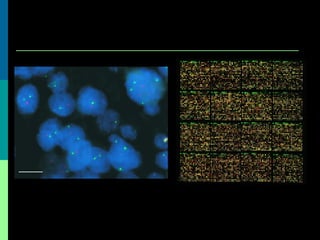
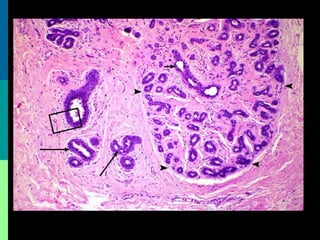
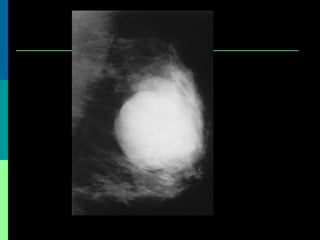
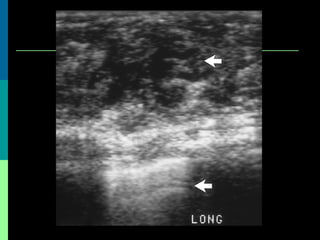
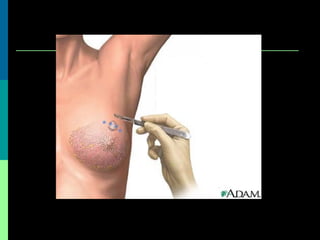
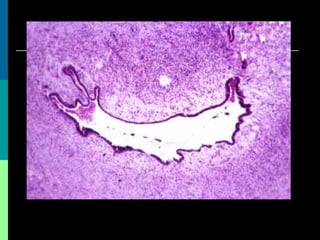
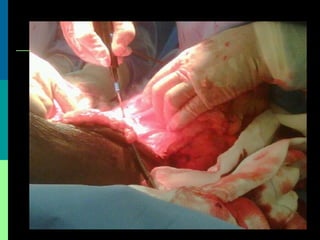
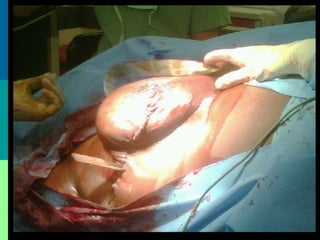
This document discusses breast cancer awareness month and provides details on a case of a 37-year-old woman presenting with a rapidly growing mass in her right breast. The mass was examined and found to be a large, firm, irregular mass. Differential diagnoses included giant fibroadenoma, phyllodes tumor, and carcinoma. Further workup and histological analysis determined it was a phyllodes tumor, a rare benign breast tumor that can occasionally become malignant. Treatment options and outcomes for phyllodes tumors are summarized.