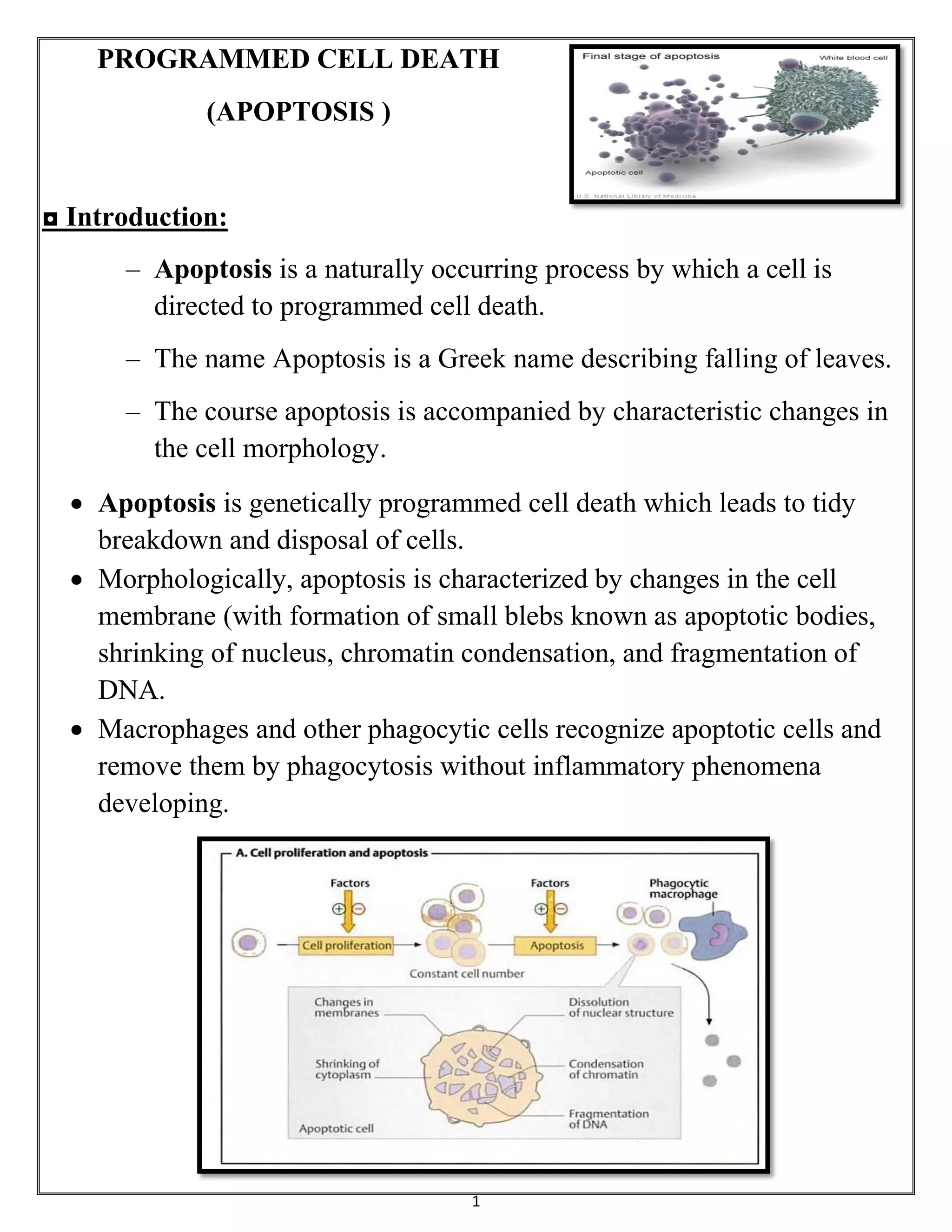
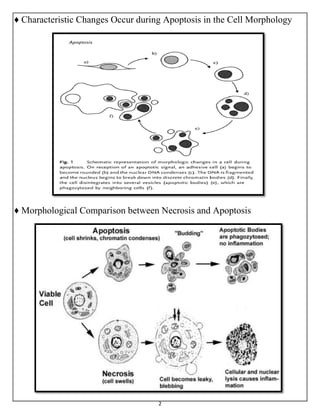
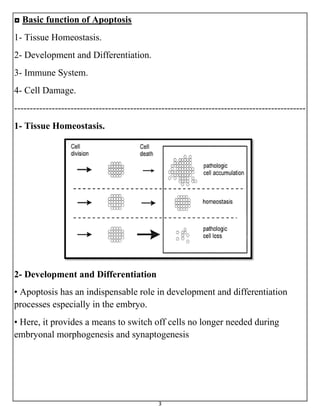
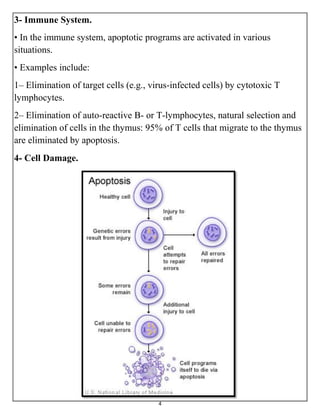
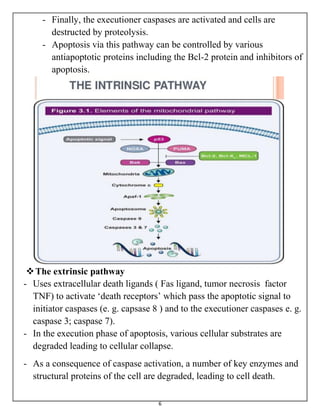
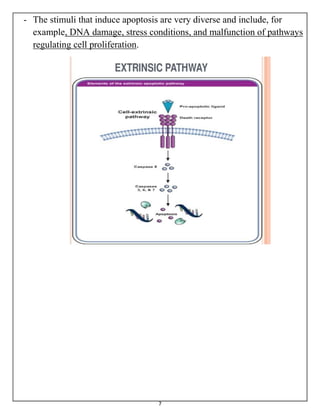
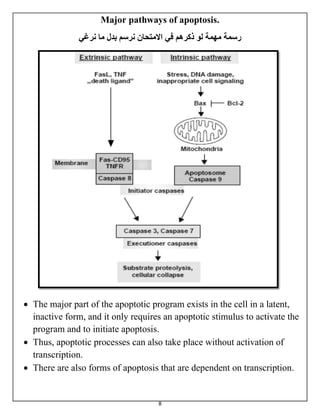
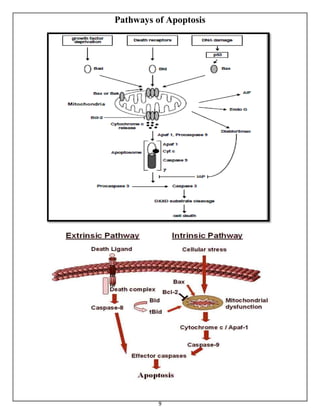
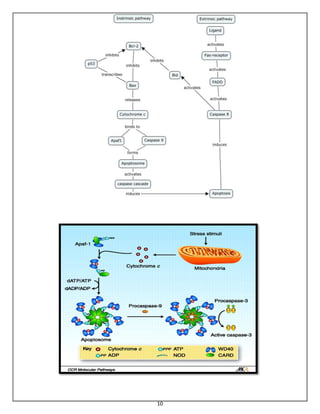
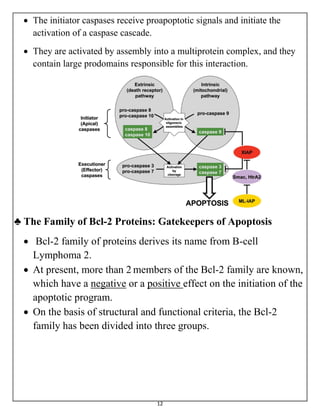
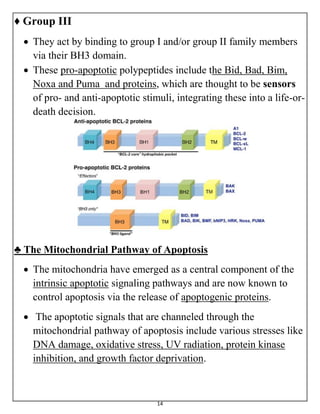
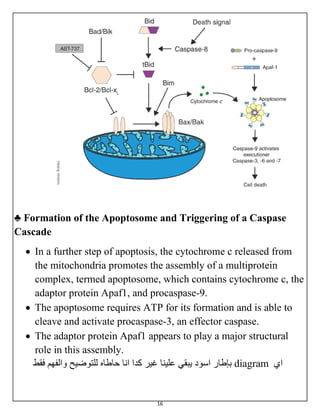
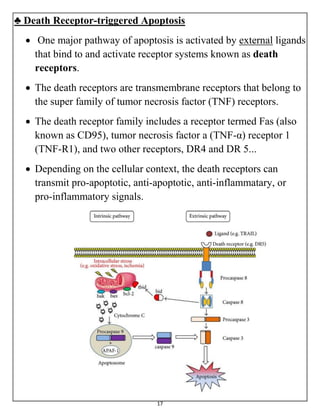
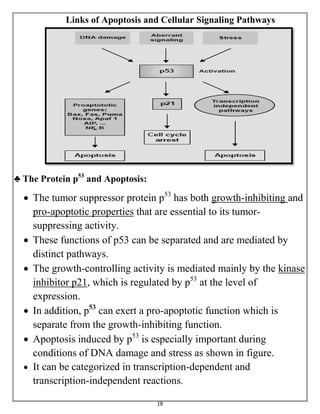
The document summarizes programmed cell death or apoptosis. It describes apoptosis as a naturally occurring, genetically programmed process where a cell undergoes an organized breakdown. During apoptosis, cells shrink, break into membrane-bound fragments called apoptotic bodies, and are removed by phagocytes without causing inflammation. The document outlines the major pathways of apoptosis, including the intrinsic mitochondrial pathway and extrinsic death receptor pathway, and discusses the roles of caspase proteases and Bcl-2 family proteins in apoptosis signaling and regulation.