Electron Configuration
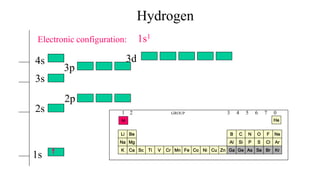
- 1. 1s 2s 2p 3s 3p 3d4s Electronic configuration: FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He 1s1 Ne Ar Kr Hydrogen 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 2. Helium 1s 2s 2p 3s 3p 3d4s Electronic configuration: 1s2 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 3. 1s 2s 2p 3s 3p 3d4s Electronic configuration : 1s2 Lithium 2s1 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 4. 1s 2s 2p 3s 3p 3d4s Electronic configuration : 1s2 Beryllium 2s2 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 5. 1s 2s 2p 3s 3p 3d4s Electronic configuration : 2p1 Boron 2s21s2 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 6. 1s 2s 2p 3s 3p 3d4s Electronic configuration : 1s2 Carbon 2s22p2 The next electron does not pair with the first electron in 2p, but occupies the empty box (orbital) to keep them as far apart as possible. FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 7. 1s 2s 2p 3s 3p 3d4s Electronic configuration : 1s2 Nitrogen 2s22p3 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 8. 1s 2s 2p 3s 3p 3d4s Electronic configuration : 2p4 Oxygen 2s21s2 Now that all 3 p orbitals are filled the next electron must pair up. FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 9. 1s 2s 2p 3s 3p 3d4s Electronic configuration : 1s22s22p5 Fluorine FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 10. Neon 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p6 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 11. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s1 Sodium FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 12. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 Magnesium FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 13. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p1 Aluminium FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 14. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p2 Silicon FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 15. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p3 Phosphorus FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 16. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p4 Sulphur FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 17. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p5 Chlorine FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 18. Argon 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 19. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s1 Potassium FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 20. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s2 Calcium FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 21. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s2 3d1 Scandium FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 22. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s2 3d2 Titanium FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 23. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s2 3d3 Vanadium FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 24. Chromium 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s1 3d5 Notice that one of the 4s electrons has been transferred to 3d so that 3d is now a half filled shell with extra stability. 4s and 3d contain only unpaired electrons. FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 25. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 3d5 Manganese 4s2 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 26. Iron 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s23d6 FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 27. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s23d7 Cobalt FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 28. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s23d8 Nickel FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 29. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 4s13d10 Copper Notice that again one of the 4s electrons has been promoted to 3d so that 3d is now a completely filled shell with extra stability. FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B
- 30. 1s 2s 2p 3s 3p 3d4s 1s2Electronic configuration: 2s22p63s2 3p6 3d104s2 Zinc FeMnVTiScK Na ZnCuNiCr Co Mg Ca Be H As O S Se F Cl BrGa C Si Ge N P He Ne Ar Kr 1 2 GROUP 3 4 5 6 7 0 Li Al B