1. Polyolefins are polymers produced from olefin or alkene monomers, with ethylene being the simplest and most common monomer. They are produced via polymerization of ethylene extracted from petroleum through cracking of longer hydrocarbon chains.
2. Low-density polyethylene (LDPE) is produced via free radical polymerization of ethylene under high pressures of 1000-3000 atm and temperatures of 200-275°C. It is used for products like bottles and tubing due to its low cost, chemical resistance, and flexibility.
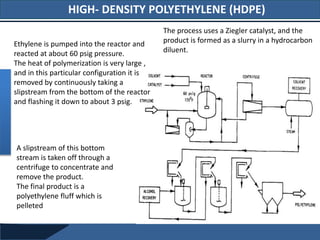
3. High-density polyethylene (HDPE) is produced using Ziegler-Natta catalysts under lower pressures than LDPE. It is stronger