Functional group in Biiology
•Download as PPTX, PDF•
0 likes•71 views
Functional group has specific properties, when attached to a larger molecule, in turn give the larger molecules specific properties.
Report
Share
Report
Share
Recommended
Bio chapter 4 – Carbon

KEY CONCEPTS
4.1 Organic chemistry is the study of carbon compounds
4.2 Carbon atoms can form diverse molecules by bonding to four other atoms
4.3 A few chemical groups are key to molecular function
Lewis diagrams lesson

This is a small presentation designed around Lewis Numbers. The content would be appropriate for a grade 10 Physical Science learner, in a CAPS curriculum school.
Recommended
Bio chapter 4 – Carbon

KEY CONCEPTS
4.1 Organic chemistry is the study of carbon compounds
4.2 Carbon atoms can form diverse molecules by bonding to four other atoms
4.3 A few chemical groups are key to molecular function
Lewis diagrams lesson

This is a small presentation designed around Lewis Numbers. The content would be appropriate for a grade 10 Physical Science learner, in a CAPS curriculum school.
Exposición de oxidos metalicos

Es un resumen de como se forman los óxidos metálicos y algunos ejercicios
science-subject-for-high-school-9th-grade-chemistry.pptx

chemistry 9
relating the characteristics of carbon atom to the structure and properties of hydrocarbons
More Related Content
What's hot
Exposición de oxidos metalicos

Es un resumen de como se forman los óxidos metálicos y algunos ejercicios
What's hot (18)
Similar to Functional group in Biiology
science-subject-for-high-school-9th-grade-chemistry.pptx

chemistry 9
relating the characteristics of carbon atom to the structure and properties of hydrocarbons
Tiếng Anh chuyên ngành Sinh học [04 lecture presentation]![Tiếng Anh chuyên ngành Sinh học [04 lecture presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Tiếng Anh chuyên ngành Sinh học [04 lecture presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
(Base on Campbell's biology)
DO NOT RE-UP!!!
Leave your mail if you want to download this book!
Amino acids structure and properties

Detailed Amino acid structure, Zwitter ions, acid base properties of Amino acids, Chirality, L and D forms of amino acids,standard and non standard amino acids, Essential and non essential amino acids,Learn all amino acids, their properties in detail,methods to quantify amino acids
Isomerism - Structural Isomerisms in Organic compounds

Structural Isomerism in Organic compounds - Chain Isomerism, Positional Isomerism, Functional Isomerism, Metamerism and Tautomerism explained with suitable examples. In Introduction, Definition of Isomerism and Stereoisomerism given simply to understand the difference between Structural and Stereo isomerism.
https://youtu.be/a0snq_oz50A
Stereochemistry

The aim in this lecture is to study
1. isomer
2. Enantiomers
3. Chiral carbon atom
4. Optical activity
5. Mesocompound
6.
Dr.S.K Isomerism.pptx

