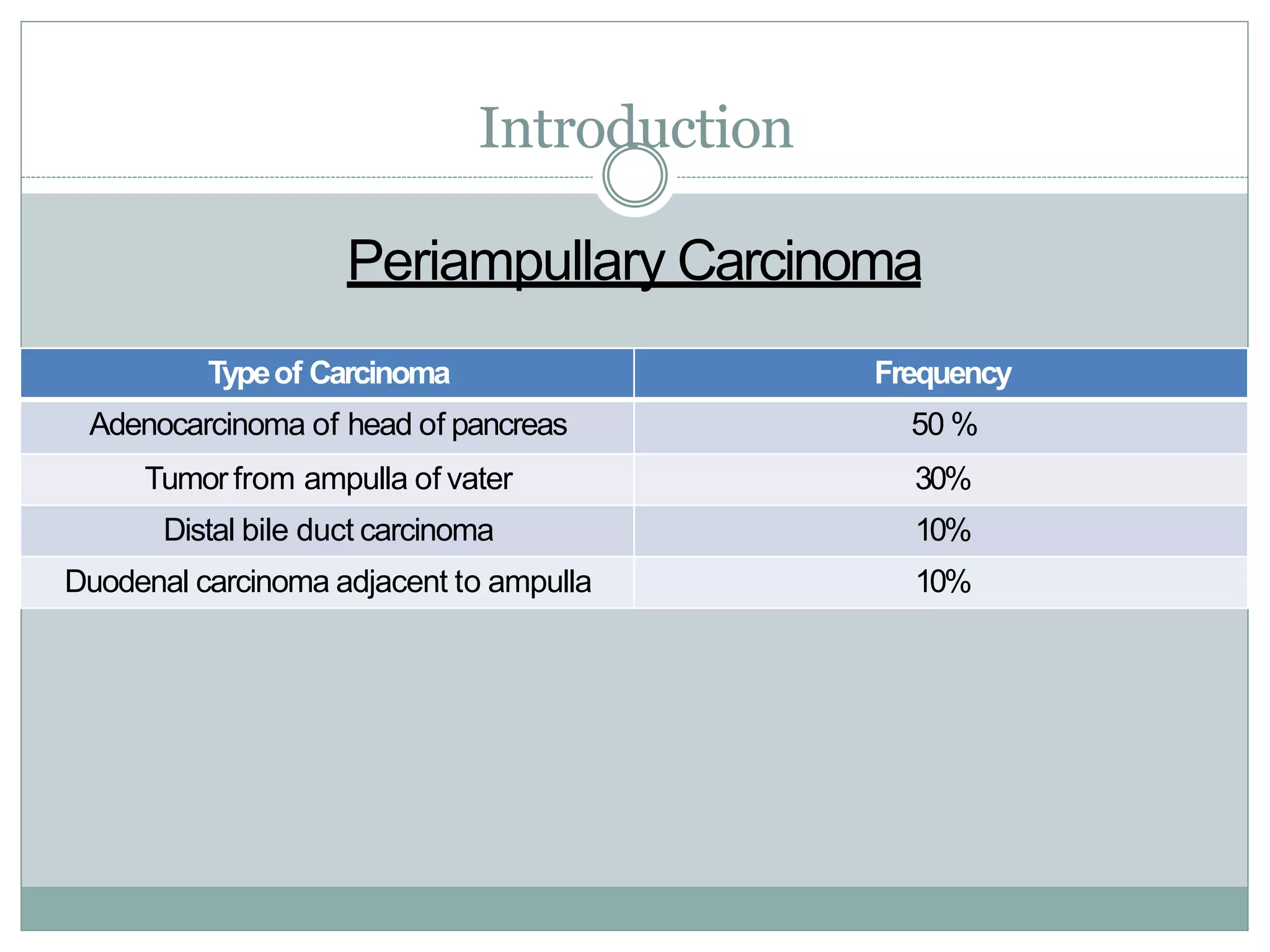
Periampullary carcinoma refers to cancers that form near the ampulla of Vater. The four main types are adenocarcinoma of the pancreatic head, ampullary tumors, distal bile duct carcinomas, and duodenal carcinomas adjacent to the ampulla. Evaluation involves imaging like CT, MRI, and EUS to determine resectability. Resectable tumors are treated with surgery followed by chemotherapy, while borderline resectable tumors receive chemotherapy and radiation before surgery. Unresectable tumors are treated with chemotherapy and/or radiation. Adjuvant chemotherapy may improve survival for resected cancers.



















![ CONKO-001: Final results of the randomized, prospective,
multicenter phase III trial of adjuvant chemotherapy with
gemcitabine versus observation in patients with resected
pancreatic cancer (PC)
Background: Prognosis of patients (pts) with PC is dismal, even after
curatively intended resection. Whereas gemcitabine-based chemotherapy is
standard in advanced PC, the role of adjuvant chemotherapy is still under
discussion.
Methods: CONKO-001, a prospective, open, multicenter, controlled phase
3 study was designed to evaluate the efficacy and toxicity of gemcitabine in
PC pts after complete (R0 or R1) resection. pts were randomized to receive
either gemcitabine (G) (1g/m2 d 1, 8 and 15 every 4 weeks) for 6 months or
observation (O).
Primary study endpoint was disease free survival (DFS), secondary
endpoints included OS and toxicity.
Results: The analyses confirm the significant improvement for G in
median DFS [G:13.4 months (m), O: 6.9m, p< .001]. Estimated DFS at 3
and 5 years was 23.5% and 16.0% in the G group vs. 8.5% and 6.5% in the O
group, respectively. G significantly improves median OS [G:22.8m, O:
20.2m, p=.005]. Estimated survival at 3 and 5 years was 36.5% and 21.0%
for G pts vs. 19.5% and 9.0% for O pts, respectively.
Conclusion: Treatment with G for 6 months for pts after complete
resection of PC significantly increases DFS and OS compared with O alone.](https://image.slidesharecdn.com/periampullarycarcinoma-230819134952-c3726b0c/75/PERIAMPULLARY-CARCINOMA-pptx-22-2048.jpg)