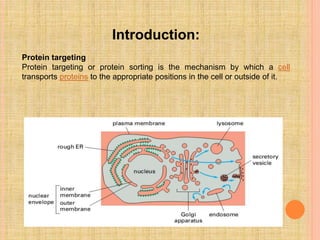
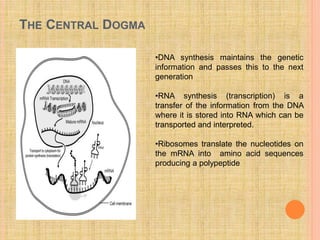
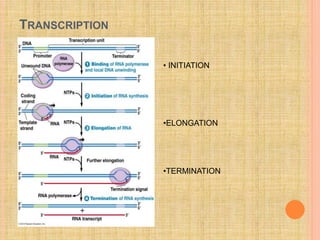
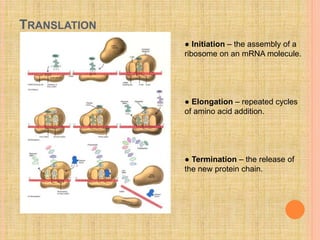
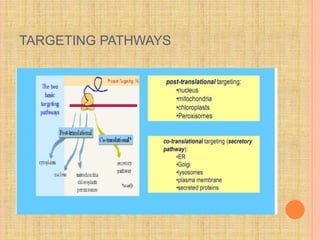
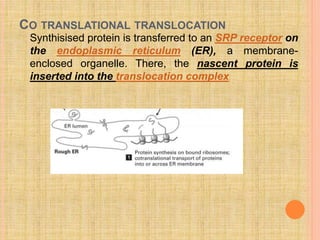
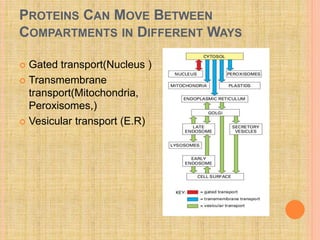
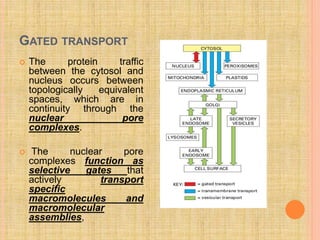
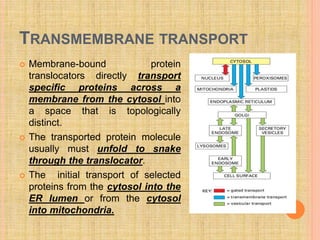
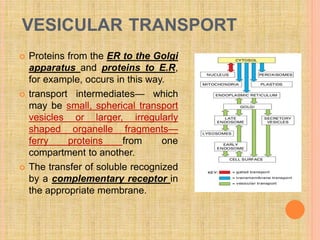
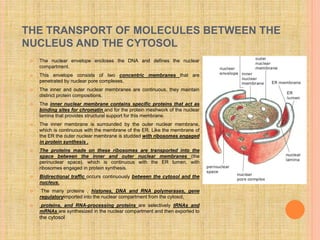
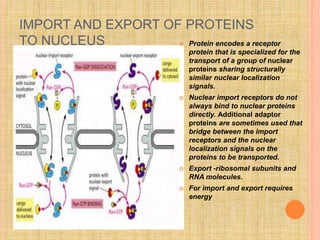
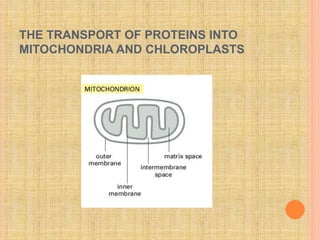
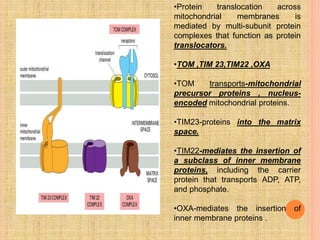
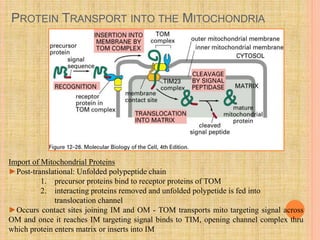
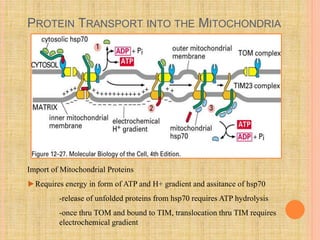
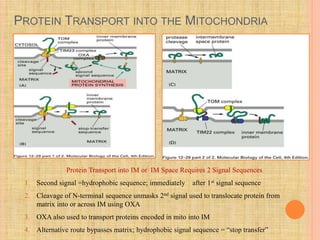
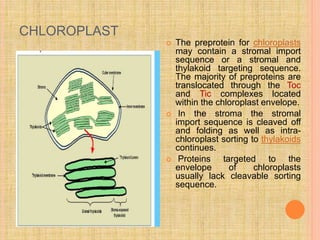
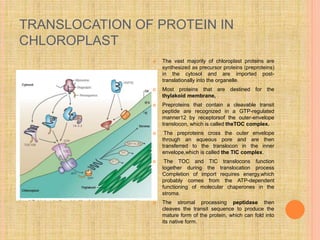
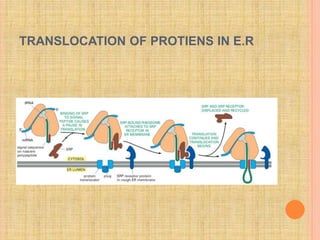
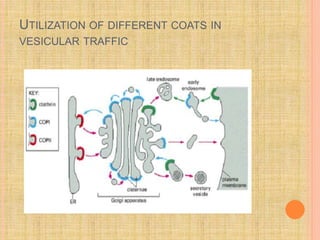
