L3b reactor sizing example problems
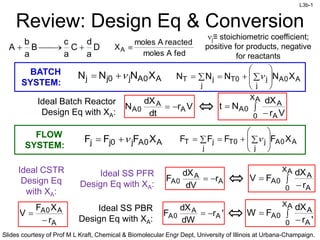
- 1. L3b-1 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. Ideal CSTR Design Eq with XA: Review: Design Eq & Conversion D a d C a c B a b A fedAmoles reactedAmoles XA BATCH SYSTEM: A0Aj0jj XNNN j A0A j j0TjT XNNNN FLOW SYSTEM: A0Aj0jj XFFF j A0A j j0TjT XFFFF r XF V A A0A Vr dt dX N A A 0A Ideal Batch Reactor Design Eq with XA: AX 0 A A 0A Vr dX Nt A A 0A r dV dX F Ideal SS PFR Design Eq with XA: AX 0 A A 0A r dX FV 'r dW dX F A A 0A Ideal SS PBR Design Eq with XA: AX 0 A A 0A 'r dX FW j≡ stoichiometric coefficient; positive for products, negative for reactants
- 2. L3b-2 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. Review: Sizing CSTRsWe can determine the volume of the CSTR required to achieve a specific conversion if we know how the reaction rate rj depends on the conversion Xj A A 0A CSTR A A0A CSTR X r F V r XF V Ideal SS CSTR design eq. Volume is product of FA0/-rA and XA • Plot FA0/-rA vs XA (Levenspiel plot) • VCSTR is the rectangle with a base of XA,exit and a height of FA0/-rA at XA,exit FA 0 rA X Area = Volume of CSTR X1 V FA 0 rA X1 X1
- 3. L3b-3 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. FA 0 rA Area = Volume of PFR V 0 X1 FA 0 rA dX X1 Area = VPFR or Wcatalyst, PBR dX 'r F W 1X 0 A 0A Review: Sizing PFRs & PBRs We can determine the volume (catalyst weight) of a PFR (PBR) required to achieve a specific Xj if we know how the reaction rate rj depends on Xj A exit,AX 0 A 0A PFR exit,AX 0 A A 0APFR dX r F V r dX FV Ideal PFR design eq. • Plot FA0/-rA vs XA (Experimentally determined numerical values) • VPFR (WPBR) is the area under the curve FA0/-rA vs XA,exit A exit,AX 0 A 0A PBR exit,AX 0 A A 0APBR dX r F W r dX FW Ideal PBR design eq. dX r F V 1X 0 A 0A
- 4. L3b-4 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. Numerical Evaluation of Integrals (A.4) Simpson’s one-third rule (3-point): 210 2X 0 XfXf4Xf 3 h dxxf hXX 2 XX h 01 02 Trapezoidal rule (2-point): 10 1X 0 XfXf 2 h dxxf 01 XXh Simpson’s three-eights rule (4-point): 3210 3X 0 XfXf3Xf3Xfh 8 3 dxxf 3 XX h 03 h2XXhXX 0201 Simpson’s five-point quadrature : 43210 4X 0 XfXf4Xf2Xf4Xf 3 h dxxf 4 XX h 04
- 5. L3b-5 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. Review: Reactors in Series 2 CSTRs 2 PFRs CSTR→PFR VCSTR1 VPFR2 VPFR2VCSTR1 VCSTR2 VPFR1 VPFR1 VCSTR2 VCSTR1 + VPFR2 ≠ VPFR1 + CCSTR2 PFR→CSTR A A0 r- F i j CSTRPFRPFR VVV If is monotonically increasing then: CSTR i j CSTRPFR VVV
- 6. L3b-6 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. Chapter 2 Examples
- 7. L3b-7 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. XA 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.85 -rA 0.0053 0.0052 0.0050 0.0045 0.0040 0.0033 0.0025 0.0018 0.00125 0.001 1. Calculate FA0/-rA for each conversion value in the tableFA0/-rA Calculate the reactor volumes for each configuration shown below for the reaction data in the table when the molar flow rate is 52 mol/min. FA0, X0 X1=0.3 X2=0.8 Config 1 X1=0.3 FA0, X0 X2=0.8 Config 2 A exit,AX in,AX A 0A nPFR dX r F V ←Use numerical methods to solve in,Aout,A nA 0A nCSTR XX r F V XA,out and XA,in respectively, are the conversion at the outlet and inlet of reactor n Convert to seconds→ min mol 52F 0A 00 1 52 8 60 67 A mol min m mol . F sin s -rA is in terms of mol/dm3∙s
