This document provides information about polypropylene, including:
- Its discovery in 1951 by Phillips Petroleum chemists who first polymerized propylene, and its commercial production starting in 1957.
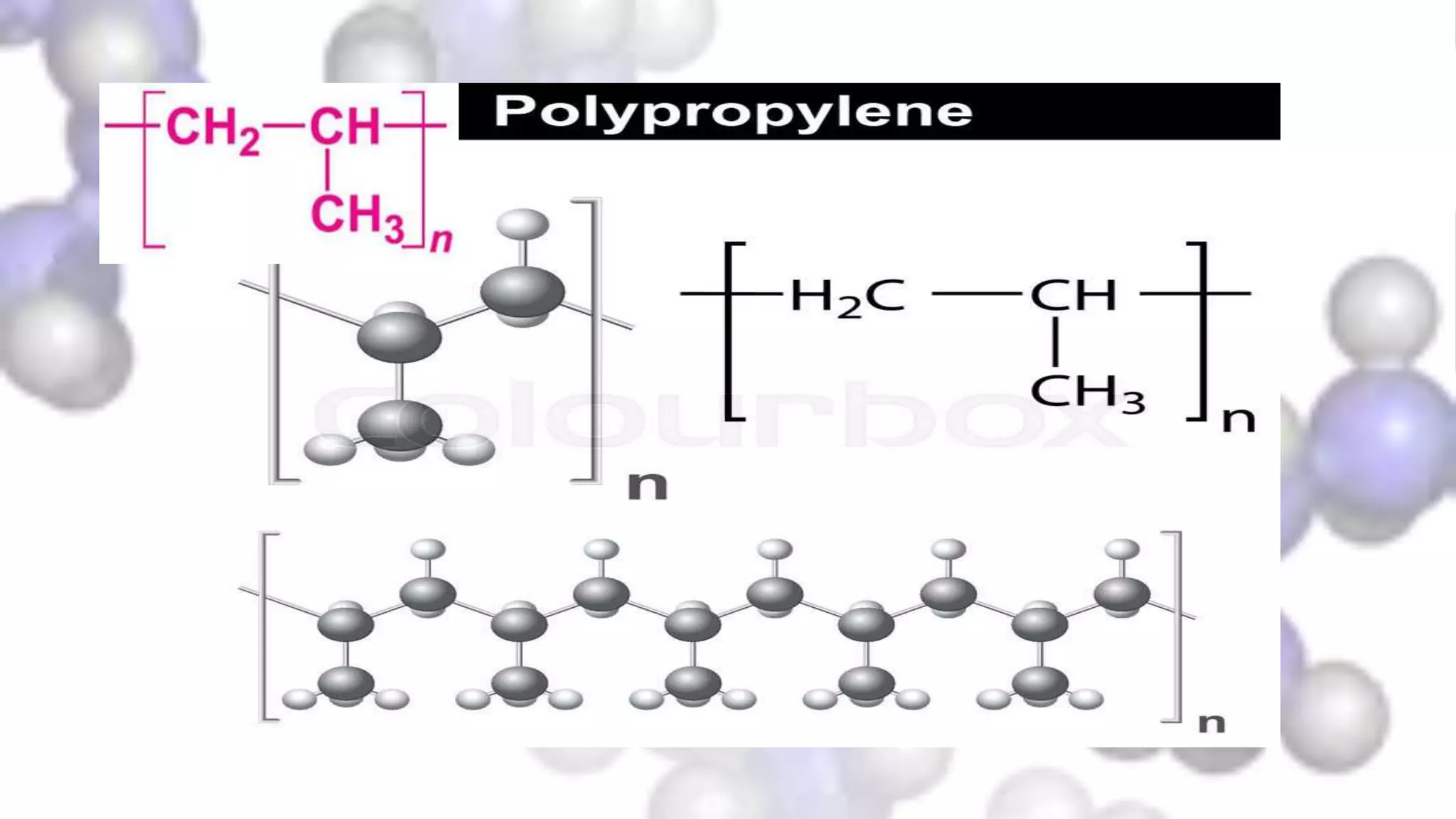
- Its structure as an addition polymer made from the monomer propylene, with methyl groups attached to every other carbon in the backbone chain.
