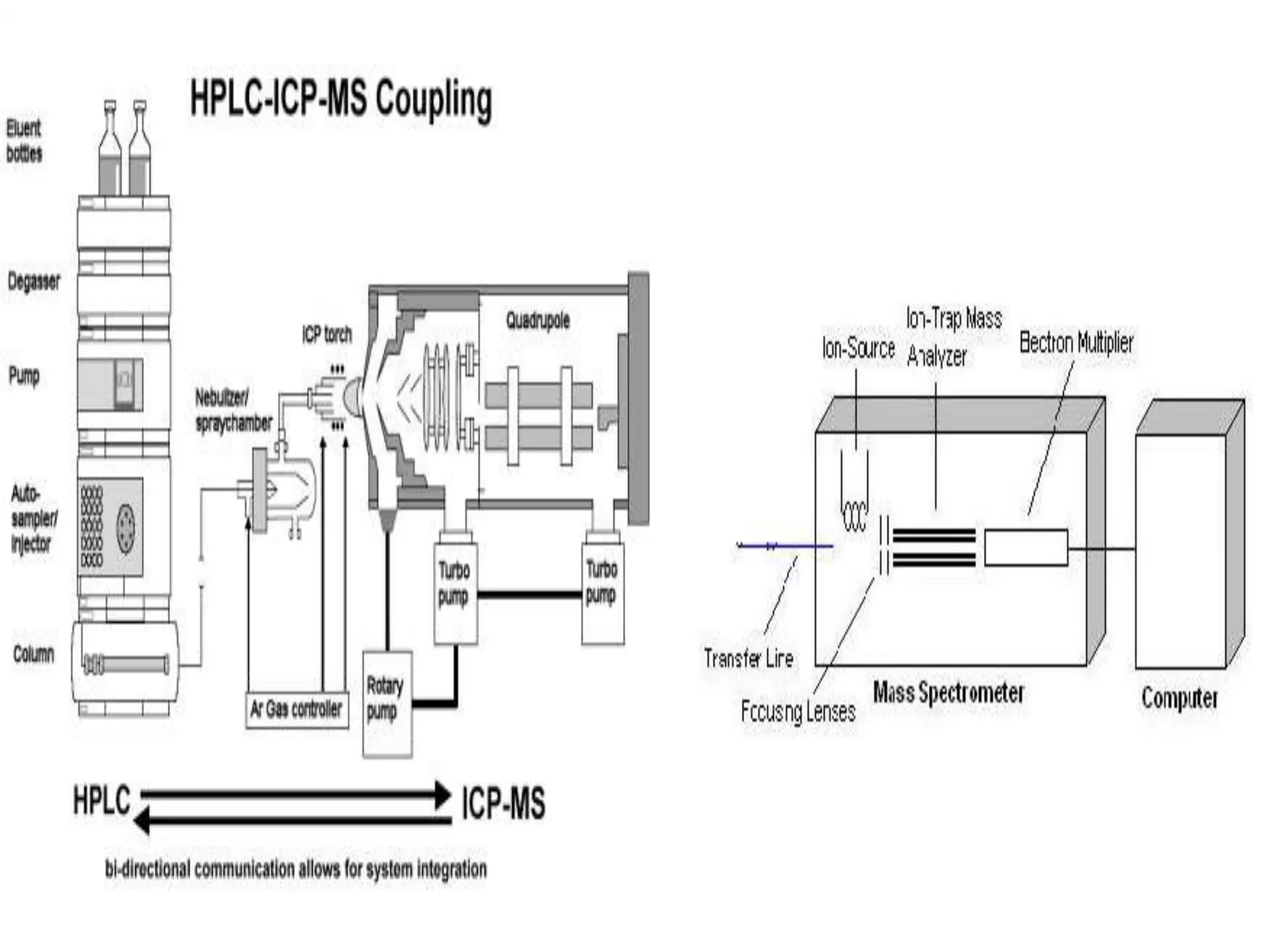
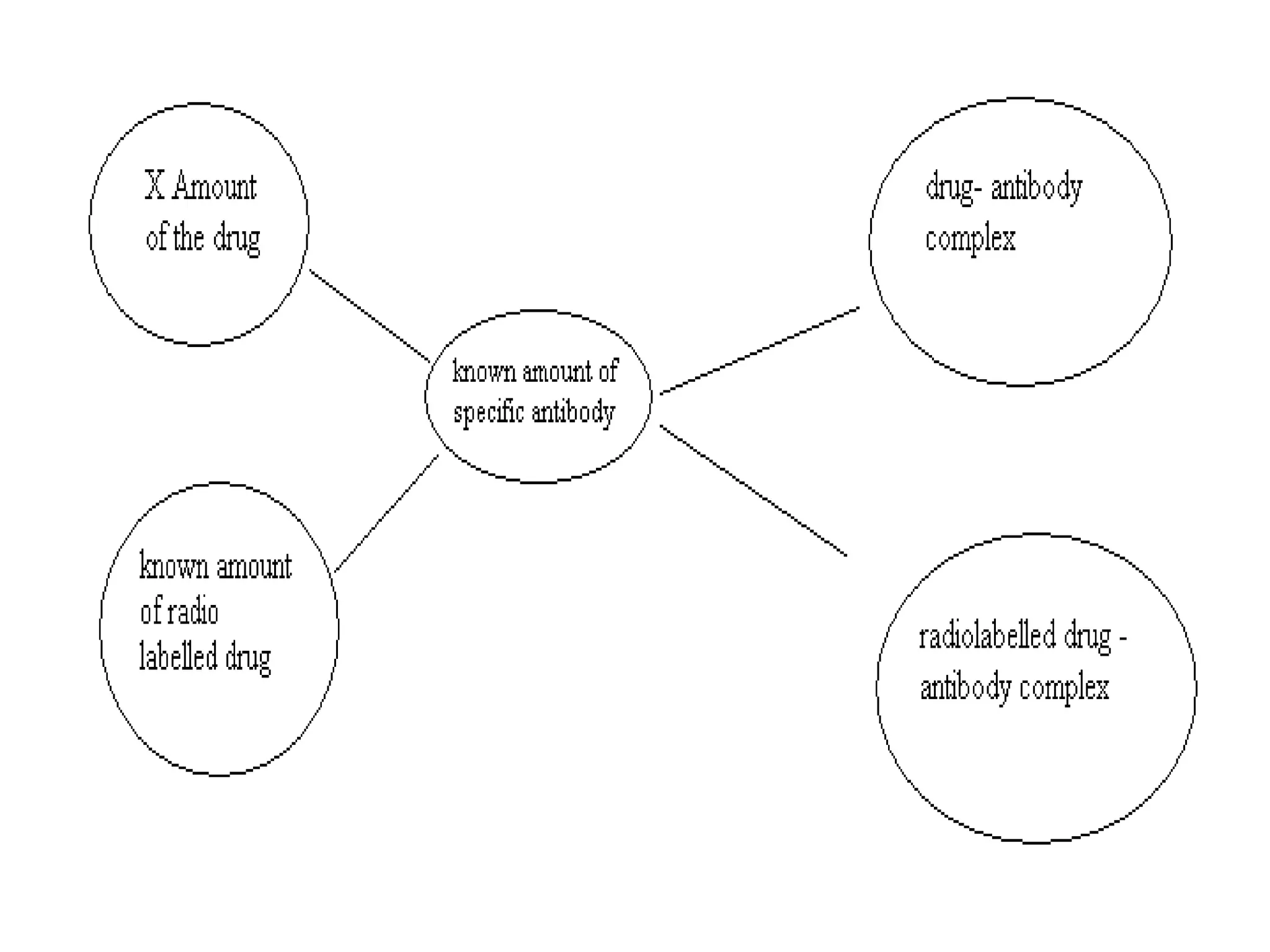
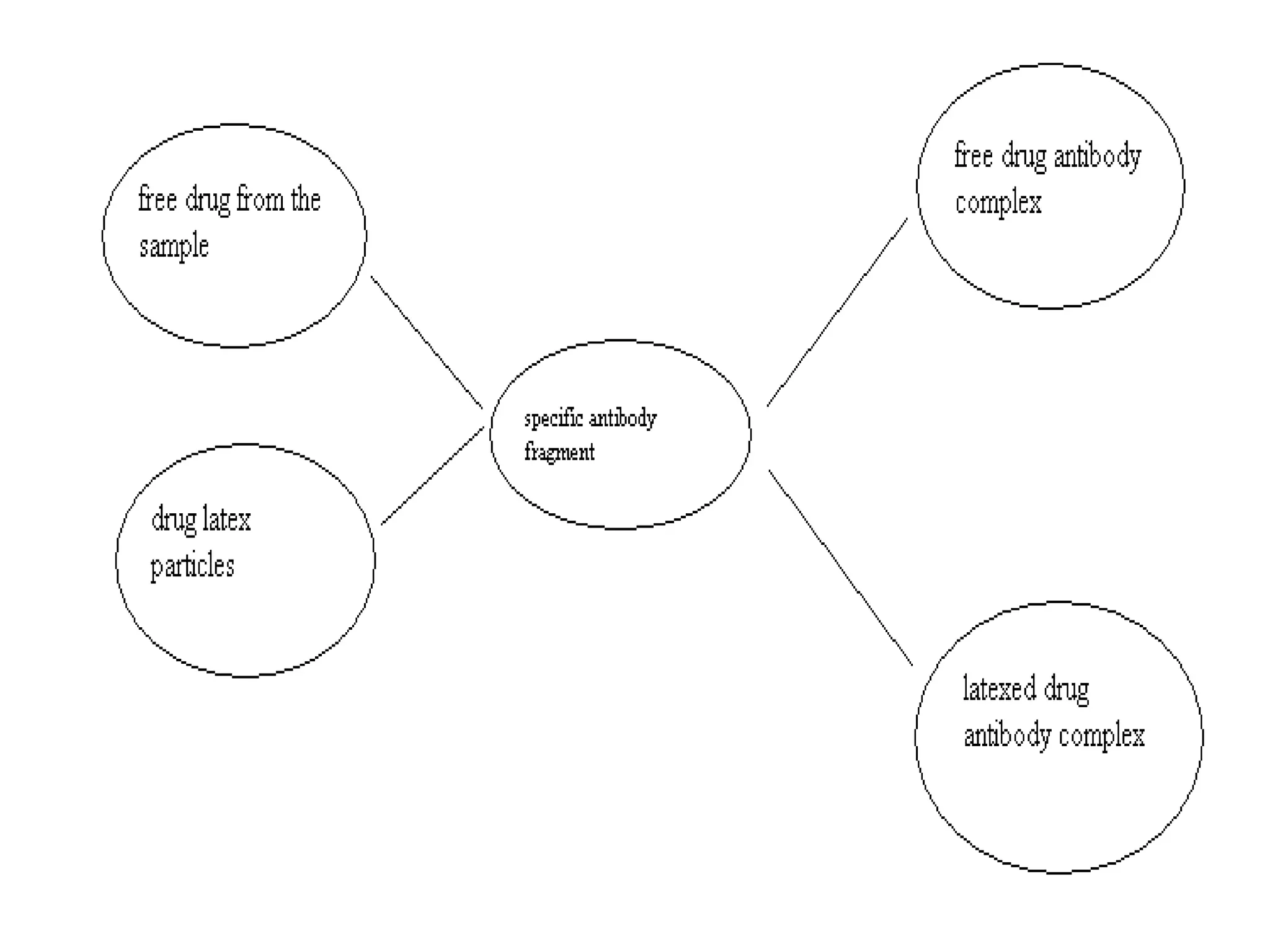
