Physical properties kinetic theory me3
•Download as PPTX, PDF•
0 likes•352 views
Kinetic theory states that all particles of matter are constantly in motion. The state of a substance, such as solid, liquid, or gas, depends on factors like temperature and volume. When energy is added to a substance, its temperature or state can change. Density is a measure of the amount of matter present in a given volume and relates to kinetic theory by describing how closely packed particles are. Physical properties like viscosity, solubility, and malleability also depend on particle motion and interaction as described by kinetic theory.
Report
Share
Report
Share
Recommended
Recommended
ICSE Class VIII Chemistry Matter Around Us - TopperLearning

Anything which occupies space or volume, has mass and can be perceived by our senses is called matter.
Physical States Of Matter

Basics of solid, liquid and gas including info about gas and temperature relationship for 8th grade science - based on McDougal Littell series
Class IV -States of Matter

Summary of chapter Solids,Liquid & Gas of class IV Science, www.learnroots.com
States of Matter: Solids, Liquids and Gases [Presentation]![States of Matter: Solids, Liquids and Gases [Presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![States of Matter: Solids, Liquids and Gases [Presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Presentation for junior high school science.
Topic: States of Matter: Solids, Liquids and Gases.
Comprehensive 33-slide presentation designed for self-directed learning, although equally useful as a classroom presentation.
Neatly laid out with clear and detailed explanations.
Covers:
• Properties of Solids, Liquids and Gases
• Particles in Solids, Liquids and Gases
• Heat Energy and State Changes
• Expansion and Contraction in Solids, Liquids and Gases
There are several digital and print worksheets available that accompany this presentation.
<a />www.GoodScienceWorksheets.com</a>
*Presentation is in Adobe Acrobat format. View in full screen mode (Ctrl L) and scroll through slides with arrows; Esc to get out of full screen view, navigate through slides using Page Thumbnails on left side of page, just like a PowerPoint presentation.
Kinetic theory 11_12

Introduction to kinetic theory of matter. Students 3r ESO - Physics and chemistry IES Cap de Llevant. Maó - Menorca
More Related Content
What's hot
ICSE Class VIII Chemistry Matter Around Us - TopperLearning

Anything which occupies space or volume, has mass and can be perceived by our senses is called matter.
Physical States Of Matter

Basics of solid, liquid and gas including info about gas and temperature relationship for 8th grade science - based on McDougal Littell series
Class IV -States of Matter

Summary of chapter Solids,Liquid & Gas of class IV Science, www.learnroots.com
States of Matter: Solids, Liquids and Gases [Presentation]![States of Matter: Solids, Liquids and Gases [Presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![States of Matter: Solids, Liquids and Gases [Presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Presentation for junior high school science.
Topic: States of Matter: Solids, Liquids and Gases.
Comprehensive 33-slide presentation designed for self-directed learning, although equally useful as a classroom presentation.
Neatly laid out with clear and detailed explanations.
Covers:
• Properties of Solids, Liquids and Gases
• Particles in Solids, Liquids and Gases
• Heat Energy and State Changes
• Expansion and Contraction in Solids, Liquids and Gases
There are several digital and print worksheets available that accompany this presentation.
<a />www.GoodScienceWorksheets.com</a>
*Presentation is in Adobe Acrobat format. View in full screen mode (Ctrl L) and scroll through slides with arrows; Esc to get out of full screen view, navigate through slides using Page Thumbnails on left side of page, just like a PowerPoint presentation.
Kinetic theory 11_12

Introduction to kinetic theory of matter. Students 3r ESO - Physics and chemistry IES Cap de Llevant. Maó - Menorca
What's hot (20)
ICSE Class VIII Chemistry Matter Around Us - TopperLearning

ICSE Class VIII Chemistry Matter Around Us - TopperLearning
Physical science structure_and_properties_of_matter

Physical science structure_and_properties_of_matter
States of Matter: Solids, Liquids and Gases [Presentation]![States of Matter: Solids, Liquids and Gases [Presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![States of Matter: Solids, Liquids and Gases [Presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
States of Matter: Solids, Liquids and Gases [Presentation]
Similar to Physical properties kinetic theory me3
Practicing science (Properties of foods)

The properties of foods which play very important role while discussing rheological and textural properties of foods.
Changing States of Matter for Middle School

