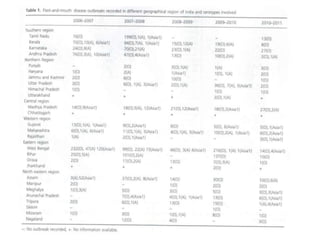
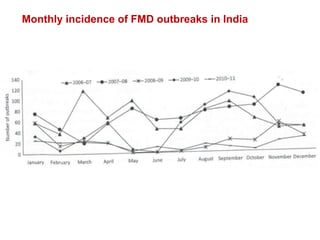
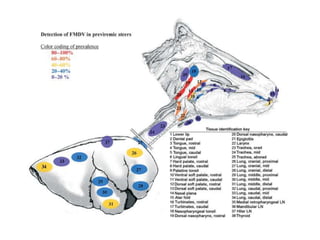
This document summarizes information about foot-and-mouth disease (FMD). It describes FMD virus, the clinical signs of disease, diagnosis, and pathogenesis. Key points include:
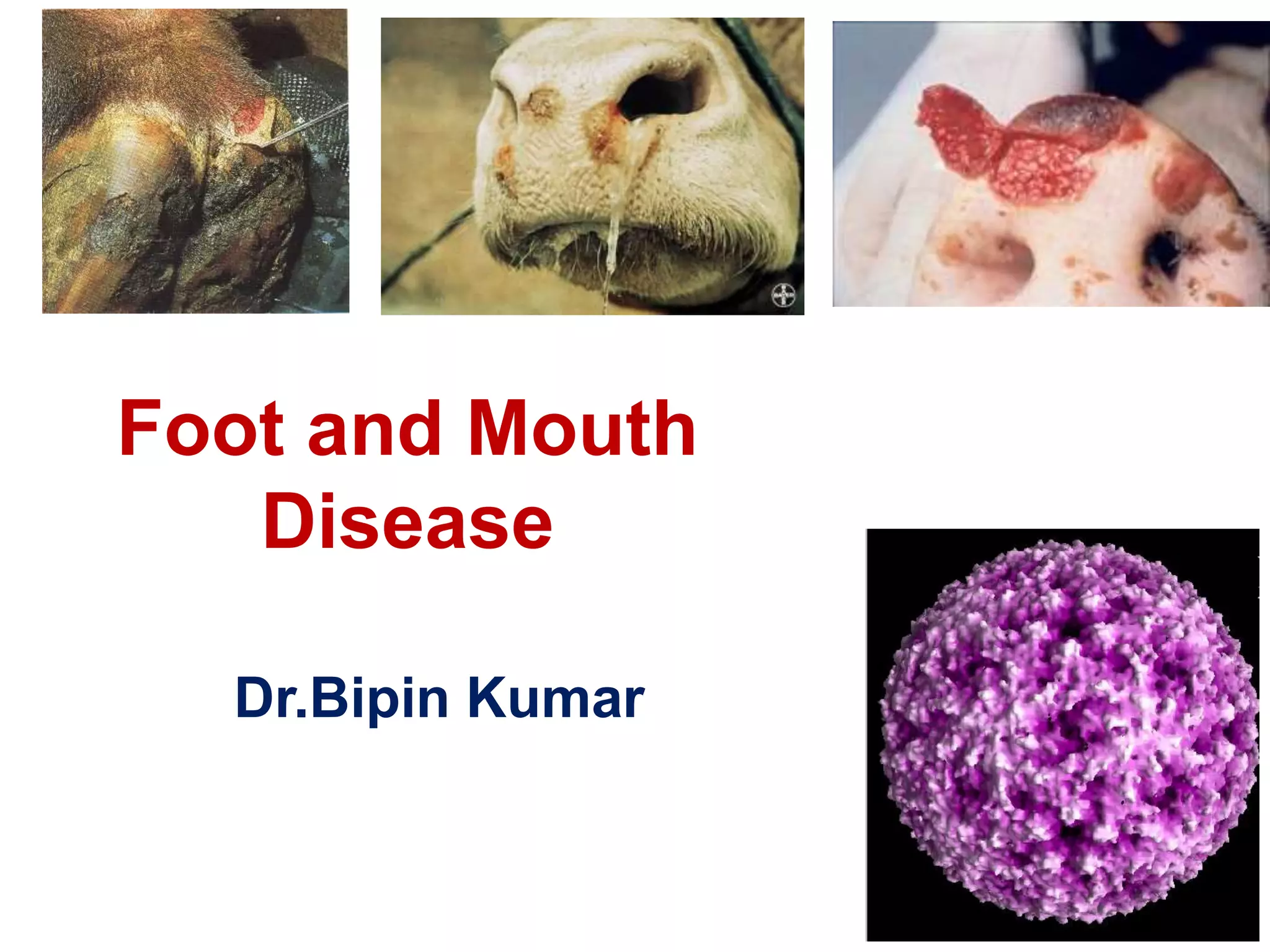
- FMD virus causes an acute, highly contagious disease in cloven-hoofed animals characterized by fever and vesicles around the mouth and feet.
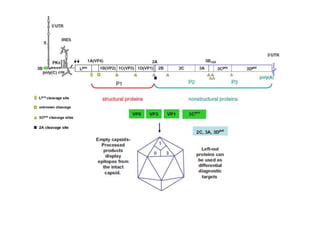
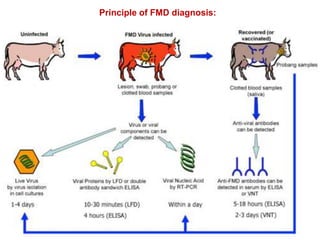
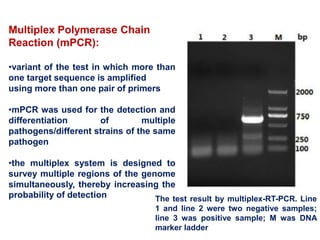
- The virus exists in 7 types and many subtypes that are diagnosed using techniques like virus isolation, ELISA, and PCR.
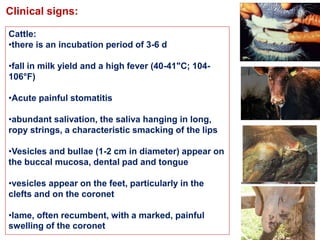
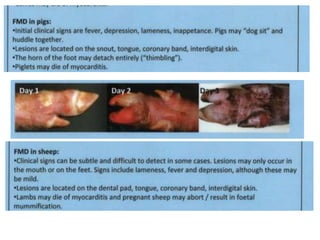
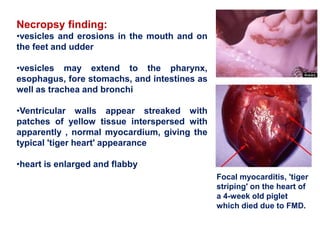
- Clinical signs vary by species but include lameness, vesicles, and decreased milk production. The disease has significant economic impacts.
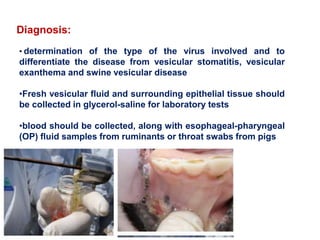
- Diagnosis involves detecting viral antigen or antibodies. Techniques include virus isolation, ELISA, and PCR which are