The presentation discusses the +1 oxidation state of copper (Cu+), detailing its electronic structure, properties, stability, and various compounds such as cuprous oxide (Cu2O) and cuprous chloride (CuCl). Furthermore, it highlights the preparation methods, uses of these compounds in applications like ruby glass production, and their significance in analytical chemistry. Additionally, biological relevance is noted with the role of copper in proteins and oxidases for electron transfer.


![+1 OXIDATION STATE OF Cu
ELECTRONIC STRUCTURE:
Copper Cu+(28): [Ar] 3d10.
PROPERTIES:
•Magnetism: Diamagnetic.
•Color and spectra: colorless.
[There are also some colored compounds:
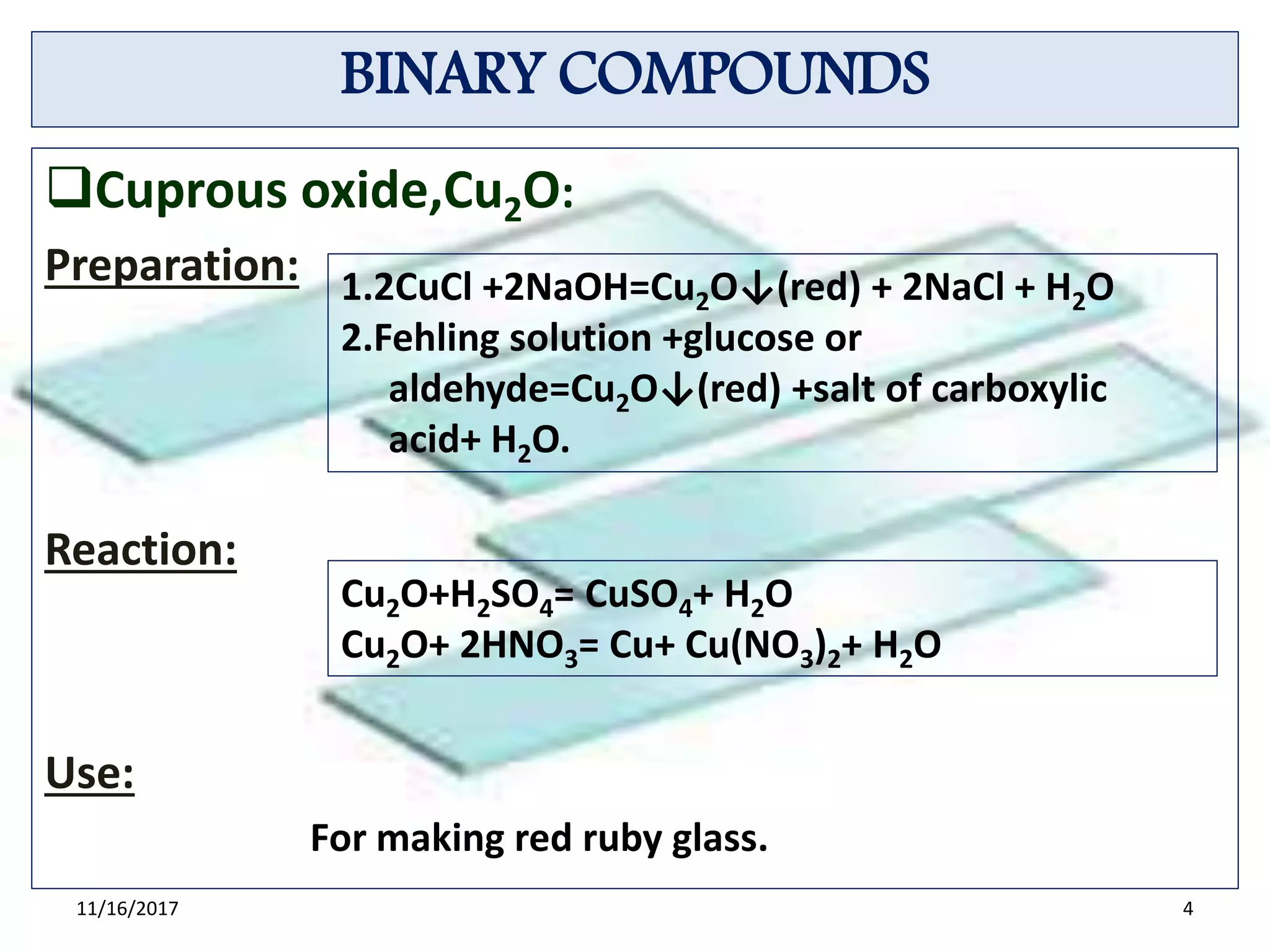
Cu2O is yellow or red
Cu2CO3 is yellow etc.