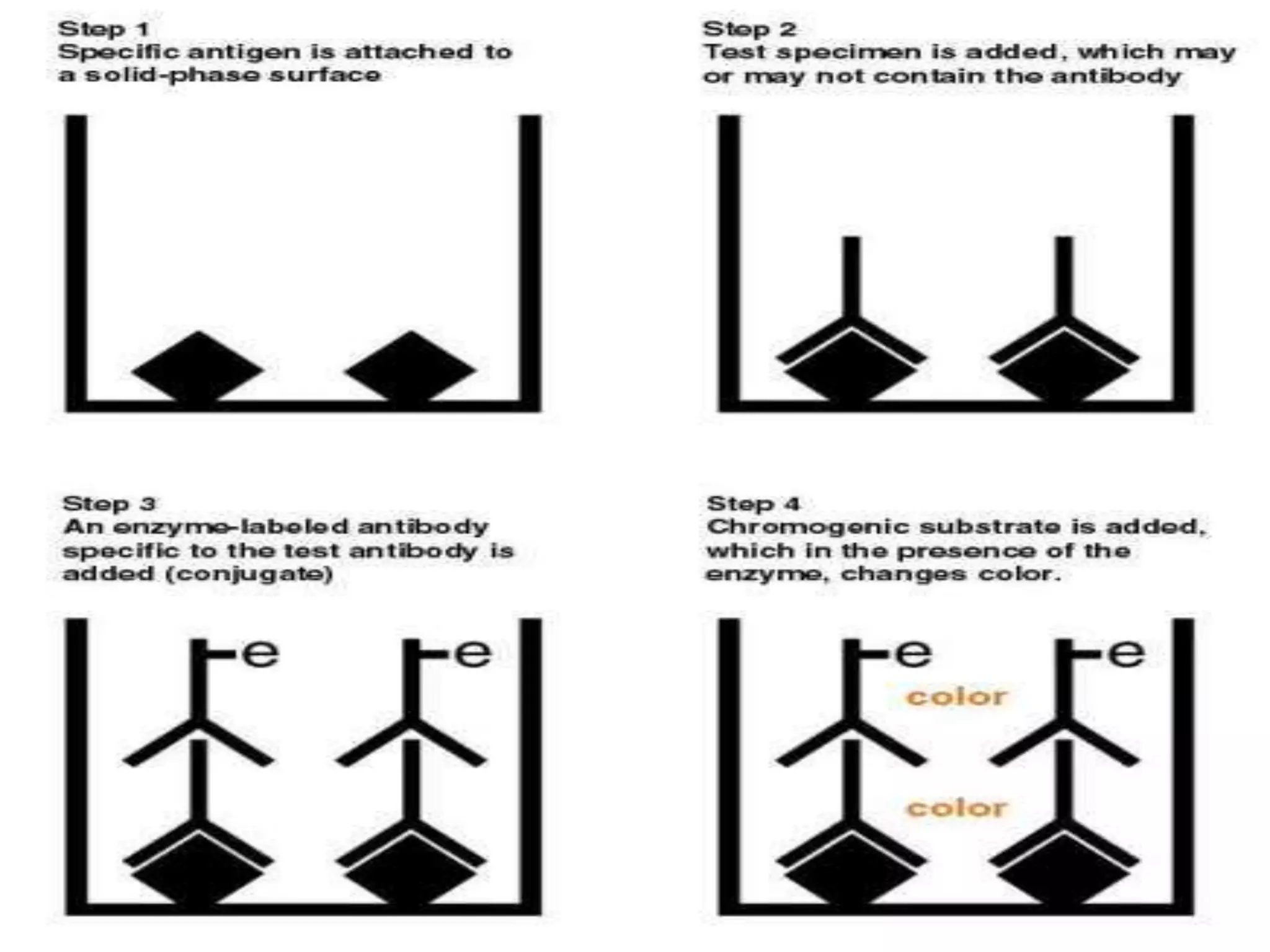
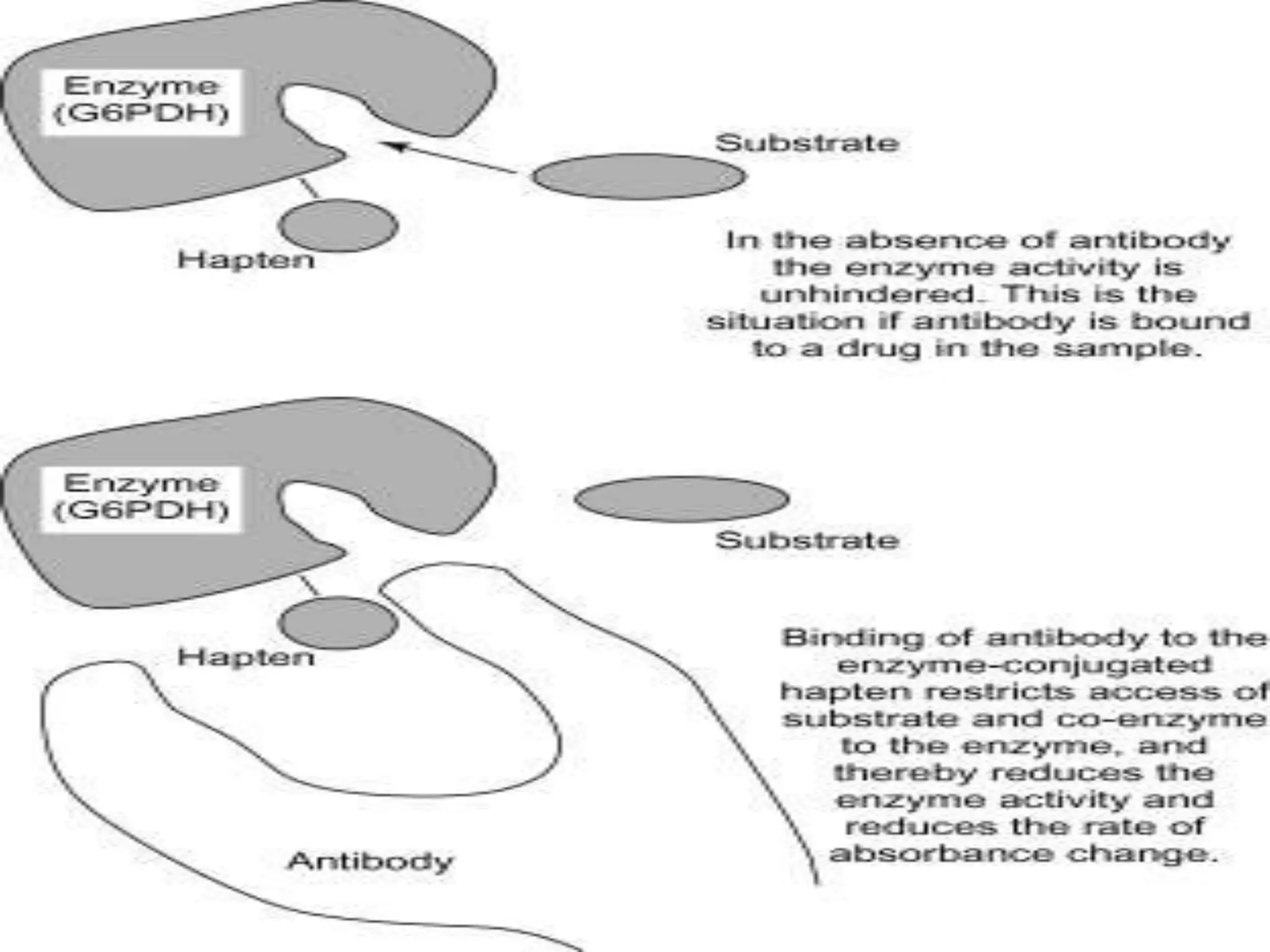
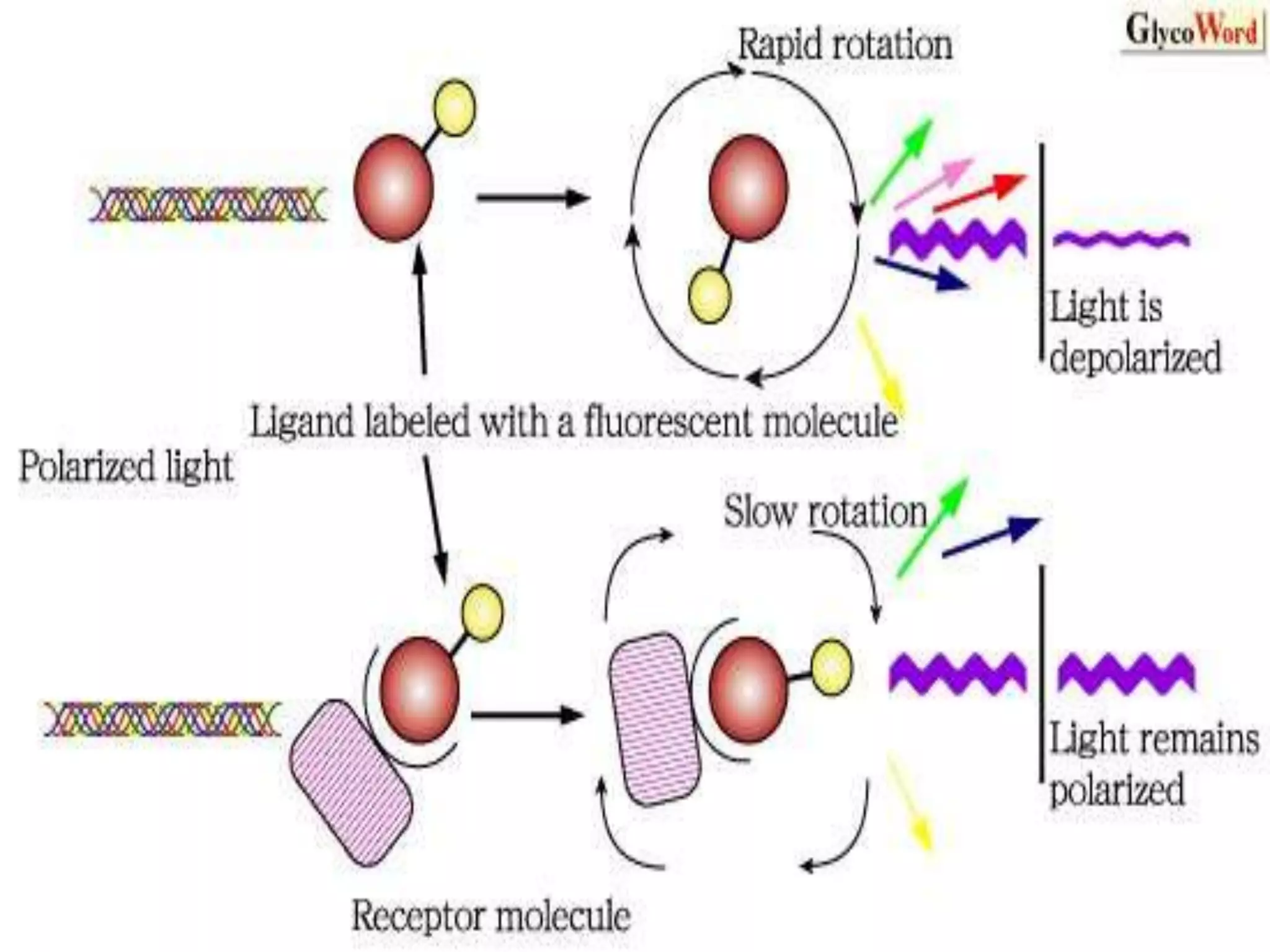
