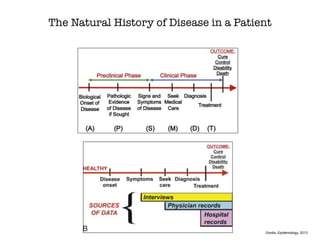
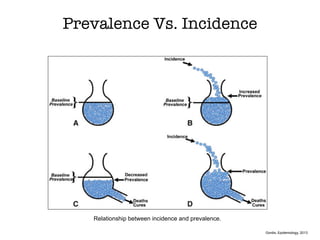
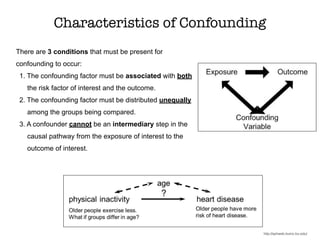
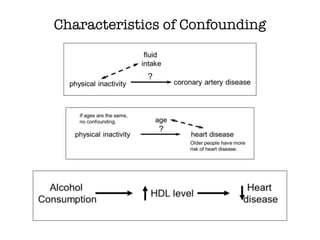
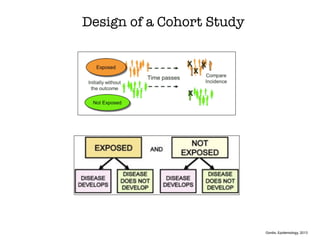
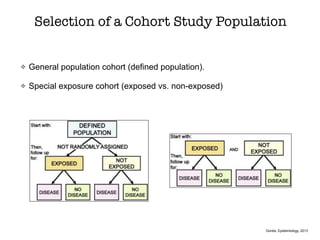
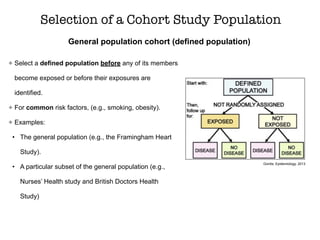
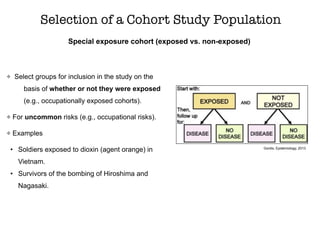
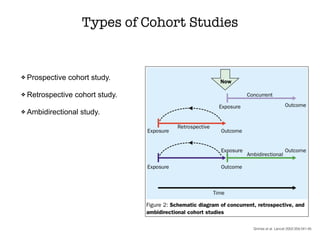
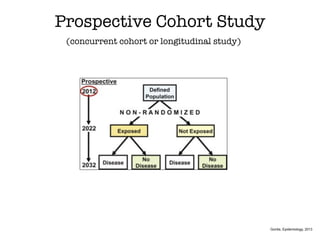
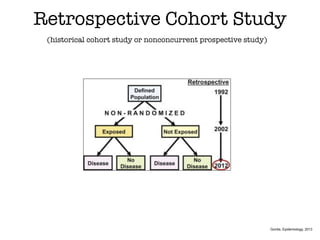
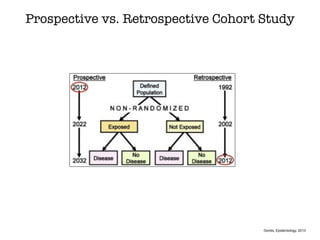
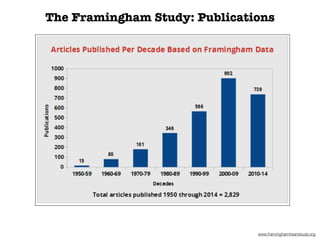
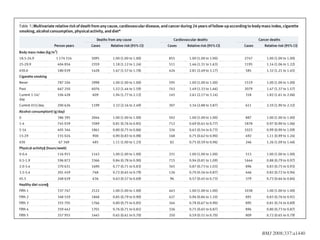
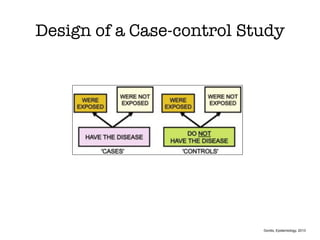
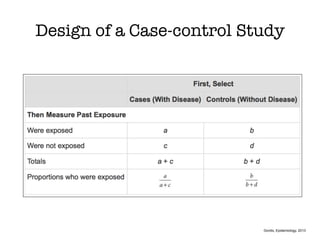
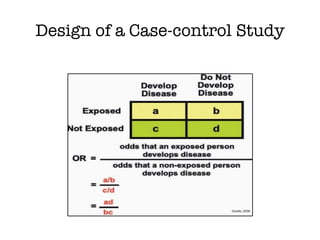
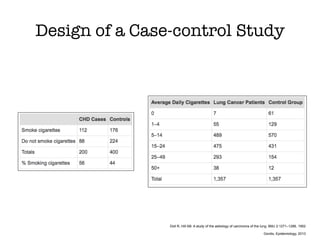
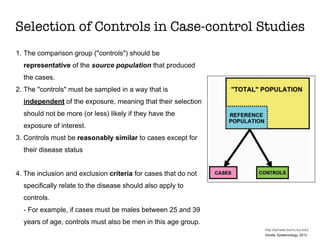
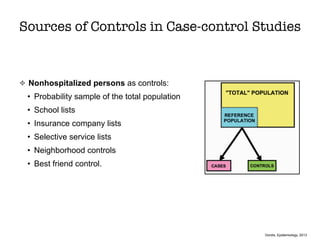
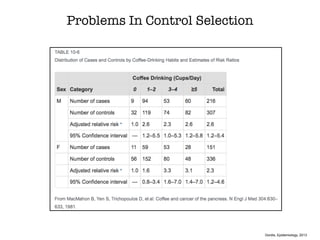
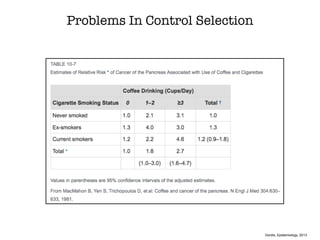
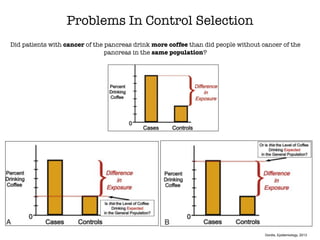
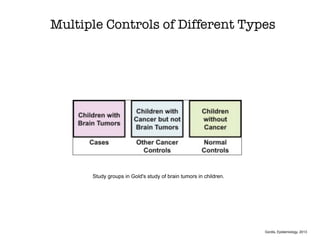