- Its properties including chemical resistance to many solvents, bases and acids, and its mechanical properties of being tough and flexible.
- Its uses in products like food containers, medical devices, ropes, piping systems and more due to its resistance to corrosion and chemicals.
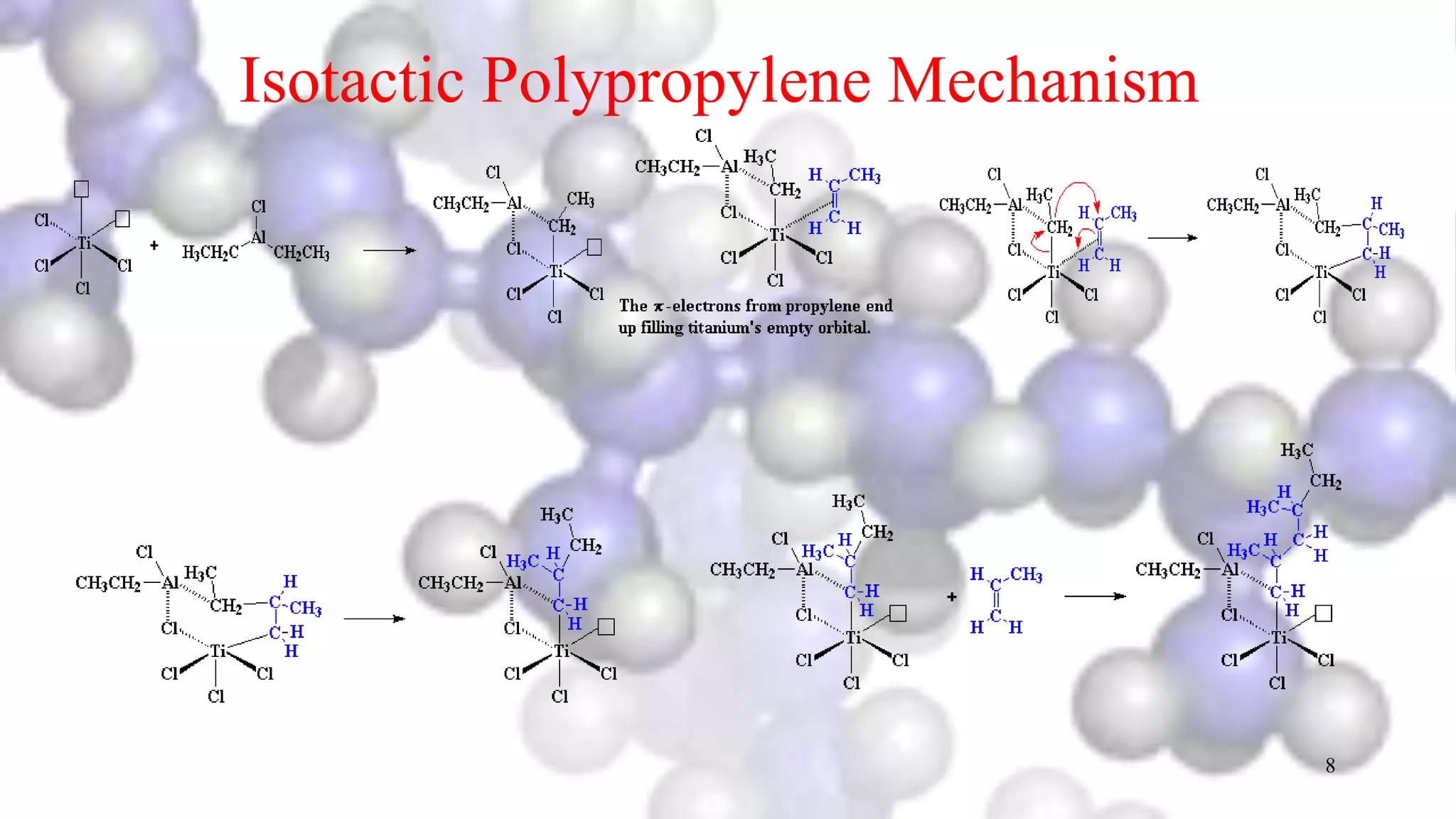
- How isotactic and syndiotactic polypropylene structures are controlled