The document discusses updated CLSI antimicrobial susceptibility testing challenges, particularly focusing on the differences between CLSI and FDA breakpoints for Enterobacteriaceae, which have been revised to better reflect current resistance mechanisms. Strategies for validating these new breakpoints in commercial AST systems are outlined, along with the need for collaboration among laboratory professionals. The presentation also emphasizes understanding pharmacokinetics and pharmacodynamics to improve patient treatment outcomes.
![Coping with Updated CLSI Antimicrobial Susceptibility Testing (AST) Challenges Janet A. Hindler, MCLS MT(ASCP) Sr. Specialist, Clinical Microbiology UCLA Medical Center [email_address] “ and a consultant with the Association of Public Health Laboratories”](https://image.slidesharecdn.com/jhindlerepaasm-v2-11-08-10-110124130941-phpapp02/75/CLSI-Antimicrobial-Susceptibility-Update-Janet-Hindler-November-Symposium-2010-1-2048.jpg)








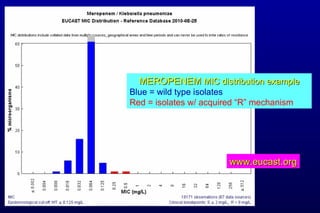
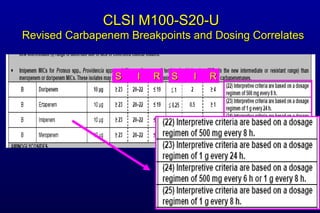
![Why did CLSI lower cephalosporin, aztreonam, and carbapenem breakpoints for Enterobacteriaceae? (3) Re: Carbapenem breakpoints Carbapenem resistance “issues” increasing rapidly Phenotypic test for carbapenemases [Modified Hodge test (MHT)] Subjective interpretation Does not detect all carbapenem “R” mechanisms](https://image.slidesharecdn.com/jhindlerepaasm-v2-11-08-10-110124130941-phpapp02/85/CLSI-Antimicrobial-Susceptibility-Update-Janet-Hindler-November-Symposium-2010-10-320.jpg)