Isomerism
History
In the year 1827, chemistry scholar Friedrich Woehler prepared cyanic acid and got surprised that the chemical composition of the formed substance was identical to the fulminic acid but the properties of this acid are very much different. This finding created shockwaves in the science scholar community as at that time it was considered that chemical compounds can only have different properties if they have different elemental
One of the most essential features of organic molecules is their propensity compositions. After some discoveries of some sort, this was considered a scientific phenomenon and Jacob Berzelius named this phenomenon Isomerism.
What is isomerism?
to show isomerism. Two or more compounds with the same molecular formula but different physical and chemical properties are known as isomers, and this property of a compound is known as Isomerism.
Types of Isomerism
Isomerism can be classified into two categories as follows:
• Structural or constitutional isomerism
• Stereoisomerism
Structural Isomerism
The constitution of a molecule refers to the quantity, kind, and connection of its atoms. The structural isomers or constitutional isomers differ in the connection of their atoms, and this phenomenon is known as structural isomerism. It is also known as constitutional isomerism.
Because they have the same molecular formula, ethanol, and dimethyl ether are isomers. C2H6O. Because the atoms in each chemical are bonded differently, they are structural isomers. The following are some examples of structural isomerism in various forms:
• Chain isomerism
• Position isomerism
• Functional isomerism
• Metamerism
• Tautomerism
• Ring chain isomerism
Chain Isomerism
Chain isomers are isomeric substances that differ solely in the arrangement of carbon atoms in the base chain, and chain isomerism is isomerism between them. The carbon atom chain or skeleton differs amongst isomers of this type.
Carbon atoms can be arranged in a straight or branched chain. Chain isomerism does not exist in molecules comprising up to three carbon atoms since they are a continuous chain with no chance of branching. It is demonstrated for compounds with four or more carbon atoms.
• Chain isomerism in alkanes: Alkanes containing four or more carbon atoms. For example, Two chain isomers are represented by the chemical formula C4H10.
• Chain isomerism in alkenes: When writing chain isomers for alkenes, the position of the double bond should not be modified. For example, Two chain isomers are represented by the chemical formula C4H8.
Position Isomerism
Position isomerization is a phenomenon that occurs when two or more compounds differ in the position of a component atom or functional group on the carbon skeleton.
C3H8O, for example, is made up of two alcohols:
Functional Isomerism
Two or more molecules with the same chemical formula but distinct functional groups are called functional isomers. Functional isomerism is the term for this occurrence.
Similar to Functional group in Biiology (20)
science-subject-for-high-school-9th-grade-chemistry.pptx

science-subject-for-high-school-9th-grade-chemistry.pptx
\\D Nashome\Users\G00589026\My Documents\Bio Ch 4 Pwpt

\\D Nashome\Users\G00589026\My Documents\Bio Ch 4 Pwpt
Tiếng Anh chuyên ngành Sinh học [04 lecture presentation]![Tiếng Anh chuyên ngành Sinh học [04 lecture presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Tiếng Anh chuyên ngành Sinh học [04 lecture presentation]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Tiếng Anh chuyên ngành Sinh học [04 lecture presentation]
Isomerism - Structural Isomerisms in Organic compounds

Isomerism - Structural Isomerisms in Organic compounds
More from Ekhlaque Khan
Chemical kinetics

This video will help you to understand how chemical reaction are involved in every day of our life and how does it occurs.
Amino acid

It contains an amino group, a carboxyl group and a side-chain that is specific to each amino acid. Act as a building blocks of proteins. forming parts of coenzymes. Works as a precursors for the biosynthesis of molecules such as heme.
Test For Carbohydrate 

This Slide is all about reagents used in Identification tests for Carbohydrate.
Reactions Of Carbohydrates 

This slide will help you to understand about chemical reactions of monosaccharides and Disaccharides. The carbohydrate can can undergo several reactions like oxidation, reduction, esterification, dehydration and tautomerization to give various products.
Water: Structure and Properties

