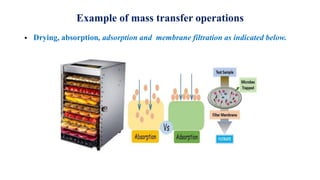
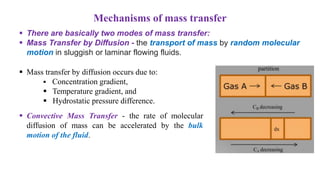
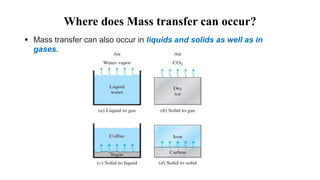
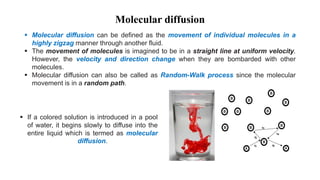
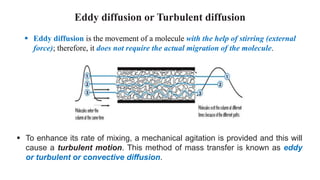
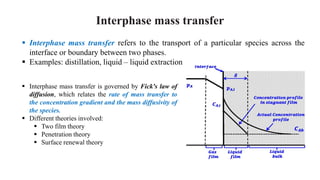
Mass transfer is the net movement of a component in a mixture from one location to another due to a concentration gradient. It occurs through mechanisms such as diffusion, where molecules move through random motion, and convection, where bulk fluid motion enhances diffusion. Mass transfer plays a key role in chemical reactions by bringing reactants together. It is quantified by mass transfer coefficients and can be modeled by theories like two film theory. Mass transfer is important in many industrial processes like distillation, drying, and absorption.