- 8. L3b-8 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. A( 0 0) AF r 3 3 mol 0.0053 d mol 0.867 s s m m d 164 1. Calculate FA0/-rA for each conversion value in the table XA 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.85 -rA 0.0053 0.0052 0.0050 0.0045 0.0040 0.0033 0.0025 0.0018 0.00125 0.001 FA0/-rA 164 Calculate the reactor volumes for each configuration shown below for the reaction data in the table when the molar flow rate is 52 mol/min. FA0, X0 X1=0.3 X2=0.8 Config 1 X1=0.3 FA0, X0 X2=0.8 Config 2 A exit,AX in,AX A 0A nPFR dX r F V ←Use numerical methods to solve in,Aout,A nA 0A nCSTR XX r F V -rA is in terms of mol/dm3∙s 164 XA,out and XA,in respectively, are the conversion at the outlet and inlet of reactor n min mol 52F 0A 00 1 52 8 60 67 A mol min m mol . F sin s Convert to seconds→
- 9. L3b-9 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. A( 0 0) AF r 3 3 mol 0.0053 d mol 0.867 s s m m d 164 1. Calculate FA0/-rA for each conversion value in the table XA 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.85 -rA 0.0053 0.0052 0.0050 0.0045 0.0040 0.0033 0.0025 0.0018 0.00125 0.001 FA0/-rA Calculate the reactor volumes for each configuration shown below for the reaction data in the table when the molar flow rate is 52 mol/min. FA0, X0 X1=0.3 X2=0.8 Config 1 X1=0.3 FA0, X0 X2=0.8 Config 2 A exit,AX in,AX A 0A nPFR dX r F V ←Use numerical methods to solve in,Aout,A nA 0A nCSTR XX r F V -rA is in terms of mol/dm3∙s 164 XA,out and XA,in respectively, are the conversion at the outlet and inlet of reactor n min mol 52F 0A 00 1 52 8 60 67 A mol min m mol . F sin s Convert to seconds→ For each –rA that corresponds to a XA value, use FA0 to calculate FA0/-rA & fill in the table
- 10. L3b-10 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. X1=0.3 FA0, X0 A( 0.85) 3A0 3 mol 0.867F s molr 0.001 dm s 867 dm 1. Calculate FA0/-rA for each conversion value in the table XA 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.85 -rA 0.0053 0.0052 0.0050 0.0045 0.0040 0.0033 0.0025 0.0018 0.00125 0.001 FA0/-rA 164 167 173 193 217 263 347 482 694 867 Calculate the reactor volumes for each configuration shown below for the reaction data in the table when the molar flow rate is 52 mol/min. FA0, X0 X1=0.3 X2=0.8 Config 1 X2=0.8 Config 2 A exit,AX in,AX A 0A nPFR dX r F V ←Use numerical methods to solve in,Aout,A nA 0A nCSTR XX r F V Convert to seconds→ min mol 52F 0A -rA is in terms of mol/dm3∙s XA,out and XA,in respectively, are the conversion at the outlet and inlet of reactor n 00 1 52 8 60 67 A mol min m mol . F sin s
- 11. L3b-11 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. XA 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.85 -rA 0.0053 0.0052 0.0050 0.0045 0.0040 0.0033 0.0025 0.0018 0.00125 0.001 FA0/-rA 164 167 173 193 217 263 347 482 694 867 FA0, X0 X1=0.3 X2=0.8 Config 1 Reactor 1, PFR from XA0=0 to XA=0.3: A A AA A A0 A 0.3 A0 PFR1 A 0 A0 X 0 A X A0 A X 0.3 0.20A .X 1A 0 F 3 0.3 0 V dX 3 F F 3 rr F rr8 3 F r 4-pt rule: 1 0.3 A0 PFR A0 3 A 16 F 3 V dX 0.1 3 3 1 r 8 934 173 5167 1.6 dm A,out 2CSTR A0 A,o A i X , nut A F XV X r 2 3 CSTR 694 0.8 3470.3 dmV Total volume for configuration 1: 51.6 dm3 + 347 dm3 = 398.6 dm3 = 399 dm3 ←Use numerical methods to solve PFR1 CSTR2 0 XA,exit A PFRn AXA,in A F V dX r
