This document discusses neutrophil disorders and their relationship to periodontal diseases. It begins with an introduction on the role of neutrophils in the innate immune system and periodontal diseases. It then describes various quantitative and qualitative neutrophil disorders. Quantitative disorders discussed include chronic benign neutropenia, cyclic neutropenia, congenital neutropenia, agranulocytosis, and Felty's syndrome. Qualitative disorders result from defects in neutrophil functions like rolling, adhesion, chemotaxis, phagocytosis, and intracellular killing. The document examines the oral complications that can result from various neutrophil disorders like gingivitis, periodontitis, and bone loss.













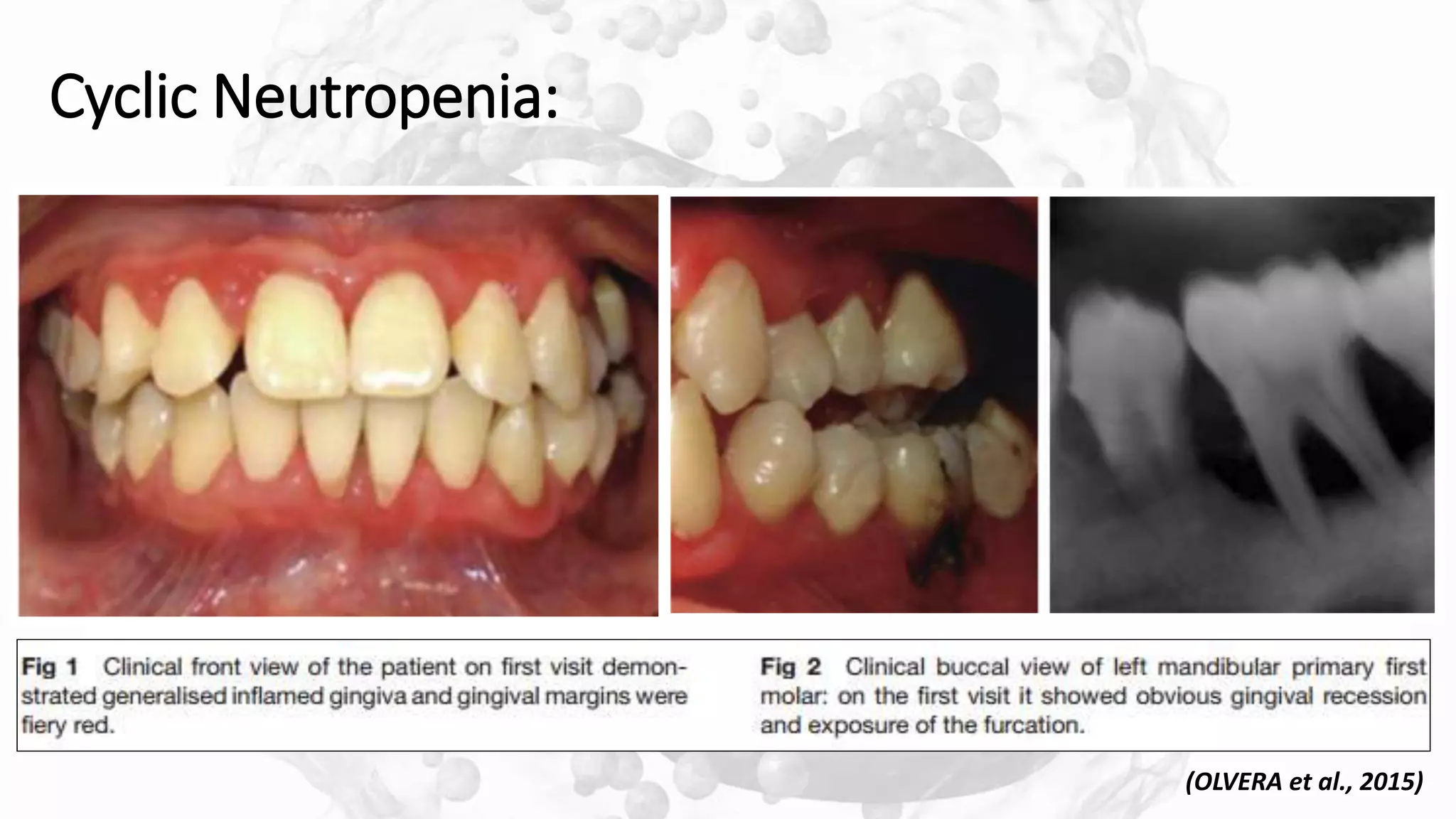
















![LAD-I
[HAJISHENGALLIS et al., 2015]](https://image.slidesharecdn.com/8pmndisordersperiodontaldiseases13120-200113174253/75/PMN-DISORDERS-PERIODONTAL-DISEASES-31-2048.jpg)





































