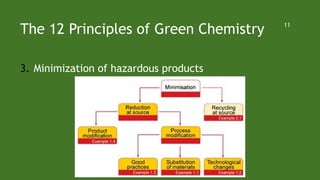
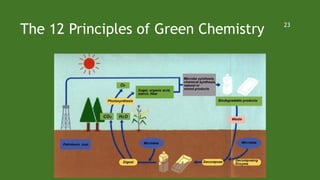
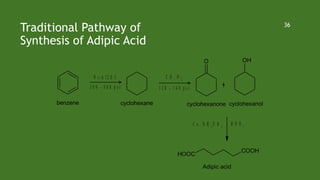
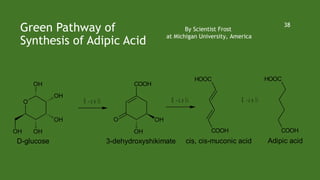
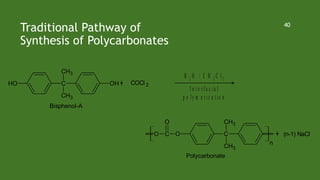
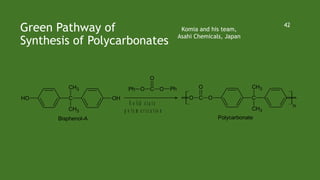
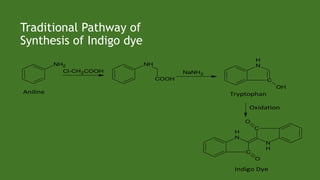
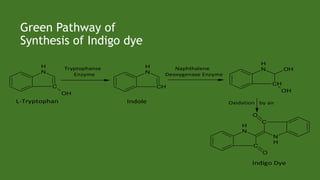
Green chemistry aims to reduce environmental impacts through innovative material choices and chemical processes that minimize waste, hazards, and energy usage. The 12 principles of green chemistry emphasize prevention of waste, safer solvents, energy efficiency, and the use of renewable resources. Green chemistry represents a transformative approach to chemical synthesis, as demonstrated by greener pathways for synthesizing compounds like adipic acid and indigo dye.