This slides will help you to understand about Water Structure & its Properties
More from Ekhlaque Khan (9)
Recently uploaded
extra-chromosomal-inheritance[1].pptx.pdfpdf![extra-chromosomal-inheritance[1].pptx.pdfpdf](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![extra-chromosomal-inheritance[1].pptx.pdfpdf](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Slide 1: Title Slide
Extrachromosomal Inheritance
Slide 2: Introduction to Extrachromosomal Inheritance
Definition: Extrachromosomal inheritance refers to the transmission of genetic material that is not found within the nucleus.
Key Components: Involves genes located in mitochondria, chloroplasts, and plasmids.
Slide 3: Mitochondrial Inheritance
Mitochondria: Organelles responsible for energy production.
Mitochondrial DNA (mtDNA): Circular DNA molecule found in mitochondria.
Inheritance Pattern: Maternally inherited, meaning it is passed from mothers to all their offspring.
Diseases: Examples include Leber’s hereditary optic neuropathy (LHON) and mitochondrial myopathy.
Slide 4: Chloroplast Inheritance
Chloroplasts: Organelles responsible for photosynthesis in plants.
Chloroplast DNA (cpDNA): Circular DNA molecule found in chloroplasts.
Inheritance Pattern: Often maternally inherited in most plants, but can vary in some species.
Examples: Variegation in plants, where leaf color patterns are determined by chloroplast DNA.
Slide 5: Plasmid Inheritance
Plasmids: Small, circular DNA molecules found in bacteria and some eukaryotes.
Features: Can carry antibiotic resistance genes and can be transferred between cells through processes like conjugation.
Significance: Important in biotechnology for gene cloning and genetic engineering.
Slide 6: Mechanisms of Extrachromosomal Inheritance
Non-Mendelian Patterns: Do not follow Mendel’s laws of inheritance.
Cytoplasmic Segregation: During cell division, organelles like mitochondria and chloroplasts are randomly distributed to daughter cells.
Heteroplasmy: Presence of more than one type of organellar genome within a cell, leading to variation in expression.
Slide 7: Examples of Extrachromosomal Inheritance
Four O’clock Plant (Mirabilis jalapa): Shows variegated leaves due to different cpDNA in leaf cells.
Petite Mutants in Yeast: Result from mutations in mitochondrial DNA affecting respiration.
Slide 8: Importance of Extrachromosomal Inheritance
Evolution: Provides insight into the evolution of eukaryotic cells.
Medicine: Understanding mitochondrial inheritance helps in diagnosing and treating mitochondrial diseases.
Agriculture: Chloroplast inheritance can be used in plant breeding and genetic modification.
Slide 9: Recent Research and Advances
Gene Editing: Techniques like CRISPR-Cas9 are being used to edit mitochondrial and chloroplast DNA.
Therapies: Development of mitochondrial replacement therapy (MRT) for preventing mitochondrial diseases.
Slide 10: Conclusion
Summary: Extrachromosomal inheritance involves the transmission of genetic material outside the nucleus and plays a crucial role in genetics, medicine, and biotechnology.
Future Directions: Continued research and technological advancements hold promise for new treatments and applications.
Slide 11: Questions and Discussion
Invite Audience: Open the floor for any questions or further discussion on the topic.
filosofia boliviana introducción jsjdjd.pptx

La filosofía boliviana y la búsqueda por construir pensamientos propios
THE IMPORTANCE OF MARTIAN ATMOSPHERE SAMPLE RETURN.

The return of a sample of near-surface atmosphere from Mars would facilitate answers to several first-order science questions surrounding the formation and evolution of the planet. One of the important aspects of terrestrial planet formation in general is the role that primary atmospheres played in influencing the chemistry and structure of the planets and their antecedents. Studies of the martian atmosphere can be used to investigate the role of a primary atmosphere in its history. Atmosphere samples would also inform our understanding of the near-surface chemistry of the planet, and ultimately the prospects for life. High-precision isotopic analyses of constituent gases are needed to address these questions, requiring that the analyses are made on returned samples rather than in situ.
Leaf Initiation, Growth and Differentiation.pdf

Leaf initiation, growth and differentiation, genetic control of leaf development.
Richard's entangled aventures in wonderland

Since the loophole-free Bell experiments of 2020 and the Nobel prizes in physics of 2022, critics of Bell's work have retreated to the fortress of super-determinism. Now, super-determinism is a derogatory word - it just means "determinism". Palmer, Hance and Hossenfelder argue that quantum mechanics and determinism are not incompatible, using a sophisticated mathematical construction based on a subtle thinning of allowed states and measurements in quantum mechanics, such that what is left appears to make Bell's argument fail, without altering the empirical predictions of quantum mechanics. I think however that it is a smoke screen, and the slogan "lost in math" comes to my mind. I will discuss some other recent disproofs of Bell's theorem using the language of causality based on causal graphs. Causal thinking is also central to law and justice. I will mention surprising connections to my work on serial killer nurse cases, in particular the Dutch case of Lucia de Berk and the current UK case of Lucy Letby.
Cancer cell metabolism: special Reference to Lactate Pathway