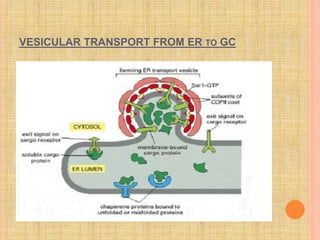
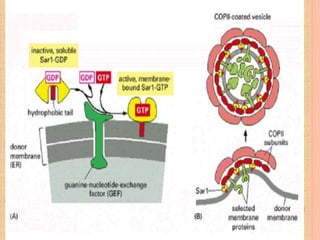
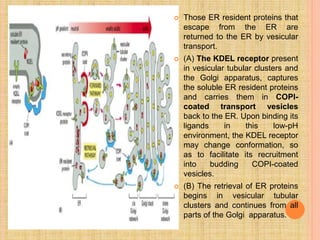
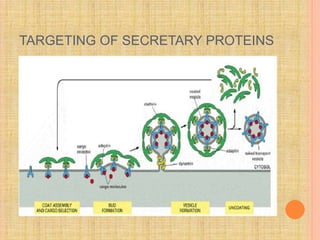
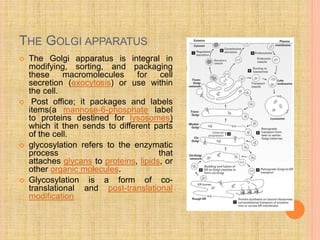
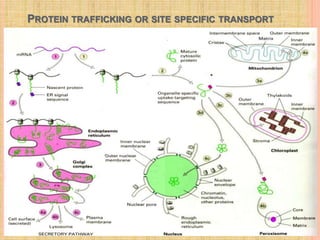
The document discusses protein targeting and sorting, which is essential for the proper distribution of newly synthesized proteins within cells, with processes varying between prokaryotes and eukaryotes. Key points include the role of signal sequences in directing proteins to their destinations, protein translocation mechanisms through membranes, and the functions of the endoplasmic reticulum and Golgi apparatus in processing and transporting proteins. It also highlights the energy requirements for these transport processes and references significant contributions in the field, including those leading to Nobel Prize recognition.