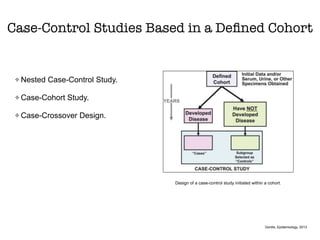
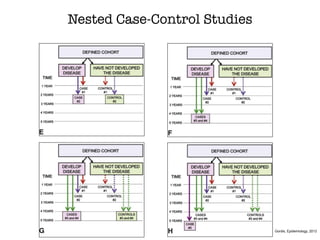
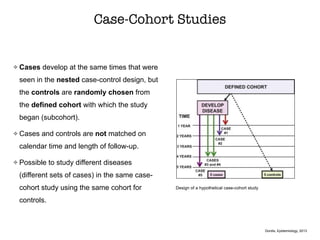
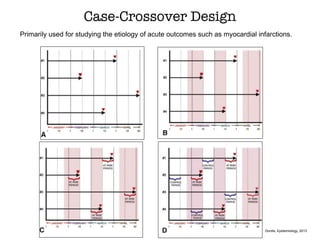
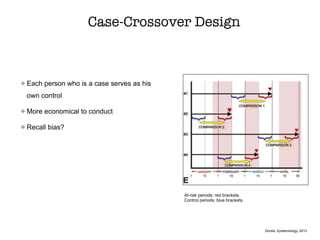
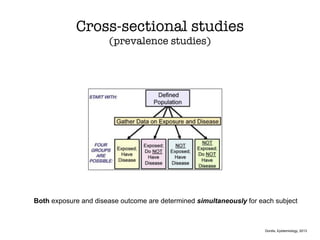
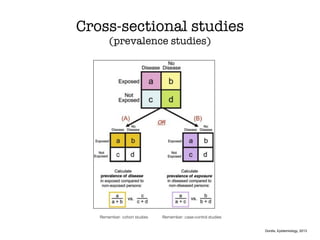
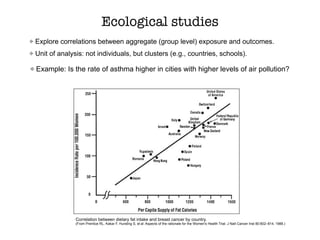
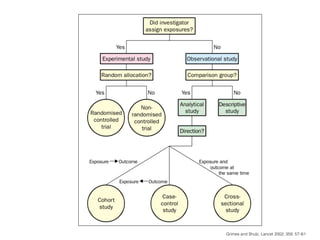
This document discusses various observational study designs used in epidemiology, including cohort and case-control studies. It provides details on how to design, conduct, and analyze these types of studies. For cohort studies, it explains how to select the cohort population and comparison groups, and describes examples like the Framingham Heart Study and Nurses' Health Study. For case-control studies, it outlines how to select cases and controls and discusses potential biases. The document emphasizes the importance of temporal sequence between exposure and outcome and minimizing biases and confounding when using these observational study designs.