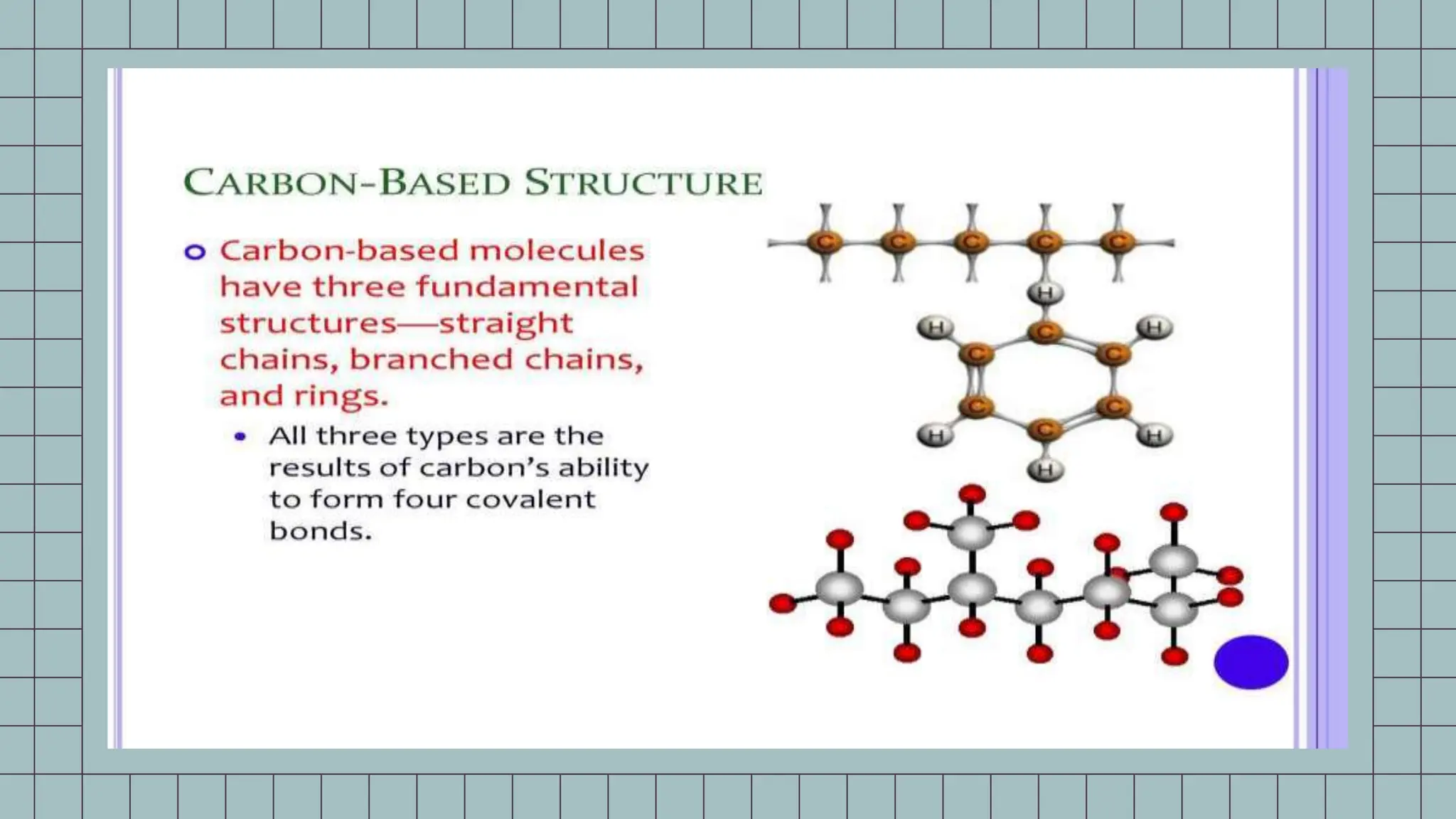
The document explains the significance of carbon atoms in the structure and properties of hydrocarbons, highlighting their ability to form stable compounds due to four valence electrons. Key concepts include the formation of chains and rings, isomerism, and the impact of functional groups on chemical reactivity. Additionally, it emphasizes the combustibility of hydrocarbons due to their high carbon content.