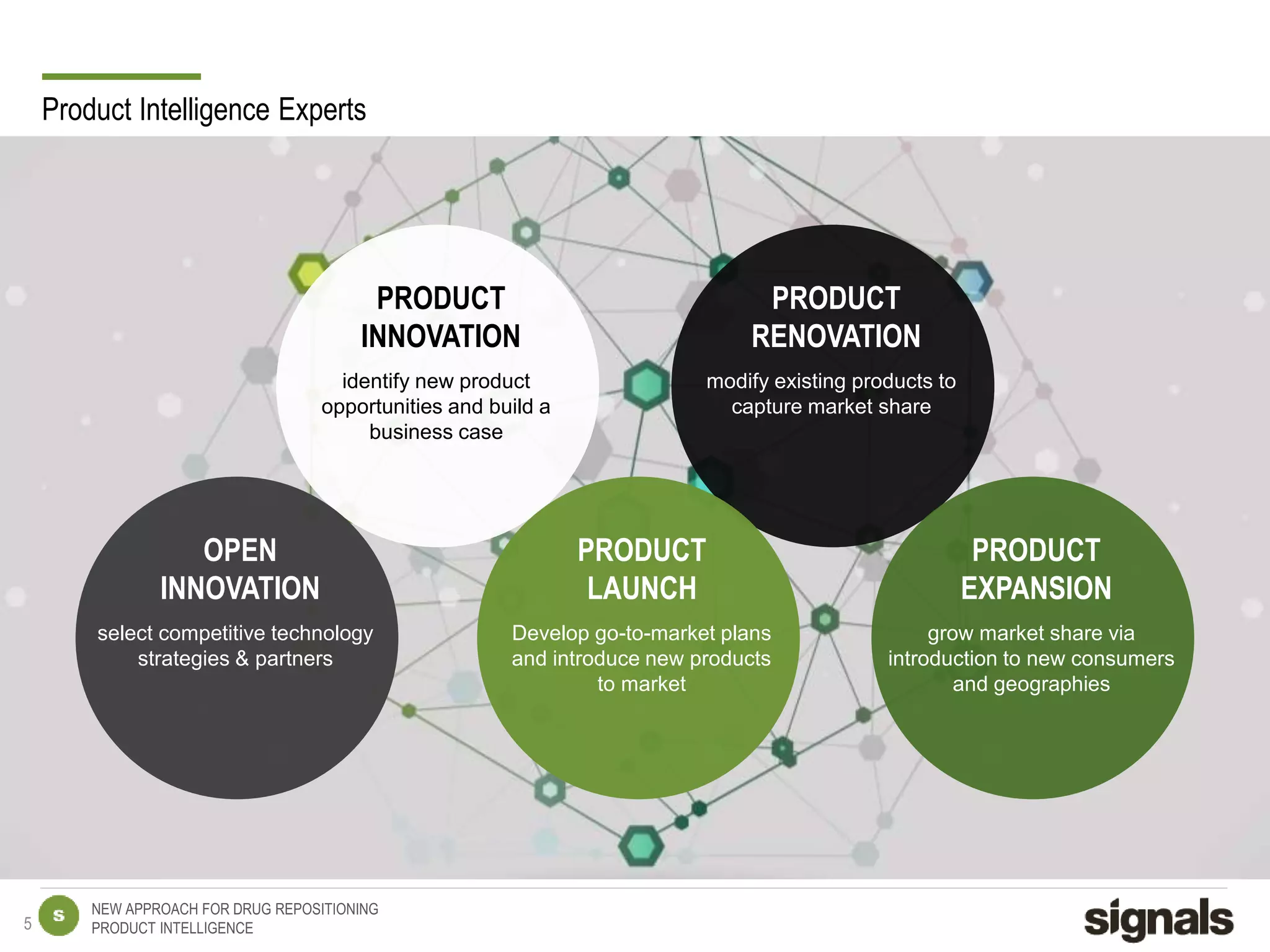
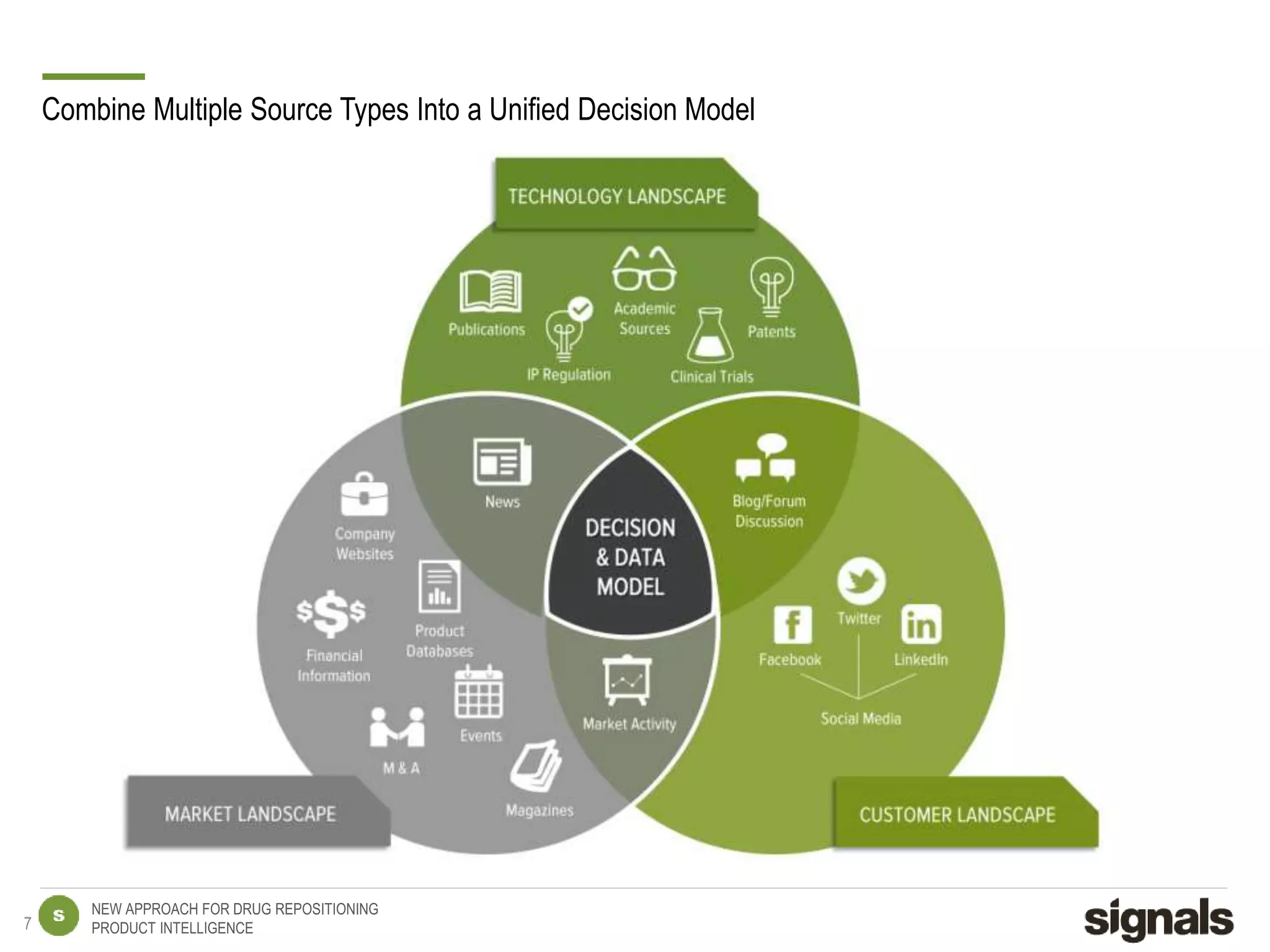
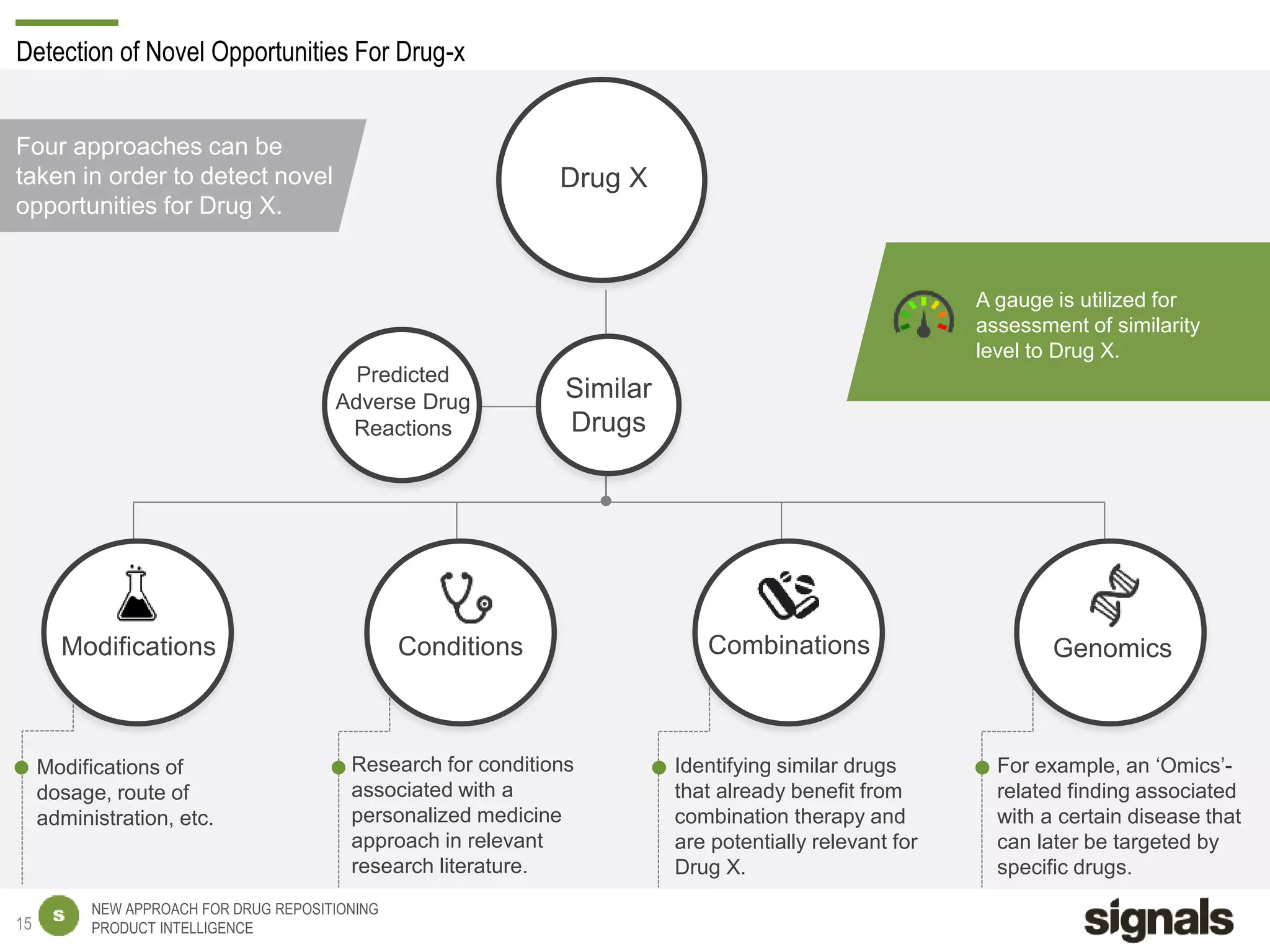
