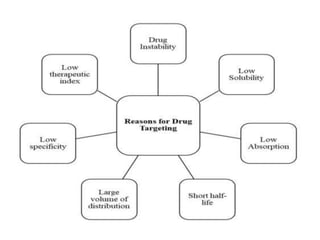
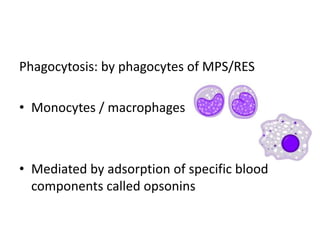
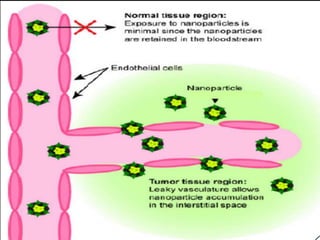
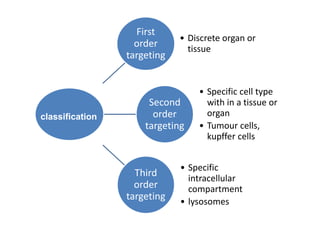
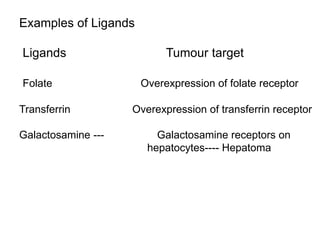
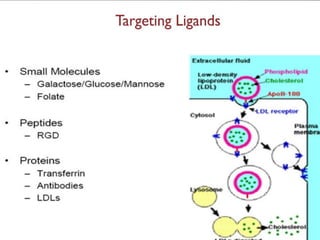
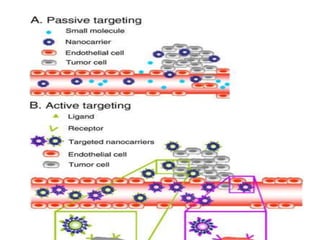
This document discusses targeted drug delivery systems. It defines targeted drug delivery as selectively delivering a pharmacologically active agent only to its site of action, not non-target tissues. Targeted delivery implies selectively localizing the drug at a pre-identified target site in a therapeutic concentration while restricting access to normal cells. This minimizes side effects and maximizes efficacy. Common approaches to targeted delivery include incorporating drugs into carrier systems, altering drug structure, or controlling drug release. Ideal targeted systems are non-toxic, biodegradable, stable, and controllably release drug at the target site.