This Changing States of Matter slideshow for middle school students is ideally used to teach students about the various processes that change one state of matter to another. The processes featured are freezing, melting, evaporation, condensation, sublimation, and deposition. Thermal energy and movement of molecules are discussed.
To download slideshow or the powerpoint version of this slideshow, visit The Homeschool Daily. Be sure to check out our full line of homeschool curriculum. Thanks for looking!
385154297-Kinetic-Molecular-Theory-and-states-of-matter-ppt.ppt

bjbsjnskamaskjdfkmnfdkkqw.lejdkdkncdjckdsjwj
Similar to Physical properties kinetic theory me3 (20)
385154297-Kinetic-Molecular-Theory-and-states-of-matter-ppt.ppt

385154297-Kinetic-Molecular-Theory-and-states-of-matter-ppt.ppt
Recently uploaded
How to Create Map Views in the Odoo 17 ERP

The map views are useful for providing a geographical representation of data. They allow users to visualize and analyze the data in a more intuitive manner.
ESC Beyond Borders _From EU to You_ InfoPack general.pdf

ESC Beyond Borders _From EU to You_ InfoPack general.pdfFundacja Rozwoju Społeczeństwa Przedsiębiorczego
Wolontariat grupowyOperation Blue Star - Saka Neela Tara

Operation “Blue Star” is the only event in the history of Independent India where the state went into war with its own people. Even after about 40 years it is not clear if it was culmination of states anger over people of the region, a political game of power or start of dictatorial chapter in the democratic setup.
The people of Punjab felt alienated from main stream due to denial of their just demands during a long democratic struggle since independence. As it happen all over the word, it led to militant struggle with great loss of lives of military, police and civilian personnel. Killing of Indira Gandhi and massacre of innocent Sikhs in Delhi and other India cities was also associated with this movement.
The Roman Empire A Historical Colossus.pdf

The Roman Empire, a vast and enduring power, stands as one of history's most remarkable civilizations, leaving an indelible imprint on the world. It emerged from the Roman Republic, transitioning into an imperial powerhouse under the leadership of Augustus Caesar in 27 BCE. This transformation marked the beginning of an era defined by unprecedented territorial expansion, architectural marvels, and profound cultural influence.
The empire's roots lie in the city of Rome, founded, according to legend, by Romulus in 753 BCE. Over centuries, Rome evolved from a small settlement to a formidable republic, characterized by a complex political system with elected officials and checks on power. However, internal strife, class conflicts, and military ambitions paved the way for the end of the Republic. Julius Caesar’s dictatorship and subsequent assassination in 44 BCE created a power vacuum, leading to a civil war. Octavian, later Augustus, emerged victorious, heralding the Roman Empire’s birth.
Under Augustus, the empire experienced the Pax Romana, a 200-year period of relative peace and stability. Augustus reformed the military, established efficient administrative systems, and initiated grand construction projects. The empire's borders expanded, encompassing territories from Britain to Egypt and from Spain to the Euphrates. Roman legions, renowned for their discipline and engineering prowess, secured and maintained these vast territories, building roads, fortifications, and cities that facilitated control and integration.
The Roman Empire’s society was hierarchical, with a rigid class system. At the top were the patricians, wealthy elites who held significant political power. Below them were the plebeians, free citizens with limited political influence, and the vast numbers of slaves who formed the backbone of the economy. The family unit was central, governed by the paterfamilias, the male head who held absolute authority.
Culturally, the Romans were eclectic, absorbing and adapting elements from the civilizations they encountered, particularly the Greeks. Roman art, literature, and philosophy reflected this synthesis, creating a rich cultural tapestry. Latin, the Roman language, became the lingua franca of the Western world, influencing numerous modern languages.
Roman architecture and engineering achievements were monumental. They perfected the arch, vault, and dome, constructing enduring structures like the Colosseum, Pantheon, and aqueducts. These engineering marvels not only showcased Roman ingenuity but also served practical purposes, from public entertainment to water supply.
Home assignment II on Spectroscopy 2024 Answers.pdf

Answers to Home assignment on UV-Visible spectroscopy: Calculation of wavelength of UV-Visible absorption
The French Revolution Class 9 Study Material pdf free download

