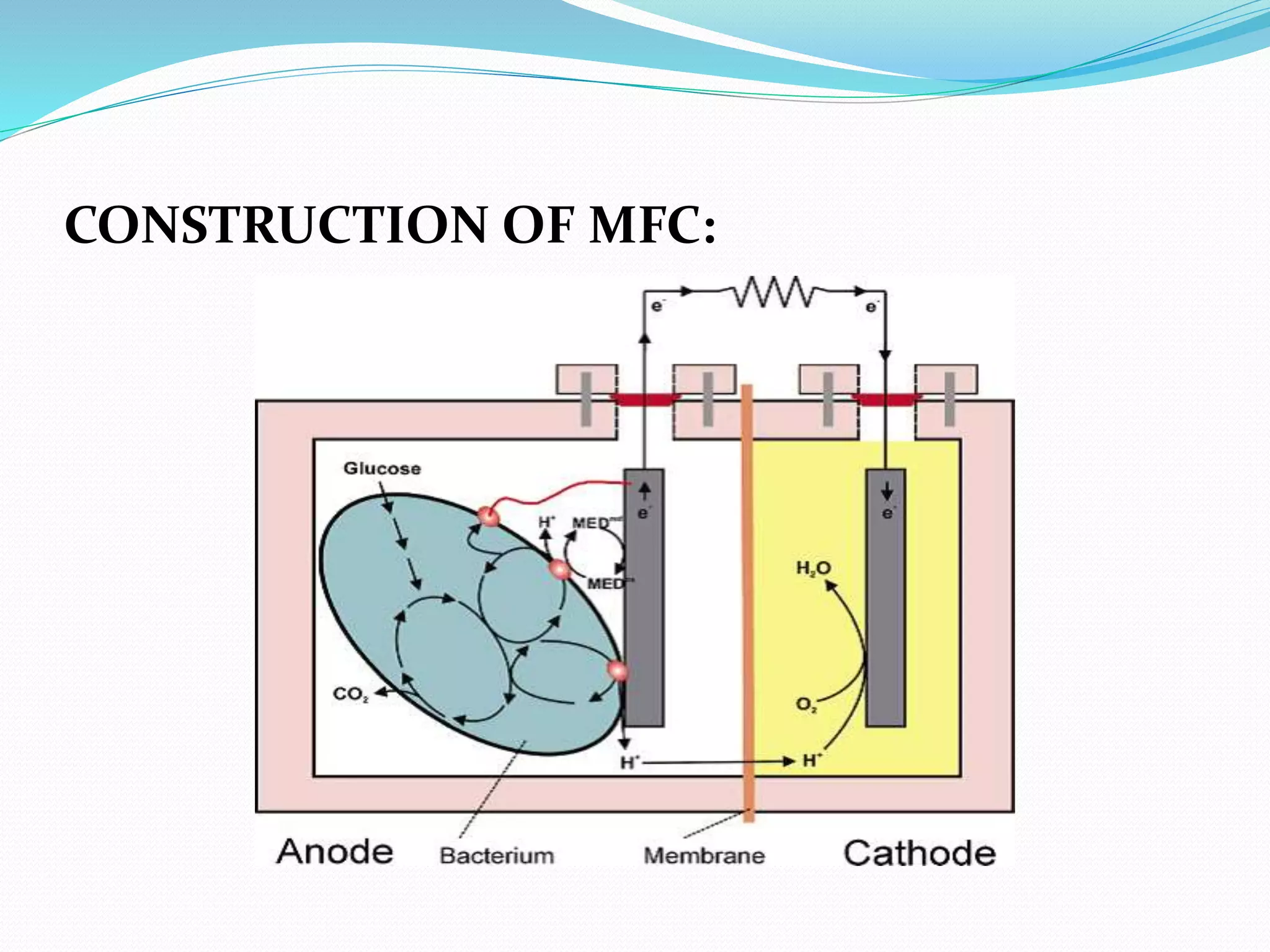
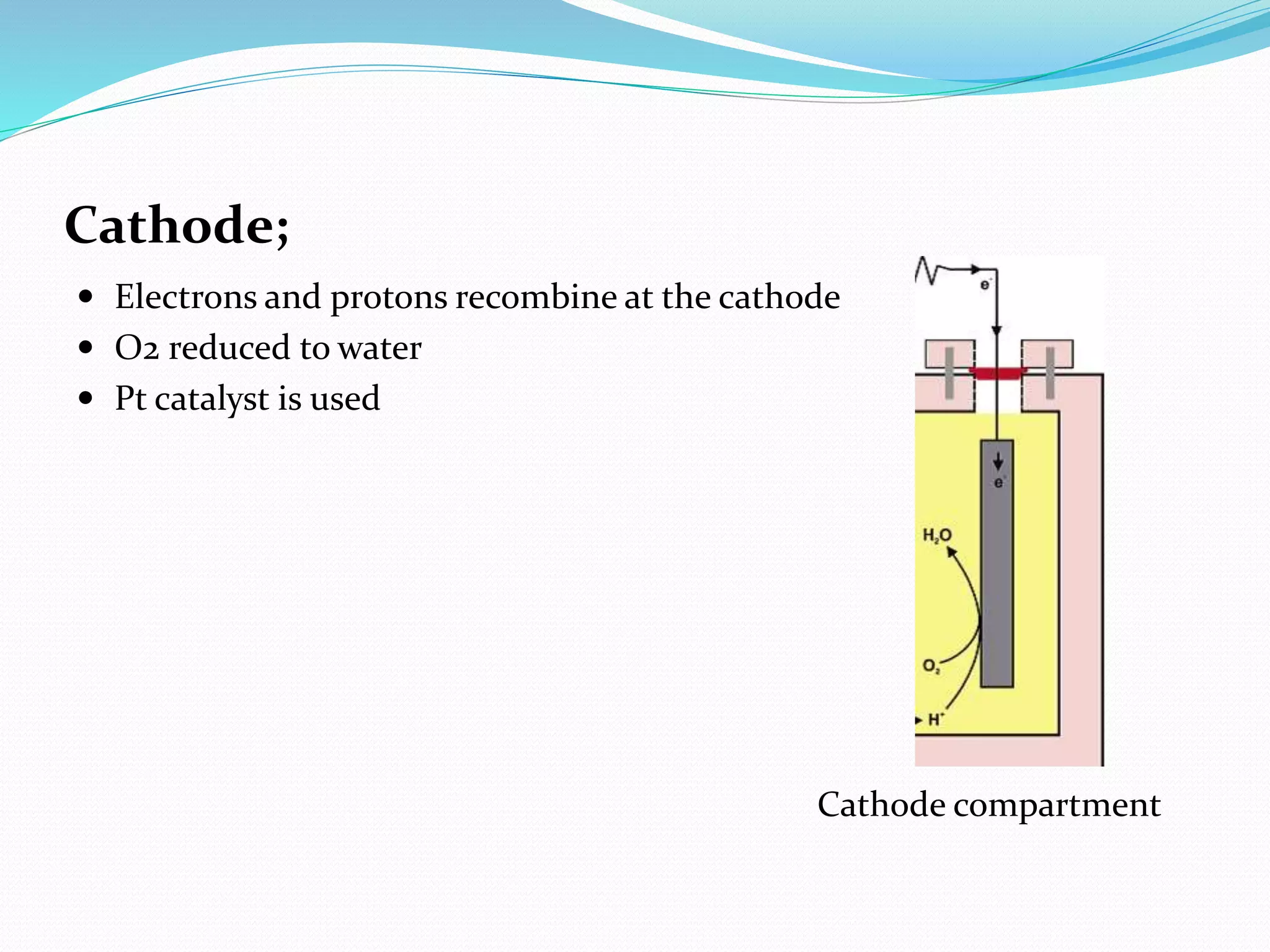
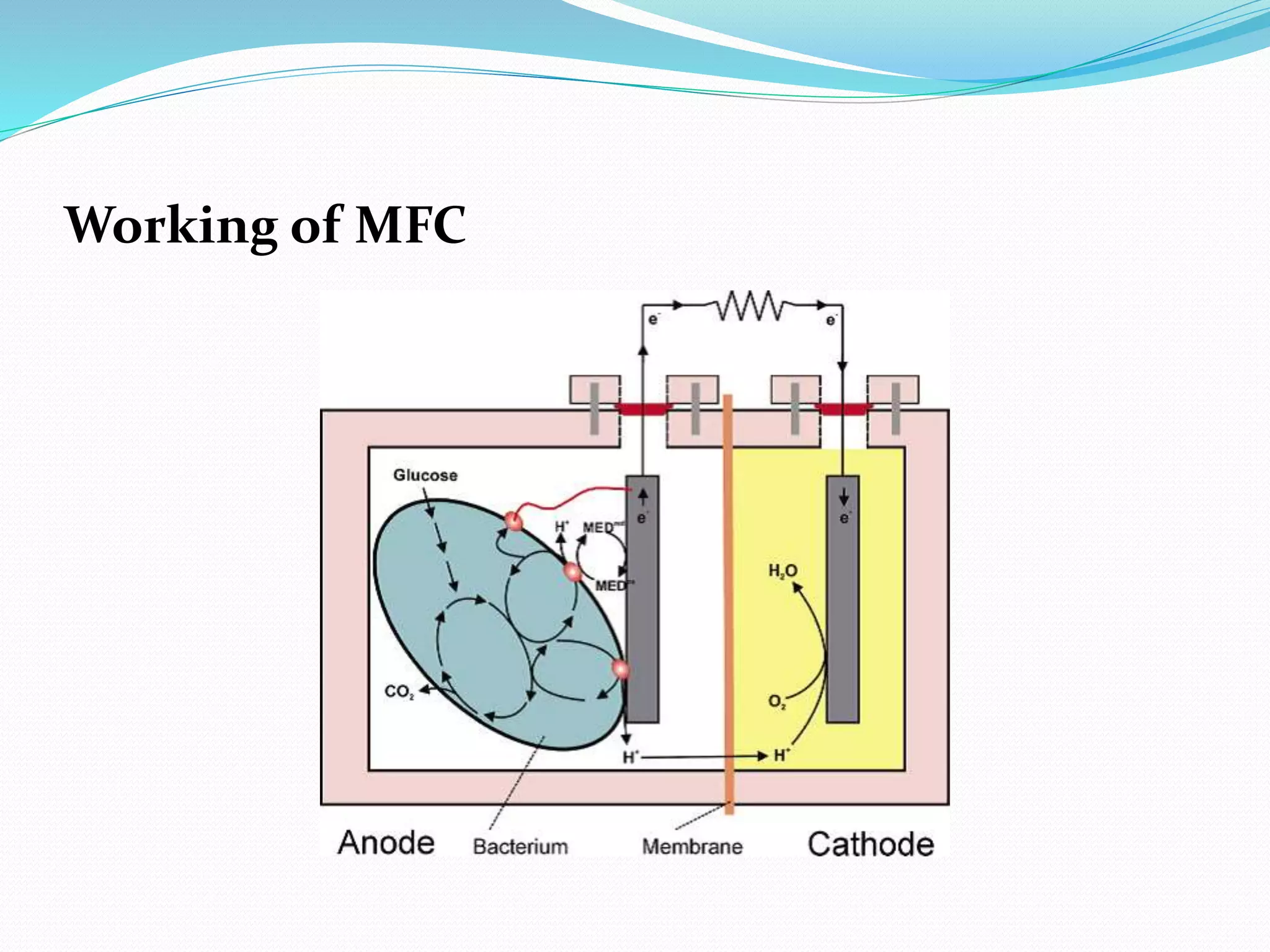
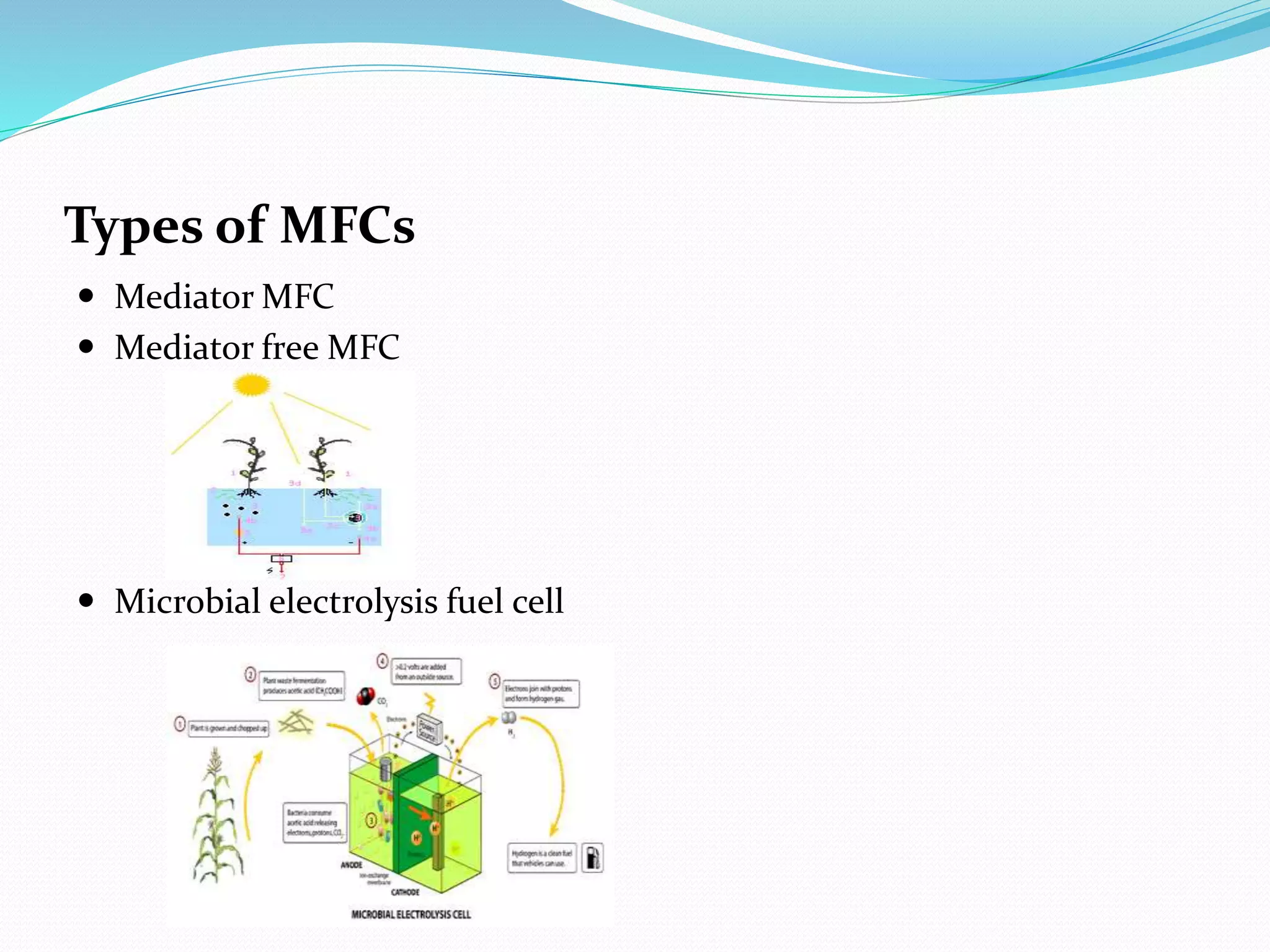
The document discusses microbial fuel cells (MFCs), which generate electricity through the catalytic reactions of microorganisms. It describes the basic components and principles of MFCs, including how bacteria at the anode convert organic substrates into protons and electrons. The protons pass through a membrane to the cathode, where the electrons from the external circuit also travel to recombine with the protons and oxygen, producing water. The document outlines various MFC designs, microbes, substrates, and applications. While MFCs can simultaneously treat wastewater and generate electricity, the technology still has low power densities and high costs compared to other energy sources.