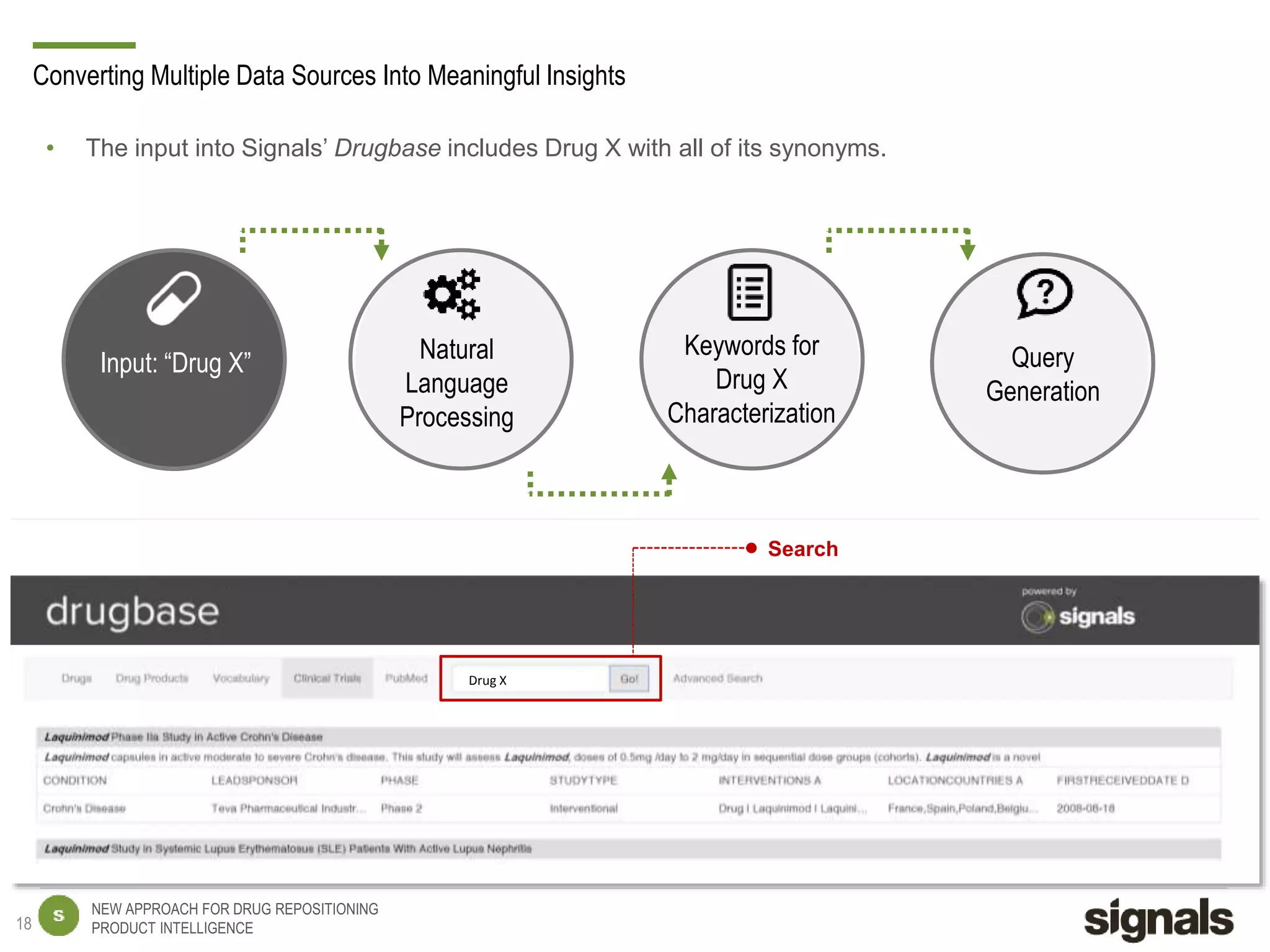
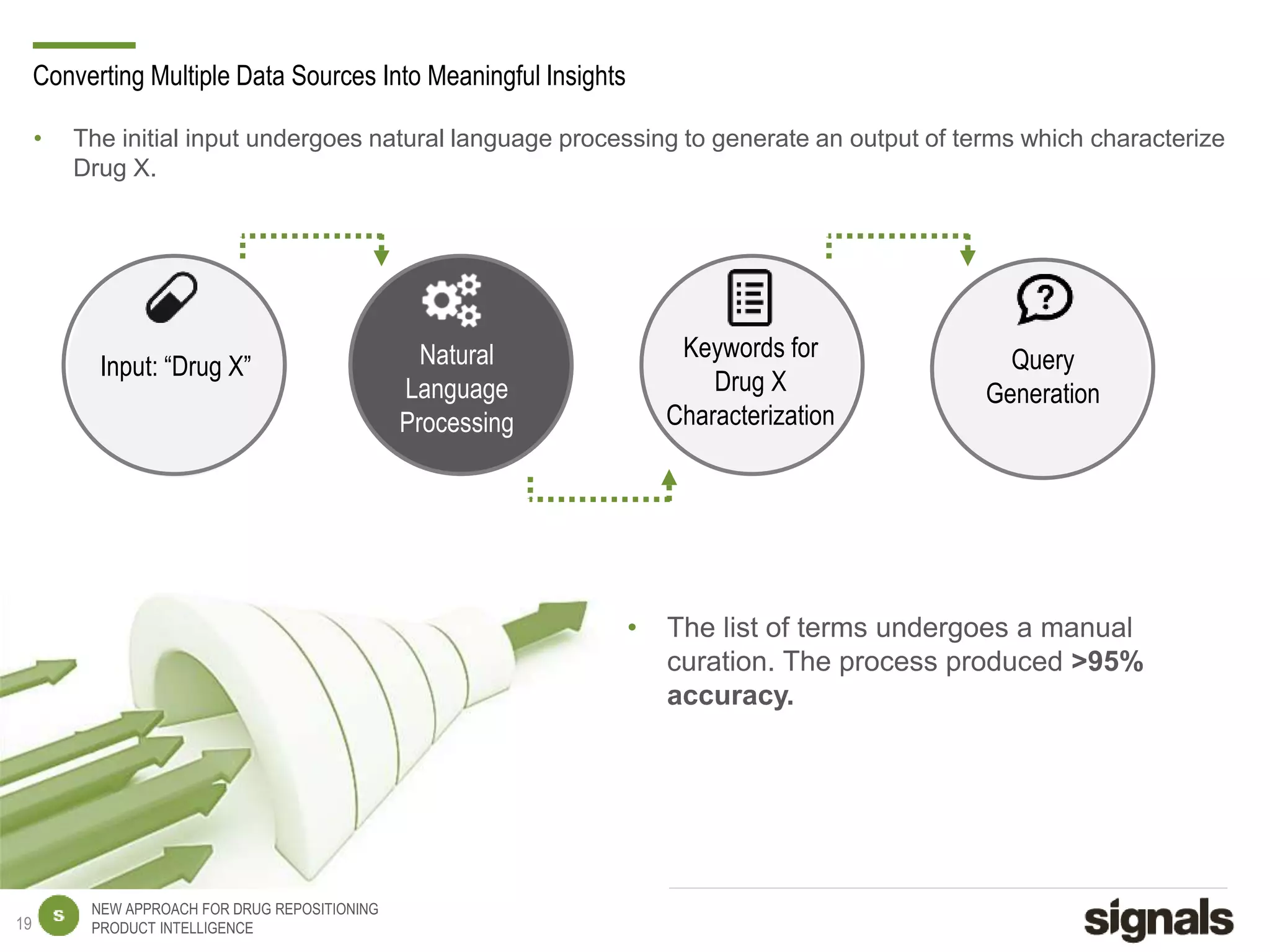
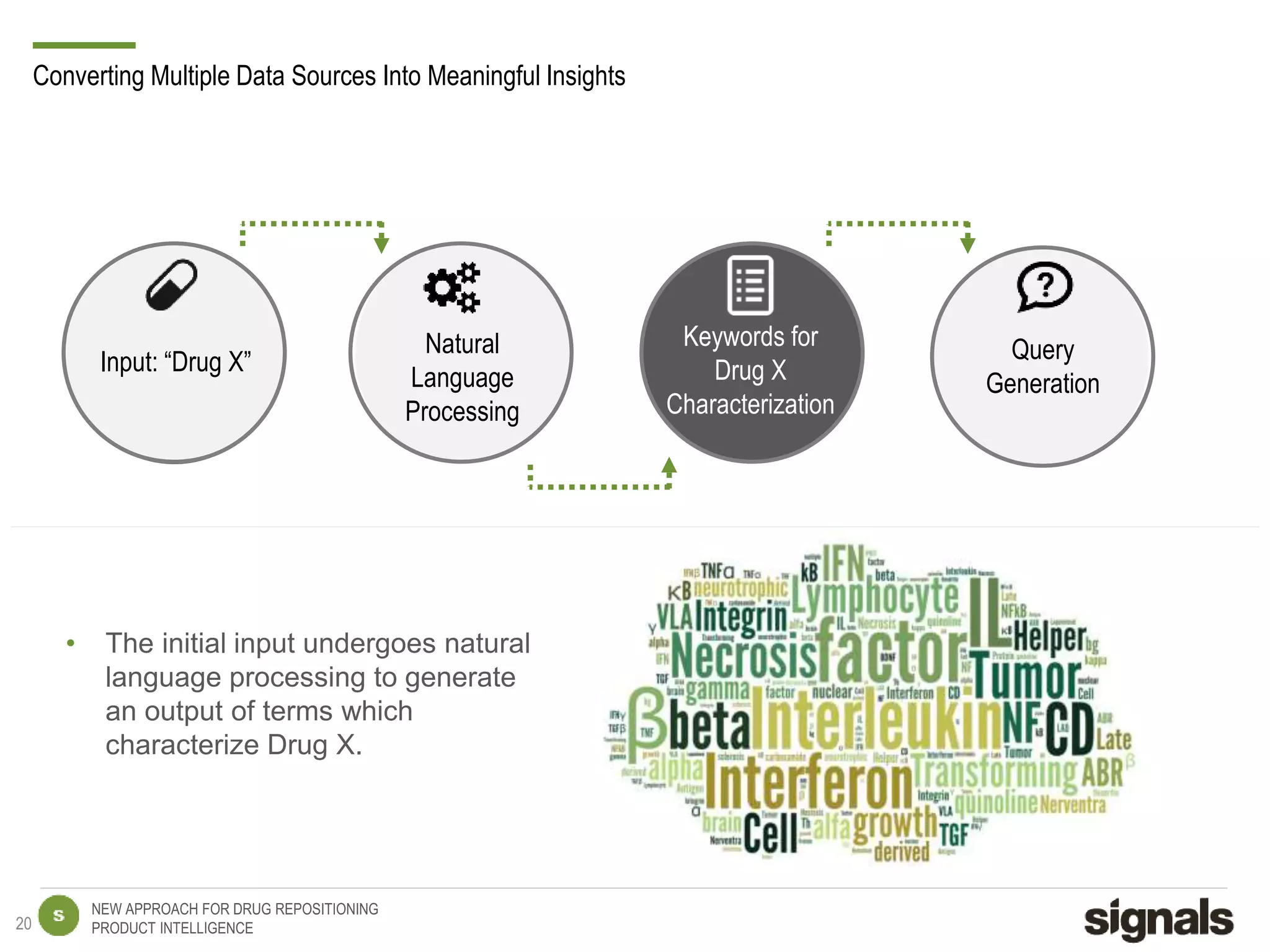