The French Revolution, which began in 1789, was a period of radical social and political upheaval in France. It marked the decline of absolute monarchies, the rise of secular and democratic republics, and the eventual rise of Napoleon Bonaparte. This revolutionary period is crucial in understanding the transition from feudalism to modernity in Europe.
For more information, visit-www.vavaclasses.com
Supporting (UKRI) OA monographs at Salford.pptx

How libraries can support authors with open access requirements for UKRI funded books
Wednesday 22 May 2024, 14:00-15:00.
GIÁO ÁN DẠY THÊM (KẾ HOẠCH BÀI BUỔI 2) - TIẾNG ANH 8 GLOBAL SUCCESS (2 CỘT) N...

GIÁO ÁN DẠY THÊM (KẾ HOẠCH BÀI BUỔI 2) - TIẾNG ANH 8 GLOBAL SUCCESS (2 CỘT) N...Nguyen Thanh Tu Collection
https://app.box.com/s/4hfk1xwgxnova7f4dm37birdzflj806wCambridge International AS A Level Biology Coursebook - EBook (MaryFosbery J...

for studentd in cabridge board
Introduction to Quality Improvement Essentials

This is a presentation by Dada Robert in a Your Skill Boost masterclass organised by the Excellence Foundation for South Sudan (EFSS) on Saturday, the 25th and Sunday, the 26th of May 2024.
He discussed the concept of quality improvement, emphasizing its applicability to various aspects of life, including personal, project, and program improvements. He defined quality as doing the right thing at the right time in the right way to achieve the best possible results and discussed the concept of the "gap" between what we know and what we do, and how this gap represents the areas we need to improve. He explained the scientific approach to quality improvement, which involves systematic performance analysis, testing and learning, and implementing change ideas. He also highlighted the importance of client focus and a team approach to quality improvement.
Template Jadual Bertugas Kelas (Boleh Edit)

Jadual Bertugas kelas dalam bentuk softcopy PowerPoint. Cikgu boleh download >> edit >> print >> laminate. Semoga bermanfaat.
The geography of Taylor Swift - some ideas

Geographical themes connected with Taylor Swift's ERAS tour - coming to the UK in June 2024
Digital Tools and AI for Teaching Learning and Research

This Presentation in details discusses on Digital Tools and AI for Teaching Learning and Research
Palestine last event orientationfvgnh .pptx

An EFL lesson about the current events in Palestine. It is intended to be for intermediate students who wish to increase their listening skills through a short lesson in power point.
CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE

Class 11 CBSE Business Studies Project ( AIDS TO TRADE - INSURANCE)
Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46

Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46
Event Link:-
https://meetups.mulesoft.com/events/details/mulesoft-mysore-presents-exploring-gemini-ai-and-integration-with-mulesoft/
Agenda
● Java 17 Upgrade Overview
● Why and by when do customers need to upgrade to Java 17?
● Is there any immediate impact to upgrading to Mule Runtime 4.6 and beyond?
● Which MuleSoft products are in scope?
For Upcoming Meetups Join Mysore Meetup Group - https://meetups.mulesoft.com/mysore/
YouTube:- youtube.com/@mulesoftmysore
Mysore WhatsApp group:- https://chat.whatsapp.com/EhqtHtCC75vCAX7gaO842N
Speaker:-
Shubham Chaurasia - https://www.linkedin.com/in/shubhamchaurasia1/
Priya Shaw - https://www.linkedin.com/in/priya-shaw
Organizers:-
Shubham Chaurasia - https://www.linkedin.com/in/shubhamchaurasia1/
Giridhar Meka - https://www.linkedin.com/in/giridharmeka
Priya Shaw - https://www.linkedin.com/in/priya-shaw
Shyam Raj Prasad-
https://www.linkedin.com/in/shyam-raj-prasad/
How to Make a Field invisible in Odoo 17

It is possible to hide or invisible some fields in odoo. Commonly using “invisible” attribute in the field definition to invisible the fields. This slide will show how to make a field invisible in odoo 17.
How to Break the cycle of negative Thoughts

