Embed presentation
Download to read offline







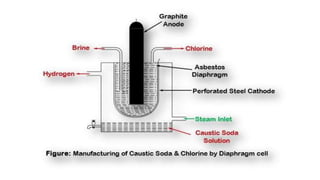



This document summarizes the commercial production process of sodium hydroxide (NaOH) through electrolysis of brine. It describes how salt is dissolved in water to create a brine solution, which is purified and then sent to diaphragm cells where electrolysis occurs. During electrolysis, sodium ions migrate to the cathode where they react with hydroxyl ions to form NaOH, while chlorine gas forms at the anode. The end products are caustic soda solution extracted from the cathode chamber and chlorine gas collected from the anode chamber.