•Stability:
Less stable than Cu2+ and Cu3+.
Cause: 1st ionisation potential is higher than the 2nd and 3rd
ionisation potential.
11/16/2017 3](https://image.slidesharecdn.com/oxidationstateofcu1-171116151923/75/Oxidation-state-of-cu-1-3-2048.jpg)

![OXIDATION STATES AND STEREOCHEMISTRY OF
COPPER (1).
OXIDATION
STATE
CO
ORDINATION
NUMBER
GEOMETRY EXAMPLES
2 linear Cu2O, KCuO, CuCl2-
Cu1, d10 3 planer Cu[CuCN]2,
[Cu(SPMe3)3]ClO4
4 tetrahedral CuI, [CuCN]3-
11/16/2017 7](https://image.slidesharecdn.com/oxidationstateofcu1-171116151923/75/Oxidation-state-of-cu-1-7-2048.jpg)
![CUPROUS COMPLEXES
1.K[Cu(CN)2]
2.(1,3-dimethyltriazeno) copper (1)
complex
Figure: a portion of spiral
chain in K[Cu(CN)2].
Figure: (1,3-dimethyltriazeno
) copper (1) complex.
2.26A◦
11/16/2017 8](https://image.slidesharecdn.com/oxidationstateofcu1-171116151923/75/Oxidation-state-of-cu-1-8-2048.jpg)
![CUPROUS COMPLEXES
3.PHOSPHINE AND ARSINE
COMPLEXES. 4.[CuH(PPh3)]6
Figure : Cubane complexes
,Cu4I4L4 (L=R3P, R3As)
Figure: the Cu6P6 group in
[CuH(PPh3)]6. the Cu-Cu
distances shown as full lines
have an average value of
2.65A◦ While those shown
broken lines average 2.54A◦11/16/2017 9](https://image.slidesharecdn.com/oxidationstateofcu1-171116151923/75/Oxidation-state-of-cu-1-9-2048.jpg)
![CUPROUS COMPLEXES
5: Cu4Cl6(Ph3PO)4 , Cu4OCl6Py4, and[(CH3)4N]4-[Cu4OCl10].
Figure: Cu4Cl6(Ph3PO)4 , Cu4OCl6Py4, and[(CH3)4N]4-[Cu4OCl10]
complexes.
11/16/2017 10](https://image.slidesharecdn.com/oxidationstateofcu1-171116151923/75/Oxidation-state-of-cu-1-10-2048.jpg)
![ORGANO DERIVATIVES
Acetylene complexes:
1.CuCl (in conc. HCl soln) + CH≡CH → CuCl(C2H2) or [CuCl2(C2H2)]-.
2.CuNH2 (soln)+ acetylenes containing CH≡C-→
Olefines complexes: They may be prepared by-
i. Direct rection with Cu1 halides.
ii. May also prepared from cupric salts by reduction with trialkyl
phosphites in ethanolic solution in presence of alkene.
Yellow or red ppt
11/16/2017 11](https://image.slidesharecdn.com/oxidationstateofcu1-171116151923/75/Oxidation-state-of-cu-1-11-2048.jpg)