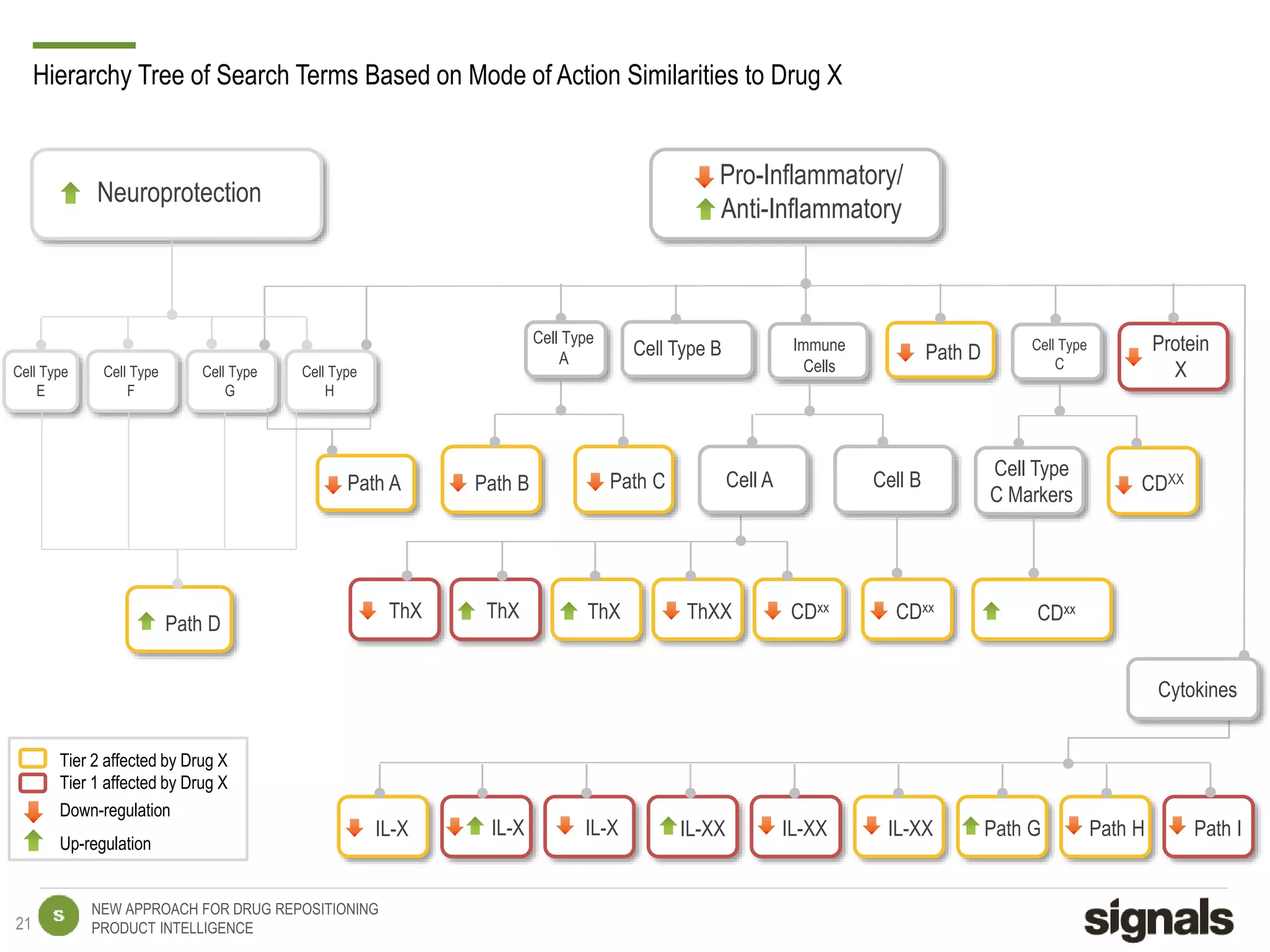
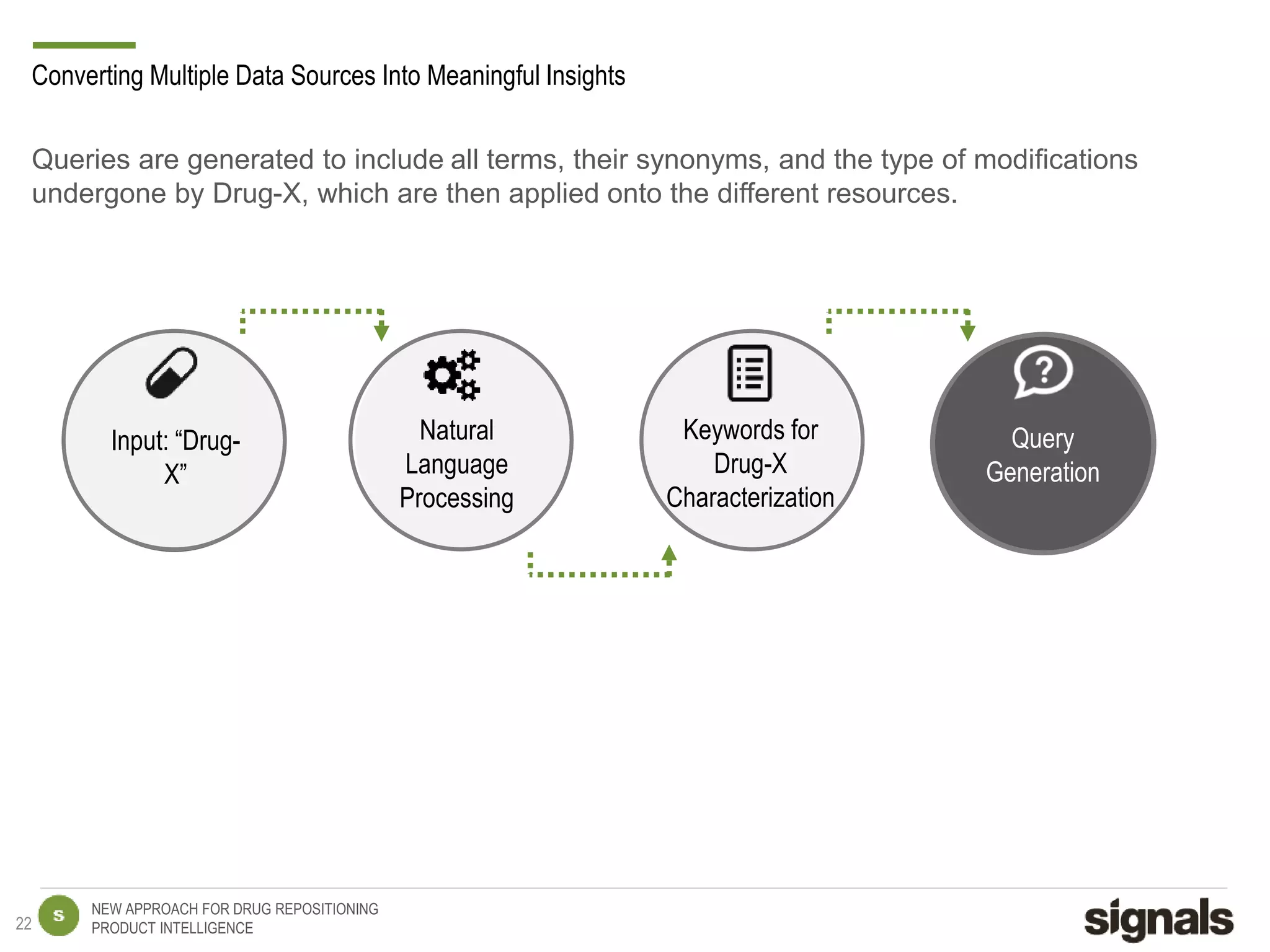
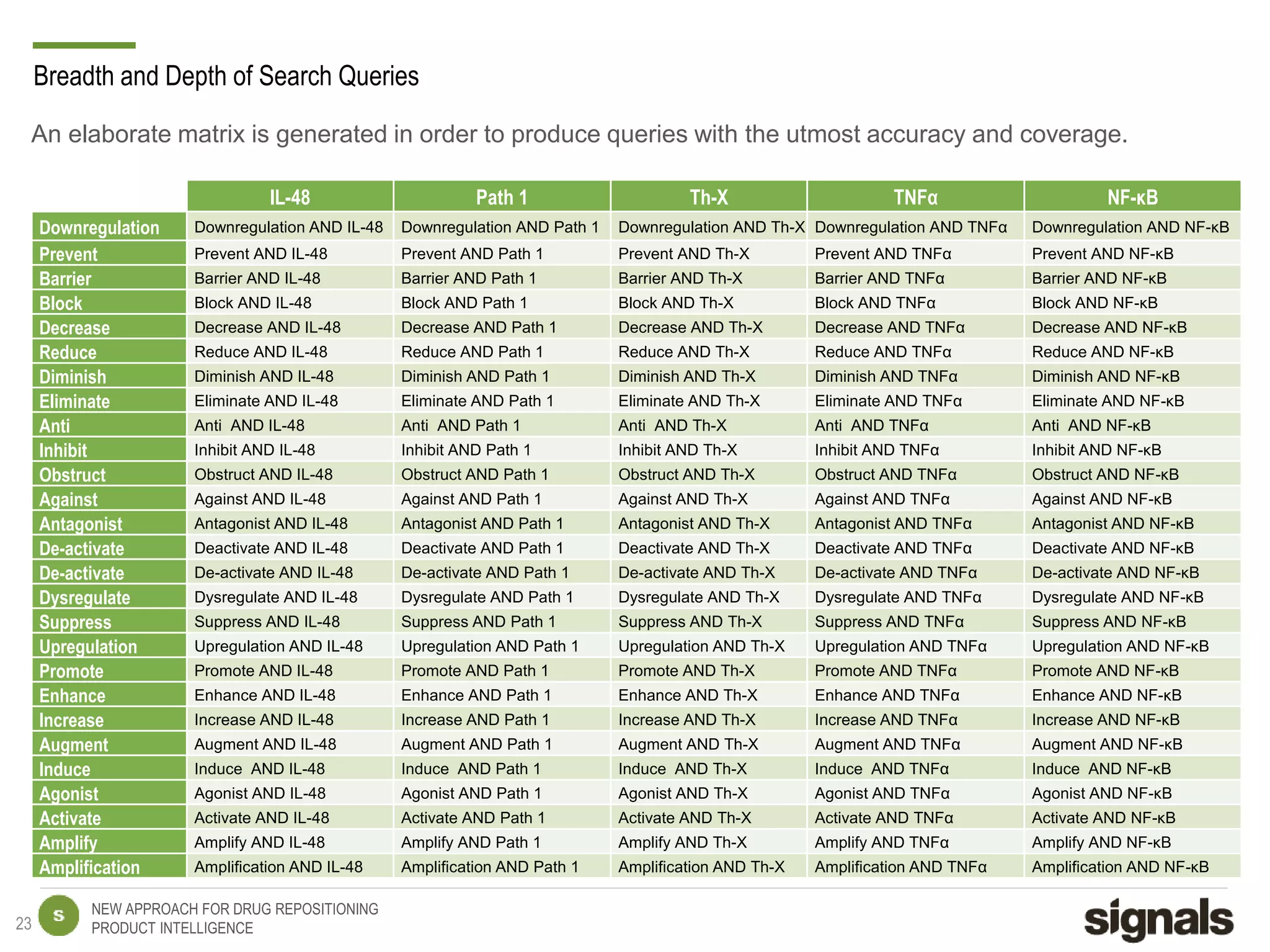
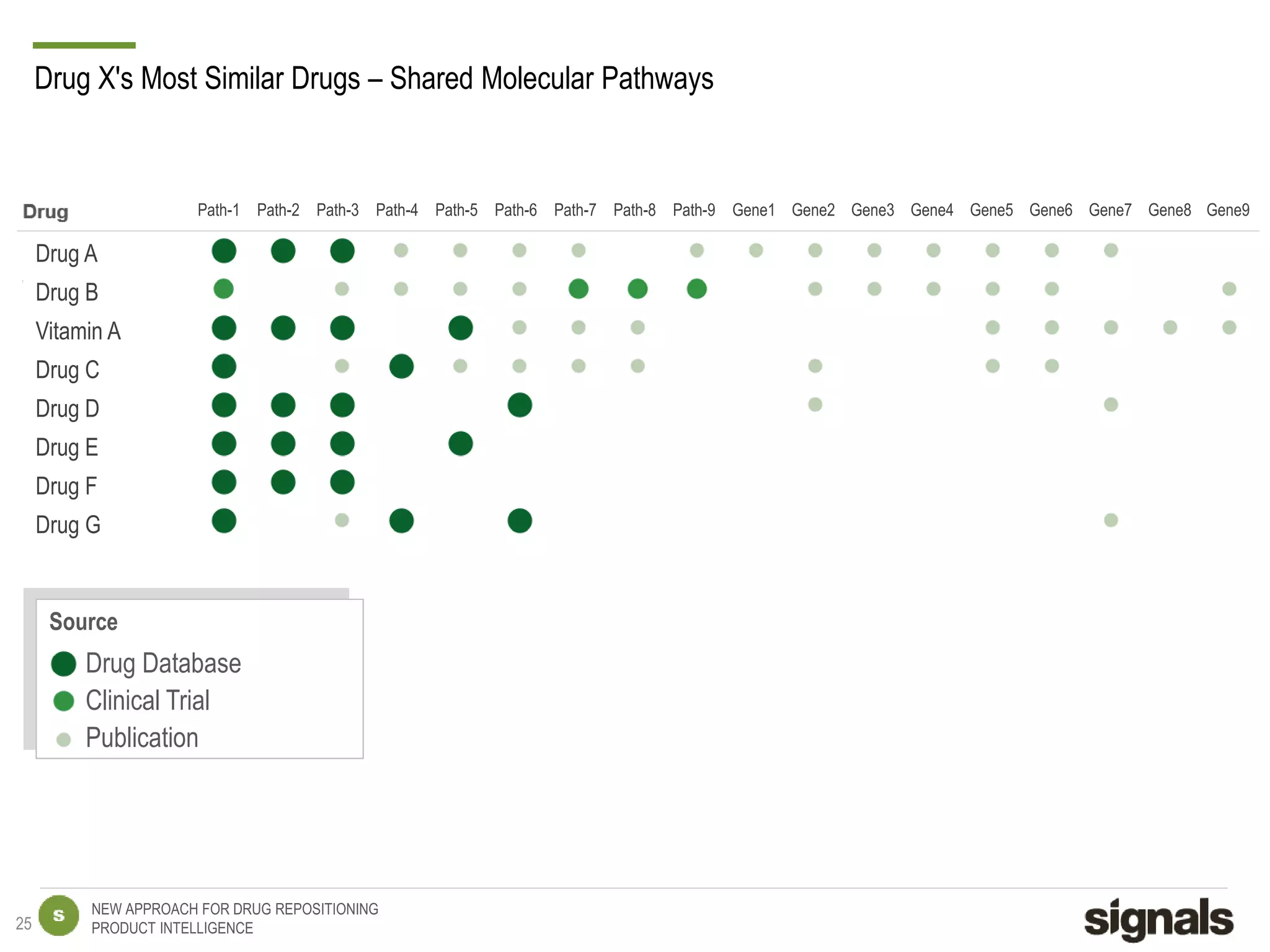
