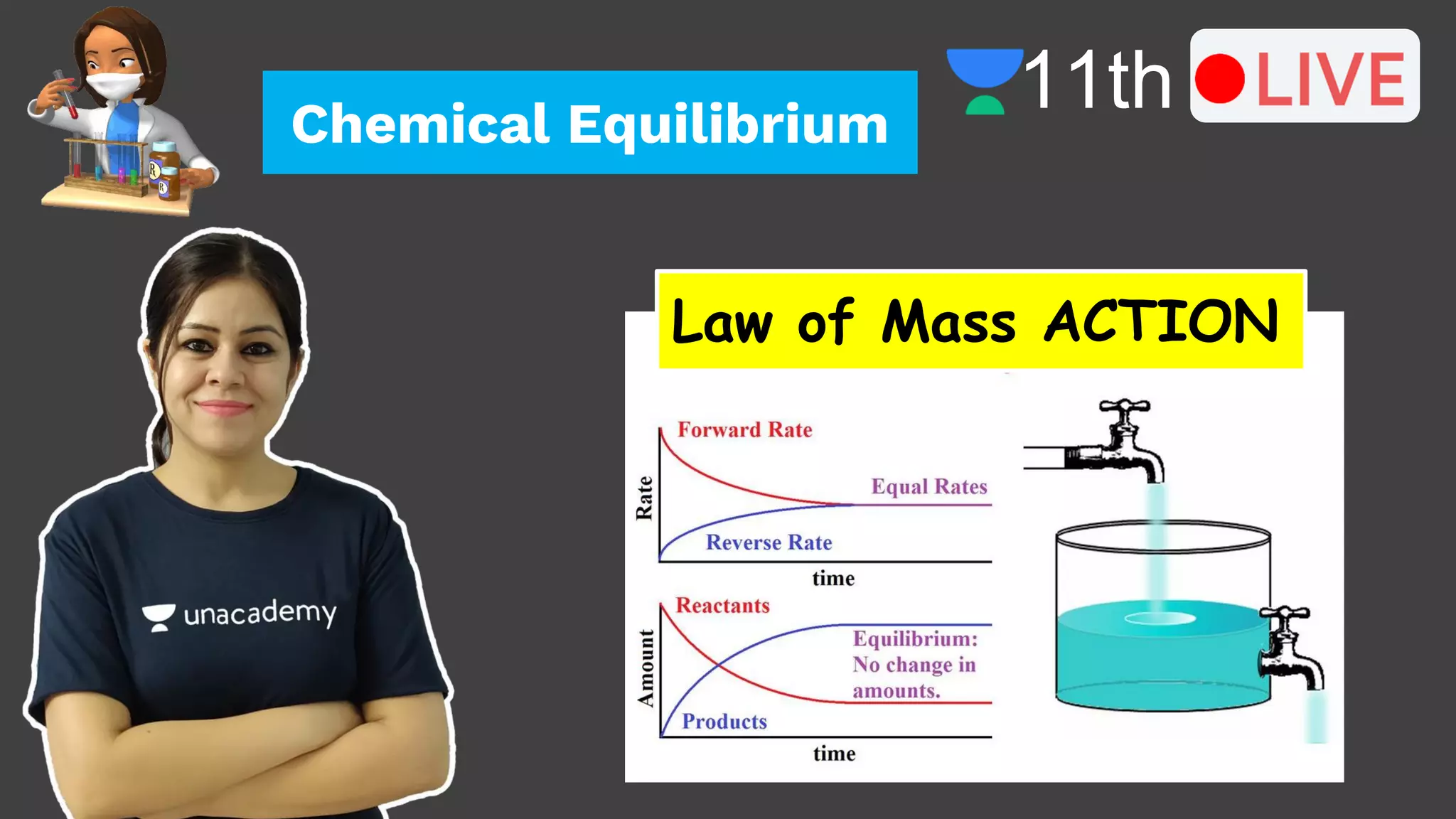
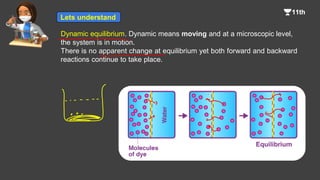
1. The document introduces the concept of chemical equilibrium, including that it is a state where the concentrations of reactants and products do not change over time, and that forward and backward reactions continue at the microscopic level.
2. It describes the law of mass action, which states that the rate of a reaction is directly proportional to the product of the molar concentrations of reactants raised to their stoichiometric coefficients.
3. It provides expressions for calculating equilibrium constants (Kc) in both gaseous and liquid/solid systems based on the law of mass action and reaction stoichiometry.



















![For any general reaction,
aA + bB-------> Products
The law of mass action may be written as
Rate = k [A]a[B]b
LAW OF MASS ACTION](https://image.slidesharecdn.com/chemicalequilibruimlecture28jan-230424084244-d95602c5/85/Chemical-Equilibruim-lecture-2-8-Jan-pdf-27-320.jpg)

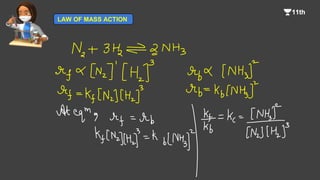
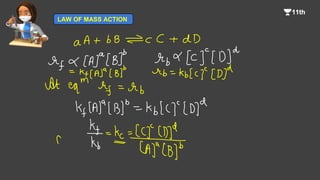
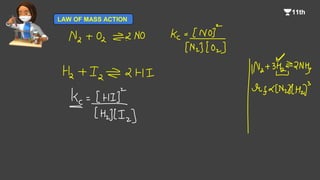






![By applying law of mass action to a reversible reaction, at equilibrium, it is
possible to derive a simple mathematical expression known as law of
chemical equilibrium.
Let us consider a simple reversible reaction,
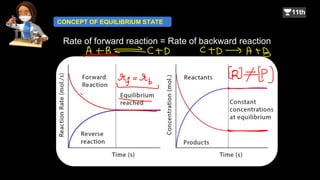
A + B C + D
Rate of forward reaction [A][B]
= kf [A][B]
where kf is the rate constant for the forward reaction.
Similarly,
Rate of backward reaction [C][D]
= kb[C][D]
where kb is the rate constant for the backward.
LAW OF MASS ACTION: Application](https://image.slidesharecdn.com/chemicalequilibruimlecture28jan-230424084244-d95602c5/85/Chemical-Equilibruim-lecture-2-8-Jan-pdf-38-320.jpg)
![Rate of forward reaction = Rate of backward reaction.
kf[A][B] = kb[C][D]
or
Since kf and kb are constants, therefore, the ratio kf/kb is also constant and
is represented by Kc.
f
b
k C D
k A B
f
c
b
k C D
K
k A B
LAW OF MASS ACTION: Application](https://image.slidesharecdn.com/chemicalequilibruimlecture28jan-230424084244-d95602c5/85/Chemical-Equilibruim-lecture-2-8-Jan-pdf-39-320.jpg)






![2. What is Kc for the following equilibrium, when the equilibrium
concentration of each substance is
[SO2] = 0.60 M, (O2) = 0.82 M and (SO3] = 1.90 M
2SO2(g) + O2(g) ⇌ 2SO3(g)
Lets Practice](https://image.slidesharecdn.com/chemicalequilibruimlecture28jan-230424084244-d95602c5/85/Chemical-Equilibruim-lecture-2-8-Jan-pdf-46-320.jpg)