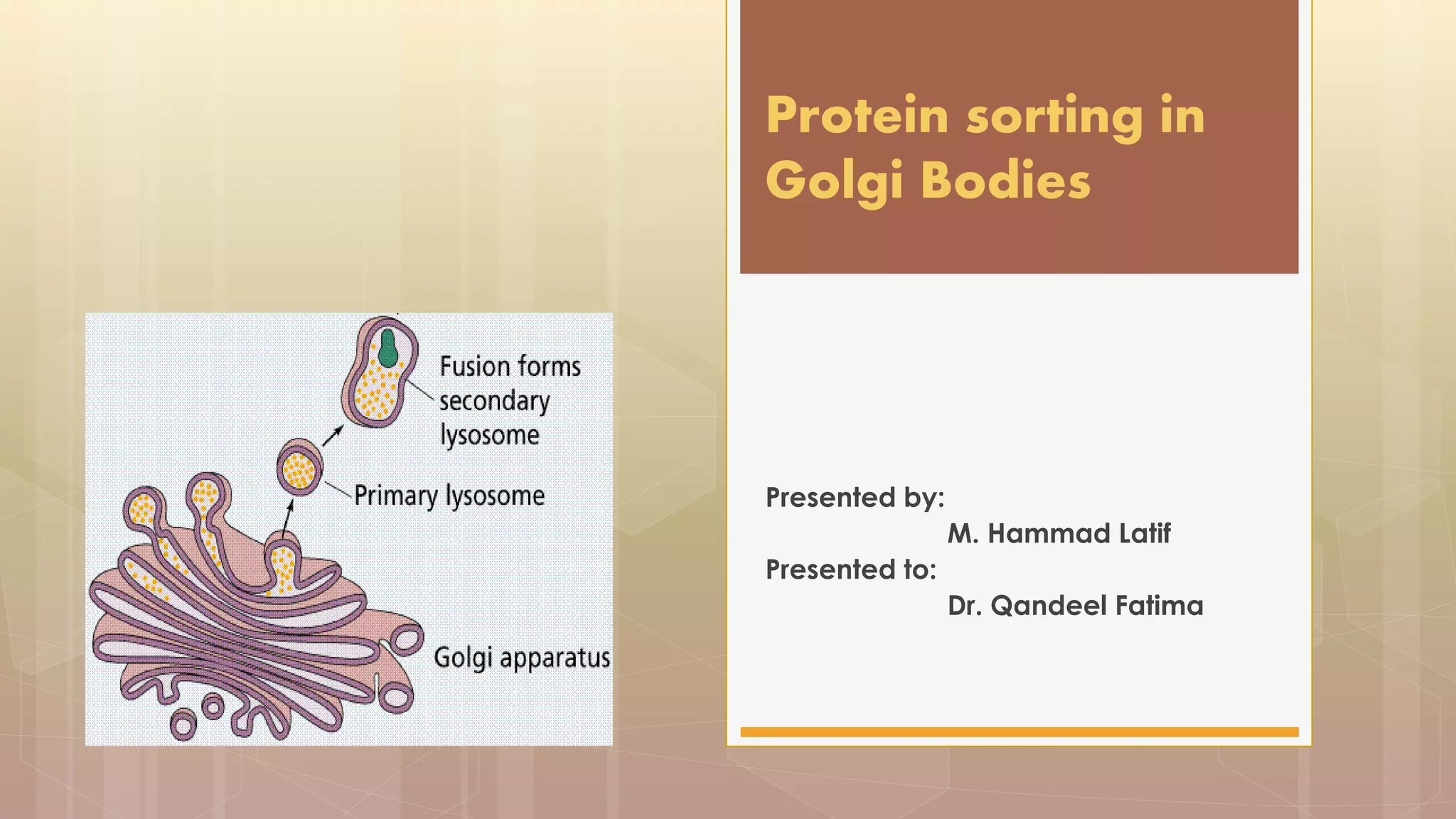
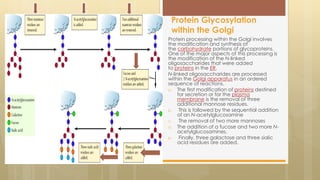
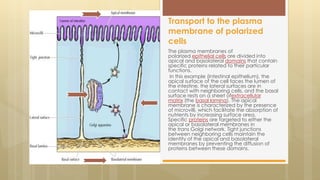
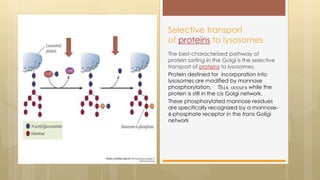
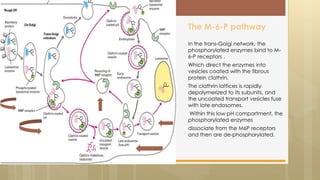
The document discusses protein sorting in Golgi bodies. It describes how proteins are modified and sorted as they pass through the cis, medial, and trans faces of the Golgi apparatus. Proteins undergo processing like glycosylation and are targeted to their final destinations, such as organelles, vesicles, or secretion. The Golgi apparatus plays a key role in modifying and sorting proteins to their correct locations within and outside the cell.