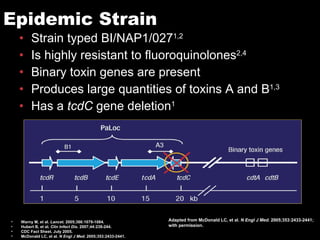
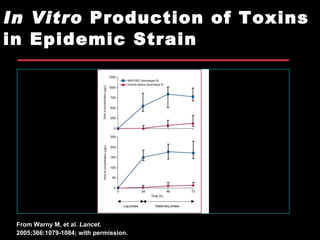
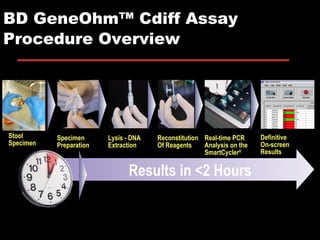
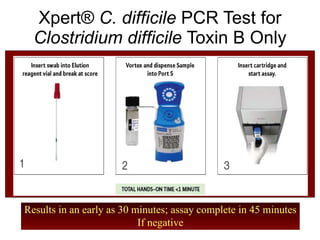
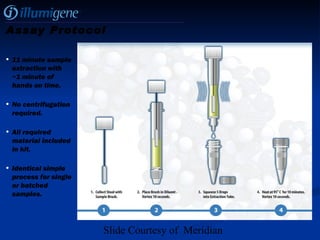
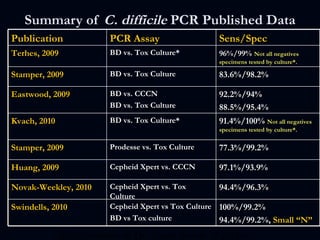
The document provides a comprehensive overview of Clostridium difficile infections (CDI), detailing its historical perspective, changing epidemiology, risk factors, pathogenesis, symptoms, laboratory diagnosis, and treatment options. It highlights the rise in CDI cases and associated mortality, the complexity of diagnosing CDI, and the importance of accurate testing and infection control measures. Additionally, it discusses the effectiveness of various treatments and the challenges posed by antibiotic use and virulent strains of the bacteria.