lipid droplet.pptx
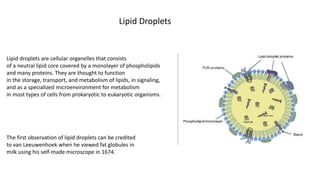
- 1. Lipid droplets are cellular organelles that consists of a neutral lipid core covered by a monolayer of phospholipids and many proteins. They are thought to function in the storage, transport, and metabolism of lipids, in signaling, and as a specialized microenvironment for metabolism in most types of cells from prokaryotic to eukaryotic organisms. Lipid Droplets The first observation of lipid droplets can be credited to van Leeuwenhoek when he viewed fat globules in milk using his self-made microscope in 1674.
- 3. In which cells and tissues, we can see them In the last 10 years, established isolation methods and advanced proteomic technology have allowed scientists to study the protein composition of lipid droplets from many cell types and tissues, such as fibroblasts , epithelia , adipocytes , hepatocytes , macrophages , pancreatic cells , mammary glands , livers , white adipose tissues , and skeletal muscles , as well as from many other popular model organisms, including plants , insects , yeast green algae , bacteria
- 6. Exact mechanism of formation of lipid droplets is still unknown, it is proposed that they bud off the membrane of the endoplasmic reticulum. 1. Acetyl co A is made from pyruvate dehydrogenase and mitochondria. 2. Acetyl co A converts malonyl co A by ACC.(acetyl co A carboxylase ) 3. Malonyl co A converts to free fatty acids(FFA) by FAS ( fatty acid synthase) 4. Glycerol and FFA come together to the ER membrane . Inside ER lumen they conjugate together. 5. ER embedded proteins such as GPAT4 & DGAT1/2 facilitate this process. 6. Those proteins condense FFA in glycerol and make TAG(tri acyl glycerol) & DAG(di acyl glycerol). 7. ER membrane makes lenses . TAG & DAG accumulate in to the ER membrane lenses. 8. FITM2 & BSCL2 are proteins which are found in neck of ER membrane budding lenses( most initial step of lipid droplet). 9. Those proteins facilitates the movement of TAG & DAG in to the lenses and are very important for the formation of lipid droplet. 10. Budding ER membrane is covered by perilipin proteins( plin1/2), that stabilize membrane of lipid droplet and protect from lipases. The formation of droplets is not spontaneous but is facilitated by ER proteins and specific lipids. Seipin, a protein that resides at ER– droplet junctions, functions early to stabilize the nascent structures, ensures droplets of correct morphology, prevents the accumulation of phosphatidic acid at the ER–droplet junction, and prevents ectopic budding into the nucleus.
- 7. Lipid droplet degradation • In starvation the ATP concentration of cells are low and AMP concentration is high. • That is signaling AMPK proteins.(AMP kinase). • When AMP binds to AMPK ,AMPK activates and it inhibit the perilipin1/2 proteins of lipid droplets. • Perilipin 1/2 inhibit the degradation of lipid droplets. • So after inhibiting perilipin proteins degradation of lipid droplets is started. As well as • In G proteins activation activate adenylyl cyclase protein. • adenylyl cyclase turns to ATP then cyclic AMP(cAMP ) • CAMP binds PKA(protein kinase A) • PKA deactivate perilipin 1/2 .then starts lipid droplets degradation. And also, • There is CMA(chaperone mediated autophagy), cytosolic proteins(HSC70) that are then targeted to lysosomes and directly translocated across the lysosome membrane for degradation. And these poteins inhibits perilipin1/2 of lipid droplets .
- 9. Perilipin (PLIN) • Perilipin, also known as lipid droplet-associated protein, Perilipin 1, or PLIN, is a protein that, in humans, is encoded by the PLIN gene. The perilipins are a family of proteins that associate with the surface of lipid droplets. Phosphorylation of perilipin is essential for the mobilization of fats in adipose tissue. • Perilipin acts as a protective coating from the body’s natural lipases, such as hormone-sensitive lipase, which break triglycerides into glycerol and free fatty acids for use in metabolism, a process called lipolysis. In humans, perilipin is expressed in three different isoforms, A, B, and C, and perilipin A is the most abundant protein associated with the adipocyte lipid droplets. • Perilipin is hyperphosphorylated by PKA following β-adrenergic receptor activation.Phosphorylated perilipin changes conformation, exposing the stored lipids to hormone-sensitive lipase-mediated lipolysis. Although PKA also phosphorylates hormone-sensitive lipase, which can increase its activity, the more than 50-fold increase in fat mobilization (triggered by epinephrine) is primarily due to perilipin phosphorylation • Perilipin is part of a gene family with five currently-known members. In vertebrates, closely related genes include adipophilin (also known as adipose differentiation-related protein or Perilipin 2), TIP47 (Perilipin 3), Perilipin 4 and Perilipin 5 (also called MLDP, LSDP5, or OXPAT). Insects express related proteins, LSD1 and LSD2, in fat bodies. The yeast Saccharomyces cerevisiae expresses PLN1 (formerly PET10), that stabilizes lipid droplets and aids in their assembly.
