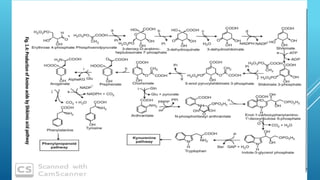
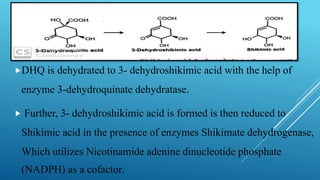
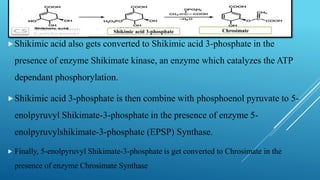
The document presents an overview of the shikimic acid pathway, a critical metabolic route found in bacteria, fungi, and plants, but absent in animals. This pathway is essential for the biosynthesis of aromatic amino acids like phenylalanine, tyrosine, and tryptophan, and involves multiple enzymatic steps that lead to the production of various phenolic compounds. Key outcomes of this pathway include the synthesis of gallic acid and other aromatic metabolites that contribute to the formation of flavonoids, lignin, and alkaloids.