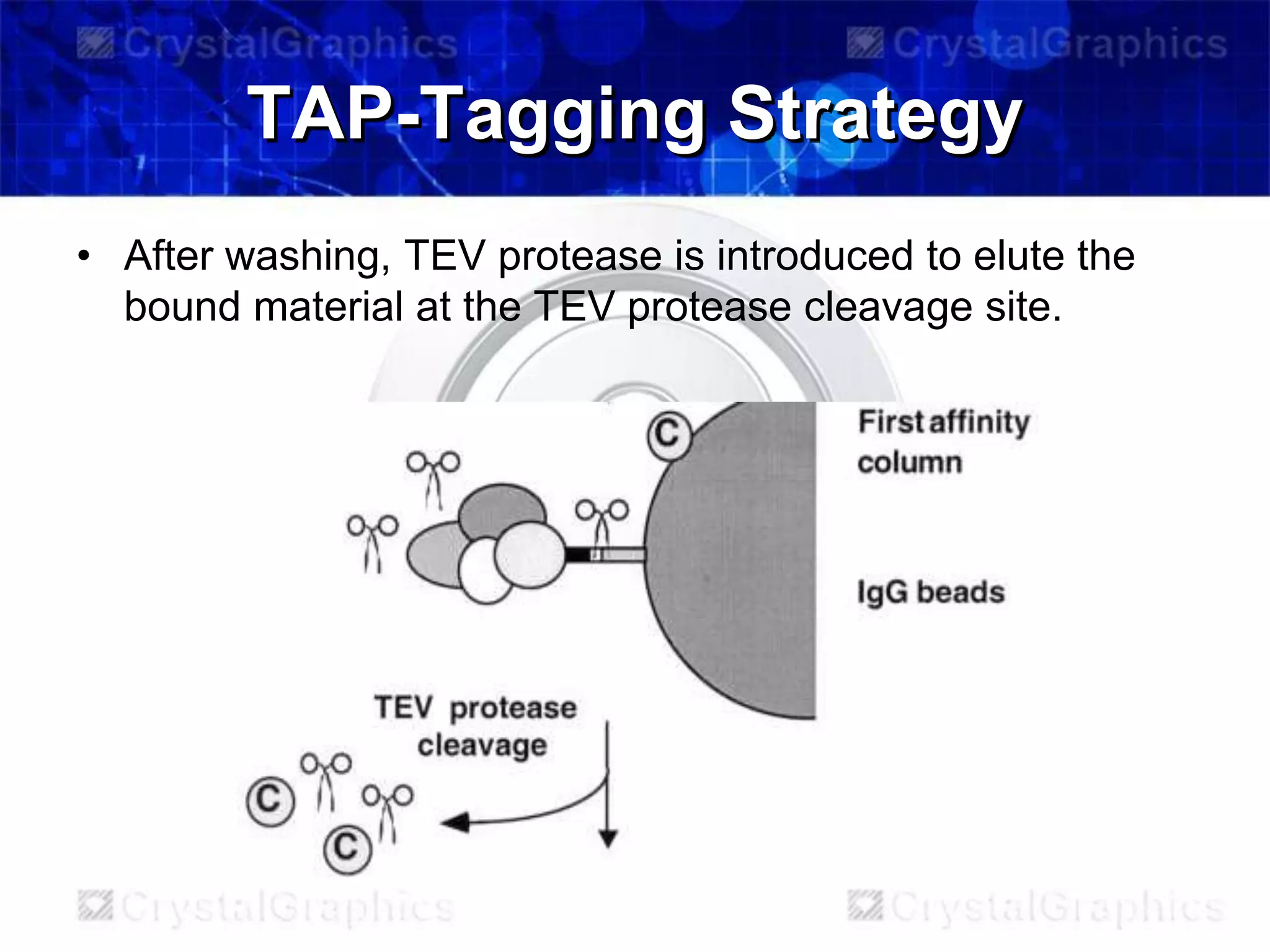
TAP-tagging is a protein purification technique that involves creating a fusion protein with a TAP tag to study protein-protein interactions. The TAP tag contains protein A and a calmodulin binding peptide separated by a TEV protease site. This allows two-step affinity purification of associated protein complexes under physiological conditions. TAP-tagging was developed in the 1990s and has been widely used to map protein interaction networks in yeast and identify new protein complexes involved in processes like pre-mRNA splicing.