Normal Cell Metabolism:
Cellular respiration describes the series of steps that cells use to break down sugar and other chemicals to get the energy we need to function.
Energy is stored in the bonds of glucose and when glucose is broken down, much of that energy is released.
Cell utilize energy in the form of ATP.
The first step of respiration is called glycolysis. In a series of steps, glycolysis breaks glucose into two smaller molecules - a chemical called pyruvate. A small amount of ATP is formed during this process.
Most healthy cells continue the breakdown in a second process, called the Kreb's cycle. The Kreb's cycle allows cells to “burn” the pyruvates made in glycolysis to get more ATP.
The last step in the breakdown of glucose is called oxidative phosphorylation (Ox-Phos).
It takes place in specialized cell structures called mitochondria. This process produces a large amount of ATP. Importantly, cells need oxygen to complete oxidative phosphorylation.
If a cell completes only glycolysis, only 2 molecules of ATP are made per glucose. However, if the cell completes the entire respiration process (glycolysis - Kreb's - oxidative phosphorylation), about 36 molecules of ATP are created, giving it much more energy to use.
IN CANCER CELL:
Unlike healthy cells that "burn" the entire molecule of sugar to capture a large amount of energy as ATP, cancer cells are wasteful.
Cancer cells only partially break down sugar molecules. They overuse the first step of respiration, glycolysis. They frequently do not complete the second step, oxidative phosphorylation.
This results in only 2 molecules of ATP per each glucose molecule instead of the 36 or so ATPs healthy cells gain. As a result, cancer cells need to use a lot more sugar molecules to get enough energy to survive.
Unlike healthy cells that "burn" the entire molecule of sugar to capture a large amount of energy as ATP, cancer cells are wasteful.
Cancer cells only partially break down sugar molecules. They overuse the first step of respiration, glycolysis. They frequently do not complete the second step, oxidative phosphorylation.
This results in only 2 molecules of ATP per each glucose molecule instead of the 36 or so ATPs healthy cells gain. As a result, cancer cells need to use a lot more sugar molecules to get enough energy to survive.
introduction to WARBERG PHENOMENA:
WARBURG EFFECT Usually, cancer cells are highly glycolytic (glucose addiction) and take up more glucose than do normal cells from outside.
Otto Heinrich Warburg (; 8 October 1883 – 1 August 1970) In 1931 was awarded the Nobel Prize in Physiology for his "discovery of the nature and mode of action of the respiratory enzyme.
WARNBURG EFFECT : cancer cells under aerobic (well-oxygenated) conditions to metabolize glucose to lactate (aerobic glycolysis) is known as the Warburg effect. Warburg made the observation that tumor slices consume glucose and secrete lactate at a higher rate than normal tissues.
Unveiling the Energy Potential of Marshmallow Deposits.pdf

Unveiling the Energy Potential of Marshmallow Deposits: A Revolutionary
Breakthrough in Sustainable Energy Science
Comparative structure of adrenal gland in vertebrates

Adrenal gland comparative structures in vertebrates
Multi-source connectivity as the driver of solar wind variability in the heli...

The ambient solar wind that flls the heliosphere originates from multiple
sources in the solar corona and is highly structured. It is often described
as high-speed, relatively homogeneous, plasma streams from coronal
holes and slow-speed, highly variable, streams whose source regions are
under debate. A key goal of ESA/NASA’s Solar Orbiter mission is to identify
solar wind sources and understand what drives the complexity seen in the
heliosphere. By combining magnetic feld modelling and spectroscopic
techniques with high-resolution observations and measurements, we show
that the solar wind variability detected in situ by Solar Orbiter in March
2022 is driven by spatio-temporal changes in the magnetic connectivity to
multiple sources in the solar atmosphere. The magnetic feld footpoints
connected to the spacecraft moved from the boundaries of a coronal hole
to one active region (12961) and then across to another region (12957). This
is refected in the in situ measurements, which show the transition from fast
to highly Alfvénic then to slow solar wind that is disrupted by the arrival of
a coronal mass ejection. Our results describe solar wind variability at 0.5 au
but are applicable to near-Earth observatories.
Structural Classification Of Protein (SCOP)

A brief information about the SCOP protein database used in bioinformatics.
The Structural Classification of Proteins (SCOP) database is a comprehensive and authoritative resource for the structural and evolutionary relationships of proteins. It provides a detailed and curated classification of protein structures, grouping them into families, superfamilies, and folds based on their structural and sequence similarities.
Recently uploaded (20)
ESR_factors_affect-clinic significance-Pathysiology.pptx

ESR_factors_affect-clinic significance-Pathysiology.pptx
THE IMPORTANCE OF MARTIAN ATMOSPHERE SAMPLE RETURN.