- 10. PET 10 Pet10p is a yeast lipid droplet protein of unknown function. We show that it binds specifically to and is stabilized by droplets containing triacylglycerol (TG). Droplets isolated from cells with a PET10 deletion strongly aggregate, appear fragile, and fuse in vivo when cells are cultured in oleic acid. Pet10p binds early to nascent droplets, and their rate of appearance is decreased in pet10Δ. Moreover, Pet10p functionally interacts with the endoplasmic reticulum droplet assembly factors seipin and Fit2 to maintain proper droplet morphology. The activity of Dga1p, a diacylglycerol acyltransferase, and TG accumulation were both 30–35% lower in the absence of Pet10p. Pet10p contains a PAT domain,a defining property of perilipins, which was not previously known to exist in yeast. We propose that the core functions of Pet10p and other perilipins extend beyond protection from lipases and include the preservation of droplet integrity as well as collaboration with seipin and Fit2 in droplet assembly and maintenance. Pet10p, an established lipid droplet protein in yeast, which binds to TG- containing droplets where it is stabilized, is important for droplet integrity, influences protein distribution, facilitates droplet formation, contributes to TG reservoirs, and collaborates with seipin and Fit2 proteins to maintain normal droplet size. Pet10p and Sps4p both contain a PAT domain, and complementation of pet10Δ with human Plin2 and Plin3 indicates that Pet10p is indeed a yeast perilipin.
- 11. Seipin is a homo-oligomeric integral membrane protein in the endoplasmic reticulum (ER) that concentrates at junctions with cytoplasmic lipid droplets (LDs). Alternatively, seipin can be referred to as Bernardinelli-Seip congenital lipodystrophy type 2 protein (BSCL2), and it is encoded by the corresponding gene of the same name, i.e. BSCL2. At protein level, seipin is expressed in cortical neurons in the frontal lobes, as well as motor neurons in the spinal cord. It is highly expressed in areas like the brain, testis and adipose tissue.Seipin's function is still unclear but it has been localized close to lipid droplets, and cells knocked out in seipin which have anomalous droplets. Hence, recent evidence suggests that seipin plays a crucial role in lipid droplet biogenesis.
- 12. RESEARCH The research described herein began with an analysis of proteins associated with droplets enriched in one of the core lipids, TG or SE. We report that Pet10p, a known droplet surface protein with unknown function (Athenstaedt et al., 1999), preferentially localizes to TG droplets, where it is protected from degradation. Pet10p is required for droplet integrity; in its absence, droplet structure is less stable, exhibiting severe aggregation in vitro. In addition, loss of the protein results in droplet fusion in vivo when cells are cultured with oleic acid (OA). PET10-null cells accumulate less TG, which for the acyltransferase Dga1p is accompanied by a loss of enzyme activity and movement from the ER to droplets. Pet10p localizes to droplets early in their assembly and promotes droplet formation. We identify a PAT domain in Pet10p and consider it a yeast perilipin, and we propose that the activities of Pet10p reflect important functions of the perilipin classes.
- 13. How its work Lipid droplets play a role in neutral lipid transport between cellular organelles. Lipids can be synthesize in cytoplasm and transferred to the ER then stored in lipid droplets. Play and active role in lipid synthesis. Lipid synthetic enzymes have been found on lipid droplets. In plant cells lipid droplets storage the energy within the organelle. that is the primary function of the plant lipid droplets. Starvation or high energy demand activities various lipases are releasing lipids as FFA that can enter the beta oxidation cycle. Lipid droplets are commonly associated in adipose tissues they found in nearly all the cell type. Lipid droplets of adipose tissues are obviously responsible for the obesity. In brain tissue LDs are formed on astrocytes in response to ROS stress. Bona fide LD diseases (neutral lipid storage diseases ) are associated with myopathy ,autism, lost of hearing And eyesight . Lipid droplets are responsible for neuron degenerative diseases. Abnormal LD dynamics can be observed in Alzheimer's disease and Huntington’s disease .
- 15. • Lipid droplets have important functions beyond energy homeostasis. • They store vitamins, signaling precursors, and other hydrophobic molecules. • They mitigate some harmful effects of ER and oxidative stress. lipid handling roles of droplets include the storage of hydrophobic vitamin and signaling precursors, and the management of endoplasmic reticulum and oxidative stress. • They function in protein maturation, storage, and turnover.• • They are motile and can exist in the nucleus. Other potential roles of lipid droplets may be connected with their intracellular motility and, in some cases, their nuclear localization.