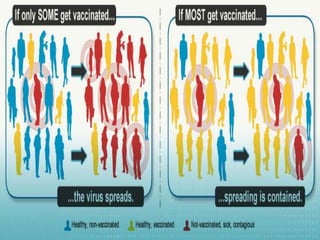
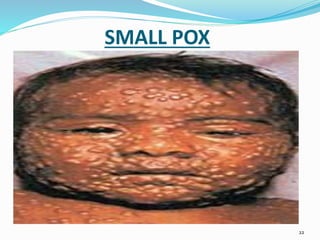
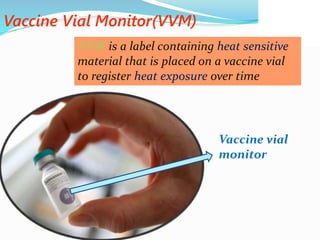
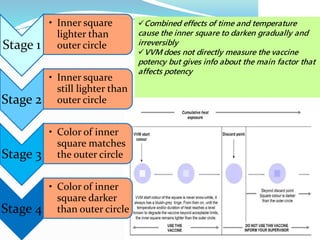
1. The document discusses immunization and the cold chain system for transporting and storing vaccines. It defines immunization and describes the different types including passive and active immunization.
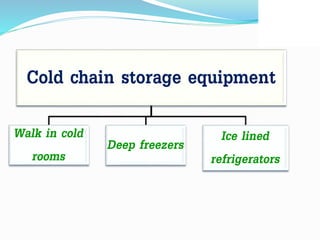
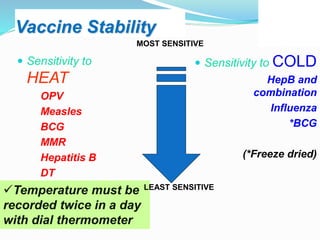
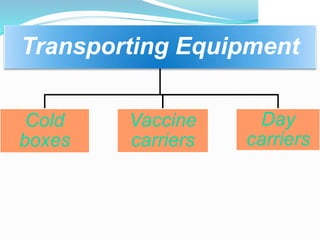
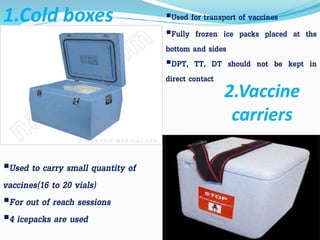
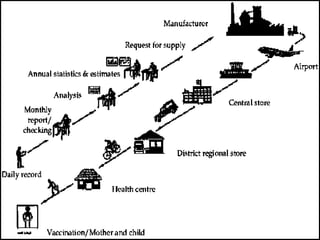
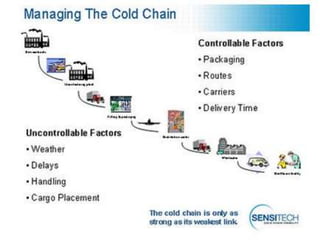
2. The cold chain is described as the system used to transport and store vaccines within recommended temperature ranges from manufacture to point of use. Proper storage and transport using equipment like walk-in cold rooms, deep freezers, and ice-lined refrigerators is important to maintain vaccine potency.
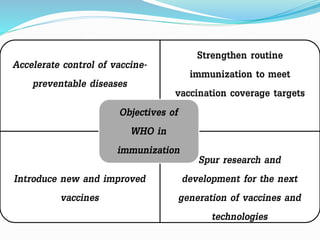
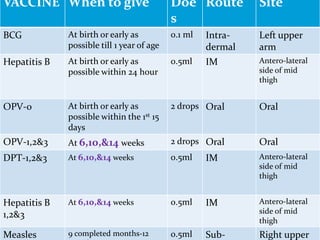
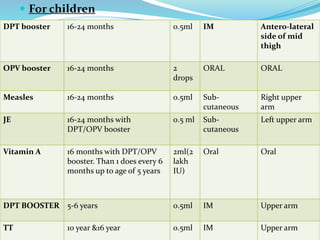
3. The national immunization schedule in India and the vaccines provided under the Universal Immunization Program are summarized, including BCG, DPT, OPV, measles, hepatitis B, and TT vaccines aimed at preventing various diseases.