THE IMPORTANCE OF MARTIAN ATMOSPHERE SAMPLE RETURN.
platelets- lifespan -Clot retraction-disorders.pptx

platelets- lifespan -Clot retraction-disorders.pptx
Cancer cell metabolism: special Reference to Lactate Pathway

Cancer cell metabolism: special Reference to Lactate Pathway
Unveiling the Energy Potential of Marshmallow Deposits.pdf

Unveiling the Energy Potential of Marshmallow Deposits.pdf
PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION

PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION
Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...

Circulatory system_ Laplace law. Ohms law.reynaults law,baro-chemo-receptors-...
Comparative structure of adrenal gland in vertebrates

Comparative structure of adrenal gland in vertebrates
Multi-source connectivity as the driver of solar wind variability in the heli...

Multi-source connectivity as the driver of solar wind variability in the heli...
Lateral Ventricles.pdf very easy good diagrams comprehensive

Lateral Ventricles.pdf very easy good diagrams comprehensive
Functional group in Biiology
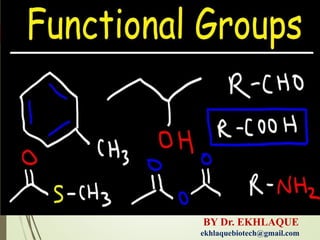
- 2. Functional groups give specific properties found together in a variety of different molecules. Each functional group has specific properties that, when attached to a larger molecule, in turn give the larger molecules specific properties. several functional groups in biology, including alcohols, aldehydes, ketones, acids, amines, phosphates, and thiols.
- 4. An important biological molecules containing functional groups is the amino acids, which have both a carboxyl group and an amino group attached to the same carbon atom, called the α carbon. Also attached to the α carbon atom are a hydrogen atom and a side chain, or R group Different side chains have different chemical compositions, structures, and properties. Each of the 20 amino acids found in proteins has a different side chain that gives it its distinctive chemical properties. Because they possess both carboxyl and amino groups, amino acids are simultaneously acids and bases.
- 5. Isomers have different arrangements of the same atoms Isomers are molecules that have the same chemical formula but different arrangements of the atoms. (The prefix iso- meaning “same,”). we will consider two: structural isomers and optical isomers.
- 6. Structural isomers It differ in how their atoms are joined together. Consider two simple molecules, each composed of 4 carbon and 10 hydrogen atoms bonded covalently, both with the formula C4H10. These atoms can be linked in two different ways, resulting in two forms of the molecule:
- 7. Optical isomers The carbon atom has four different atoms or groups attached to it. This pattern allows two different ways of making the attachments, each the mirror image of the other. Such a carbon atom is an asymmetrical carbon, and the pair of compounds are optical isomers of each other. You can imagine your right and left hands as optical isomers. Just as a glove is specific for a particular hand, some biochemical molecules can interact with one optical isomer of a compound, but are unable to “fit” the other.
- 9. Cont… The α carbon in an amino acid is an asymmetrical carbon because it is bonded to four different functional groups. Therefore, amino acids exist in two isomeric forms, called D-amino acids and L-amino acids. D and L stand for right (dextro) and left (levo), respectively. Only L-amino acids are commonly found in most organisms, and their presence is an important chemical “signature” for life.
- 11. Molecules vary in size Some are small, such as H2 and CH4. Others are larger, such as a molecule of table sugar (Sucrose, C12H22O11), which has 45 atoms. Still other molecules, especially proteins such as hemoglobin, are gigantic, sometimes containing tens of thousands of atoms.