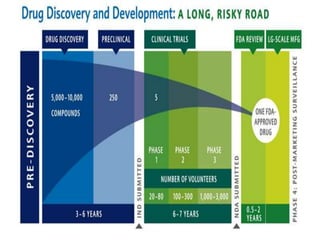
Pharmacometrics is the science of using mathematical and statistical methods to characterize and predict the pharmacokinetic and pharmacodynamic behavior of drugs. It aims to improve decision making in drug development and pharmacotherapy. Pharmacometric models integrate pharmacokinetic and pharmacodynamic models to describe the relationship between drug concentration, effect, and patient characteristics. Population pharmacometric modeling is useful for characterizing variability in these parameters between individuals.