We all have good and bad thoughts from time to time and situation to situation. We are bombarded daily with spiraling thoughts(both negative and positive) creating all-consuming feel , making us difficult to manage with associated suffering. Good thoughts are like our Mob Signal (Positive thought) amidst noise(negative thought) in the atmosphere. Negative thoughts like noise outweigh positive thoughts. These thoughts often create unwanted confusion, trouble, stress and frustration in our mind as well as chaos in our physical world. Negative thoughts are also known as “distorted thinking”.
Recently uploaded (20)
ESC Beyond Borders _From EU to You_ InfoPack general.pdf

ESC Beyond Borders _From EU to You_ InfoPack general.pdf
Home assignment II on Spectroscopy 2024 Answers.pdf

Home assignment II on Spectroscopy 2024 Answers.pdf
The French Revolution Class 9 Study Material pdf free download

The French Revolution Class 9 Study Material pdf free download
GIÁO ÁN DẠY THÊM (KẾ HOẠCH BÀI BUỔI 2) - TIẾNG ANH 8 GLOBAL SUCCESS (2 CỘT) N...

GIÁO ÁN DẠY THÊM (KẾ HOẠCH BÀI BUỔI 2) - TIẾNG ANH 8 GLOBAL SUCCESS (2 CỘT) N...
Cambridge International AS A Level Biology Coursebook - EBook (MaryFosbery J...

Cambridge International AS A Level Biology Coursebook - EBook (MaryFosbery J...
Digital Tools and AI for Teaching Learning and Research

Digital Tools and AI for Teaching Learning and Research
CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE

CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE
Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46

Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46
Physical properties kinetic theory me3
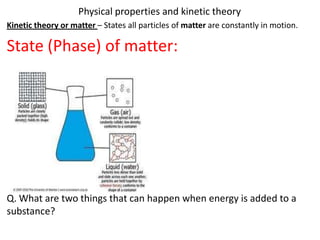
- 1. Physical properties and kinetic theory Kinetic theory or matter – States all particles of matter are constantly in motion. State (Phase) of matter: Q. What are two things that can happen when energy is added to a substance?
- 2. A: Change of temperature or state
- 3. Volume (Space Occupied) Ex. Even concrete needs to expand due to heat. Expansion joints are created in bridges and roads to allow for this natural adjustment. Q: What are two ways to measure volume?
- 4. Measuring Volume • V=lwh • Graduated Cylinder • Fluid Displacement
- 5. Mass (Amount of matter an object or substance contains) Q. What tool might be used to find the mass of an object?
- 6. Balance
- 7. Density Density – Amount of matter present in a certain volume. Density = Mass/volume Q: How does this relate to kinetic theory?
- 8. Physical properties cont. Viscosity Tendency of a substance to resist flow Results in slower or faster movement of substance particles. Low energy = low heat = thickness and slow flow. Ex. (honey from refrigerator) High energy= high heat = thinner and faster flow. Ex. (honey from microwave) Kk Substances get thicker as temperatures (energy amount) lower. Substances get thinner as temperatures rise.
- 9. How does viscosity relate to kinetic theory? • Draw and label how the molecules of a viscous substance might be different from a non- viscous substance.
- 10. Solubility (How a substance dissolves in a given volume of liquid.) Ex. Salt or sugar dissolves much better than dirt in a given volume of liquid What happens to mud or dirt when It’s put in water? Dirt in solvent(different properties than sugar)
- 11. NEW VOCABULARY!! • Dissolving - disappearing visually in a solvent • Solute – substance (usually a solid) being dissolved in a liquid. • Solvent – substance (usually a liquid) doing the dissolving.
- 12. Think back to lab… Temperature most definitely determines how quickly something can dissolve!! Sugar in hot coffee vs. cold coffee Cold Solvent Hot Solvent
- 13. Malleability Ability of a substance (usually metals) to be formed into different shapes without breaking. - Metals have many uses in the world because of this property. - Can you name a few?
- 14. Physical Change Any change that is made to a substances physical properties - observed without changing the identity (internal makeup) of a substance. - Always ask yourself if the identity of the substance is the same or not following a change. What happens to water as it goes from liquid to a solid? Is this a physical change or not ( a chemical change)?
- 15. Image Sources • Teachersource.com • Mrsdlovesscience.com • www.123rf.com
Editor's Notes
- *Review how molecules move differently in solids, liquids, and gases (note solid particles still moving)
- *A: temperature rises, or if at specific point, change of state occursPhysical property: state physical change: change of state