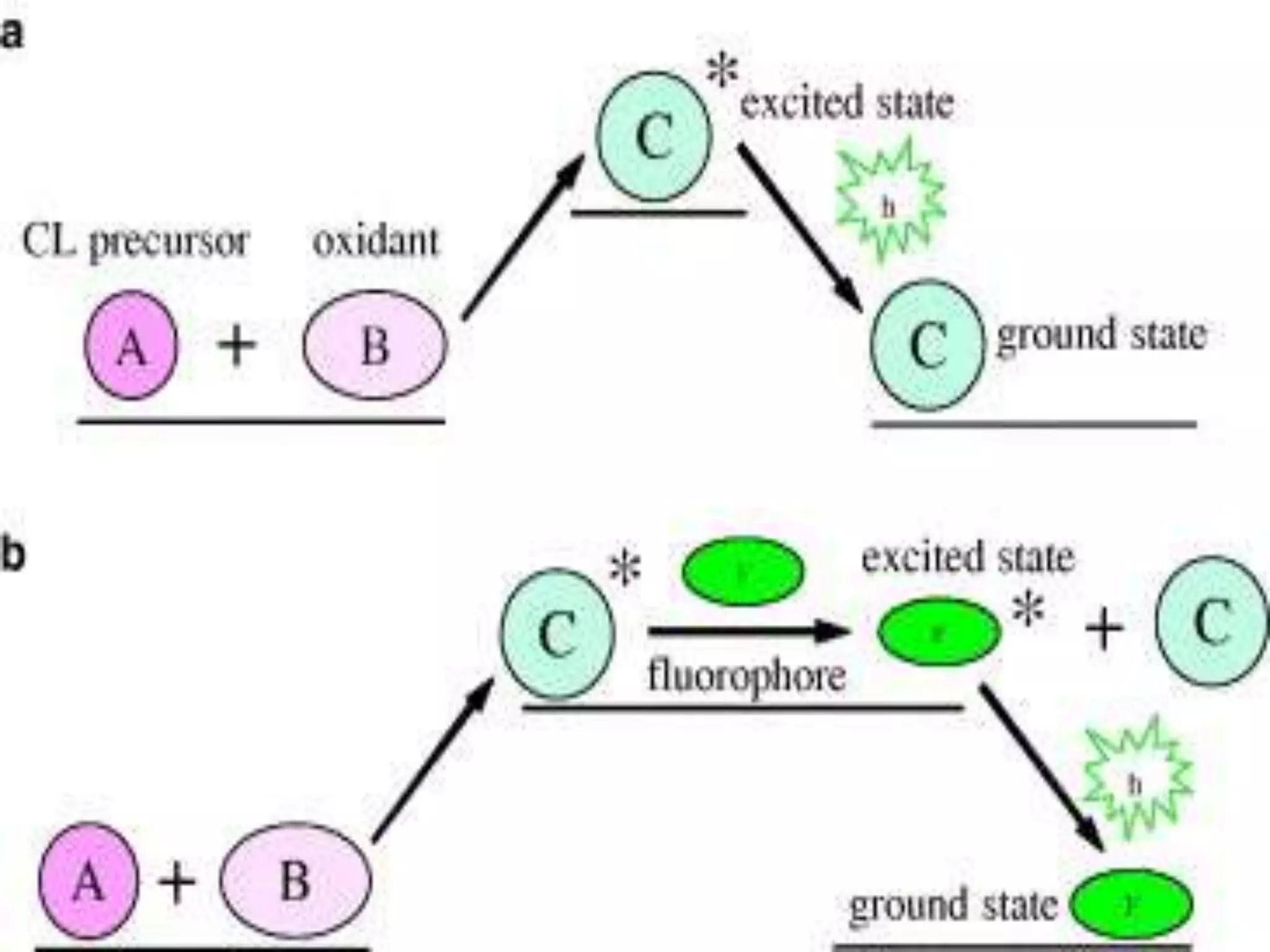
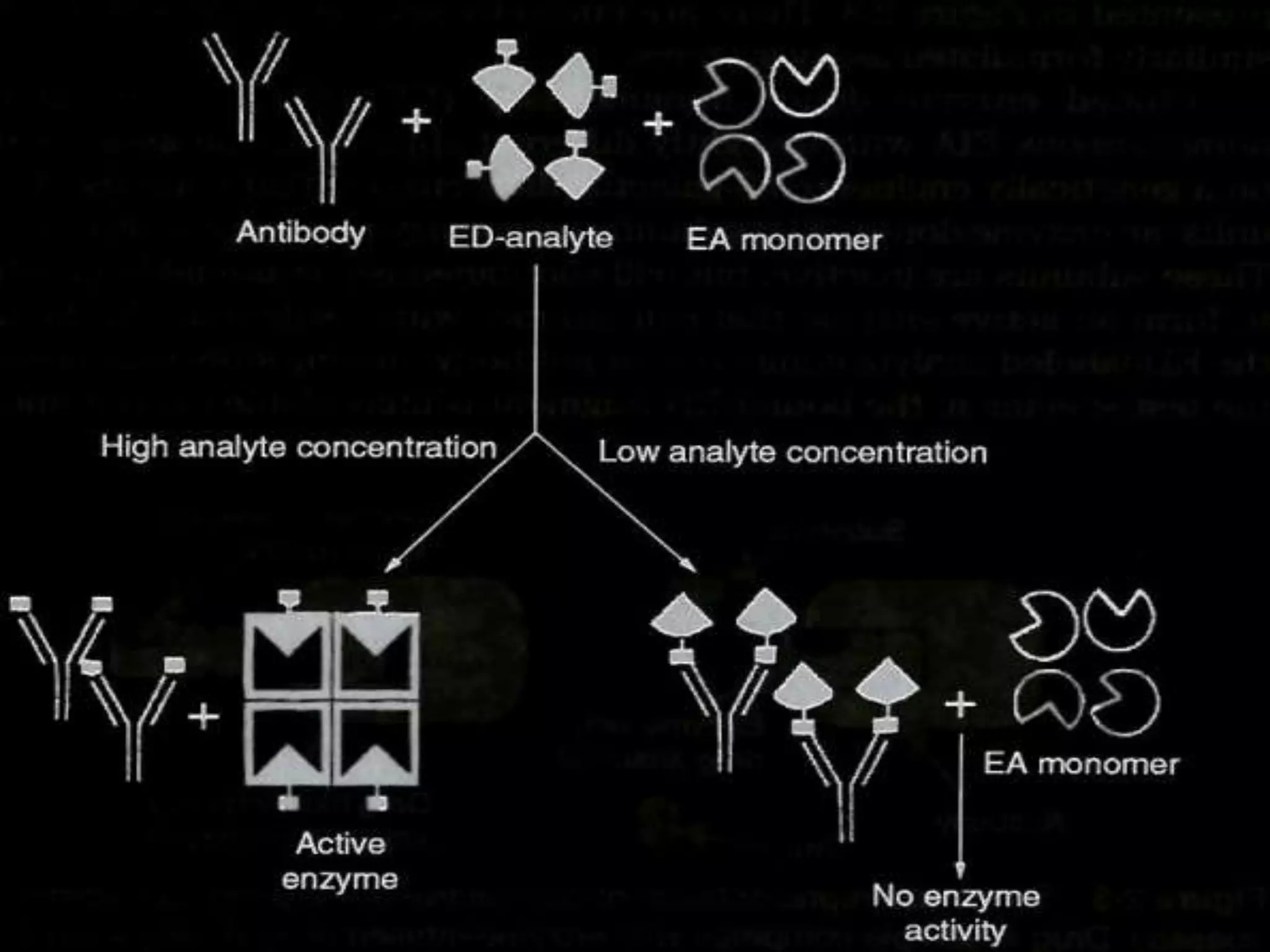
This document discusses several techniques for drug analysis including chromatography methods like high performance liquid chromatography (HPLC), gas chromatography (GC), and liquid chromatography-mass spectrometry (LC-MS). It also summarizes various immunoassay techniques such as radioimmunoassay, particle enhanced turbidimetric inhibition immunoassay, enzyme immunoassay, enzyme multiplied immunoassay technique, fluorescence polarization immunoassay, and chemiluminescence assays. Additional methods covered are affinity chrome-mediated immunoassay, and cloned enzyme donor immunoassay.