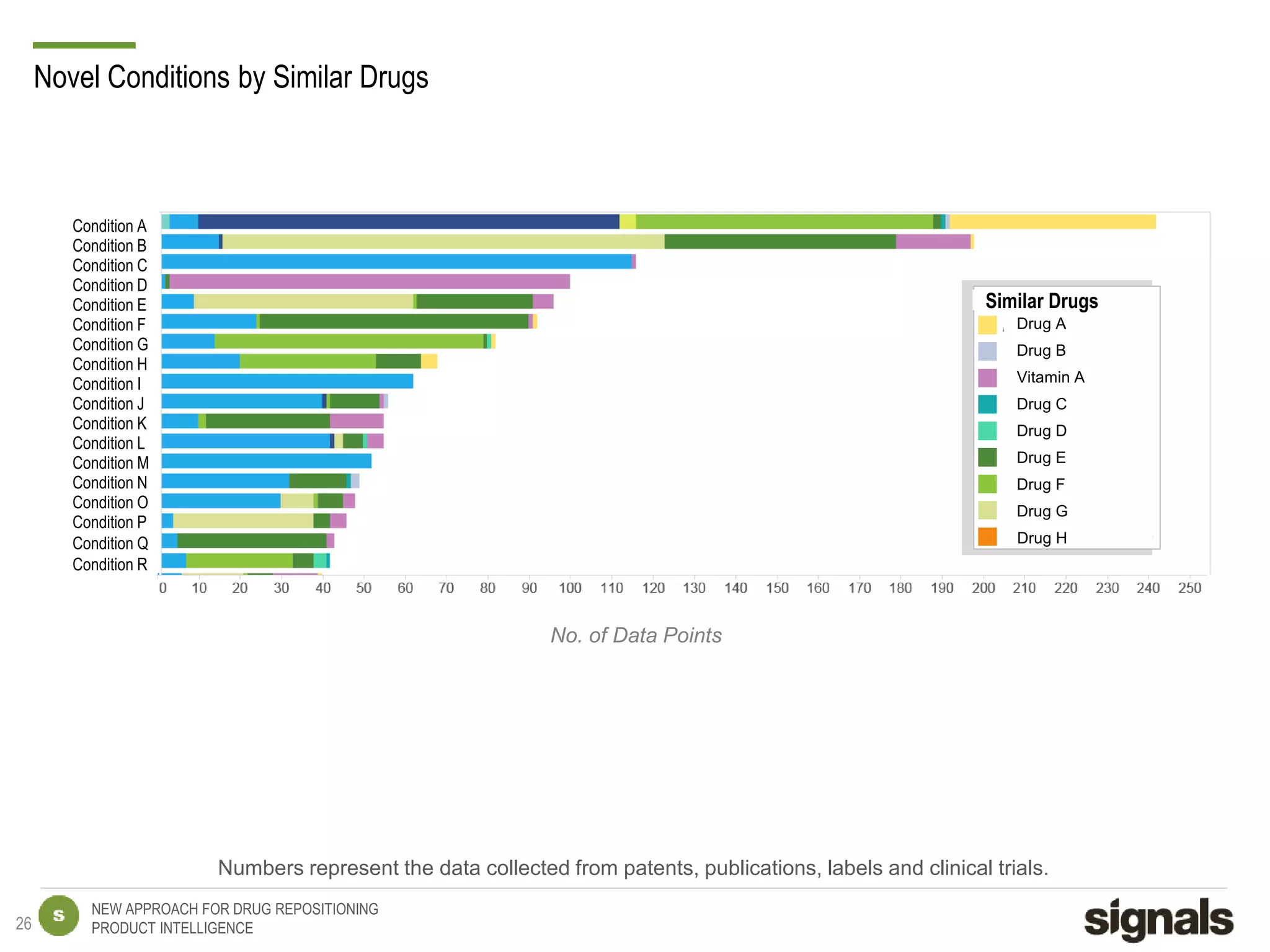
The document discusses Signals' approach to drug repositioning using big data. It introduces Signals and their product intelligence expertise. Their solution automatically produces and delivers business analytics by collecting, integrating and analyzing big data from open web sources. The presentation discusses the challenges in drug development, need for repositioning, and Signals' evidence-based data model and methodology for characterizing a drug and generating queries to identify novel opportunities for increasing its ROI by detecting similar drugs, modifications, conditions and genomic data.