- 12. L3b-12 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. XA 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.85 -rA 0.0053 0.0052 0.0050 0.0045 0.0040 0.0033 0.0025 0.0018 0.00125 0.001 FA0/-rA 164 167 173 193 217 263 347 482 694 867 Reactor 1, CSTR from XA0=0 to XA=0.3: Need to evaluate at 6 pts, but since there is no 6-pt rule, break it up 0 0 1 0 3 A A . A,outCSTR A F XV X r Total volume for configuration 2: 58 dm3 + 173 dm3 = 231 dm3 X1=0.3 FA0, X0 X2=0.8 Config 2 CSTR 3 0. 583 0193 dmV A0 PFR2 A A 0.8 0.3 F V dX r PFRV ... . 263 263 34217 3 4 3 3 8 33 2 482193 694 0 08 5 7 0 30 5 3 point rule 4 point rule 3 173 dm PFR2CSTR1 0. A0 A0 PF 0.3 R2 A A A 05 . . 5 8 A0 F F V dX dX r r Must evaluate as many pts as possible when the curve isn’t flat
- 13. L3b-13 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. ACSTR A A V X C r 0 0 CSTR A A A V C r X 00 For a given CA0, the space time needed to achieve 80% conversion in a CSTR is 5 h. Determine (if possible) the CSTR volume required to process 2 ft3/min and achieve 80% conversion for the same reaction using the same CA0. What is the space velocity (SV) for this system? space time holding time mean residenceh V time 0 5 =5 h 0=2 ft3/min ft min h hV min 3 60 5 2 3 V ft 600 V SV 0 1Space velocity: -1 h SV . h 0 2 5 1 1 Notice that we did not need to solve the CSTR design equation to solve this problem. Also, this answer does not depend on the type of flow reactor used. XA=0.8 A CSTR A AF r XV 0 A A CSTR A C r V X 0 0 0 0 V V
- 14. L3b-14 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. XA,exit PFR A A X AA,in C V dX r 0 0 A product is produced by a nonisothermal, nonelementary, multiple-reaction mechanism. Assume the volumetric flow rate is constant & the same in both reactors. Data for this reaction is shown in the graph below. Use this graph to determine which of the 2 configurations that follow give the smaller total reactor volume. FA0, X0 X1=0.3 X2=0.7 Config 2 X1=0.3 FA0, X0 X2=0.7 Config 1 A CSTR A,out A,in A V X X r C 0 0 Shown on graph XA,exit PFRn A AA,in A X V dX F r 0 CSTR A A A V X r F 0 • Since 0 is the same in both reactors, we can use this graph to compare the 2 configurations • PFR- volume is 0 multiplied by the area under the curve between XA,in & XA,out • CSTR- volume is 0 multiplied by the product of CA0/-rA,outlet times (XA,out - XA,in)
- 15. L3b-15 Slides courtesy of Prof M L Kraft, Chemical & Biomolecular Engr Dept, University of Illinois at Urbana-Champaign. A product is produced by a nonisothermal, nonelementary, multiple-reaction mechanism. Assume the volumetric flow rate is constant & the same in both reactors. Data for this reaction is shown in the graph below. Use this graph to determine which of the 2 configurations that follow give the smaller total reactor volume. FA0, X0 X1=0.3 X2=0.7 Config 2 X1=0.3 FA0, X0 X2=0.7 Config 1 • PFR- V is 0 multiplied by the area under the curve between XA,in & XA,out • CSTR- V is 0 multiplied by the product of CA0/-rA,outlet times (XA,out - XA,in) Config 1 Config 2 Less shaded area Config 2 (PFRXA,out=0.3 first, and CSTRXA,out=0.7 second) has the smaller VTotal XA=0.3 XA=0.7 XA=0.3 XA=0.7
