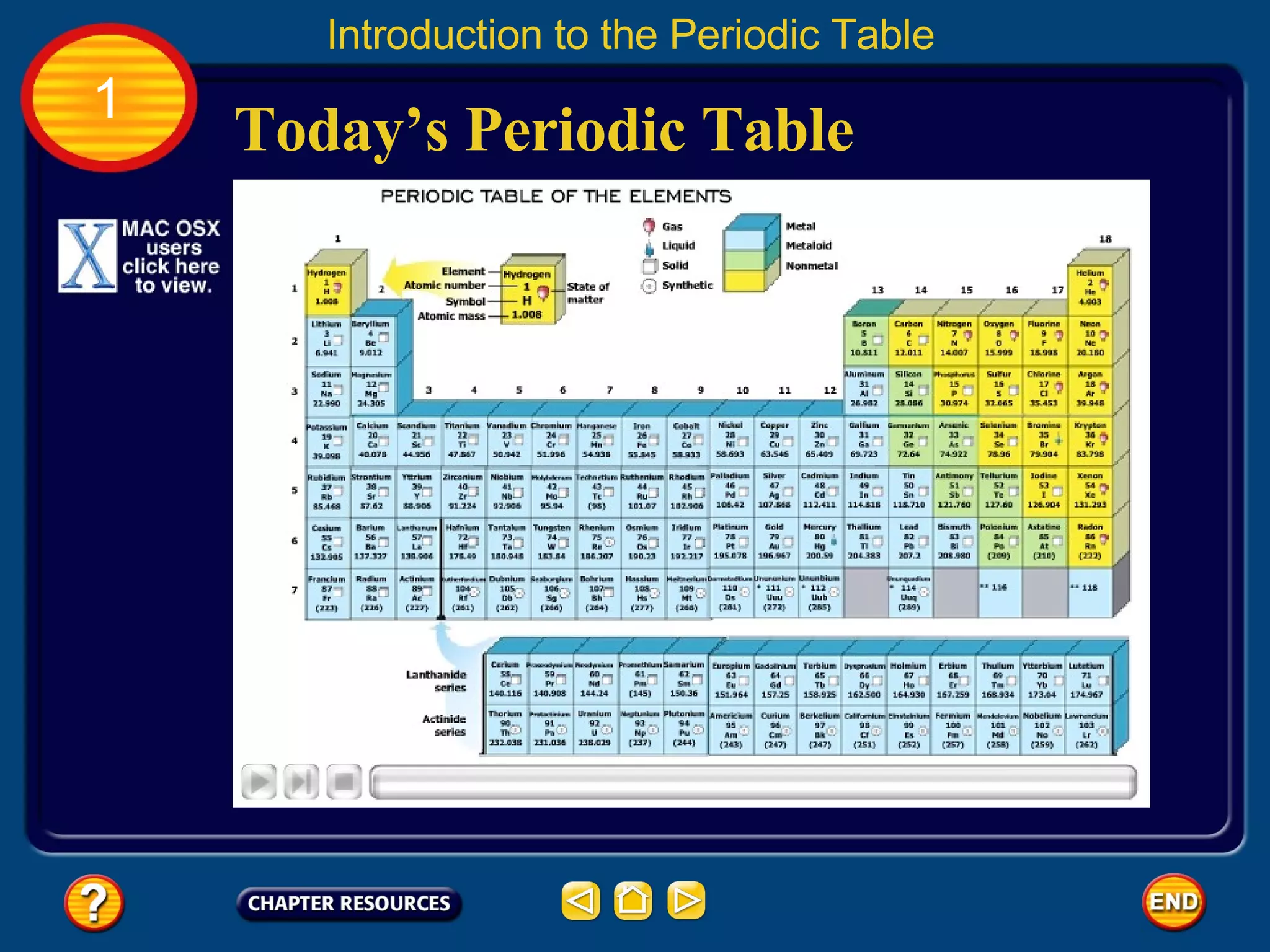
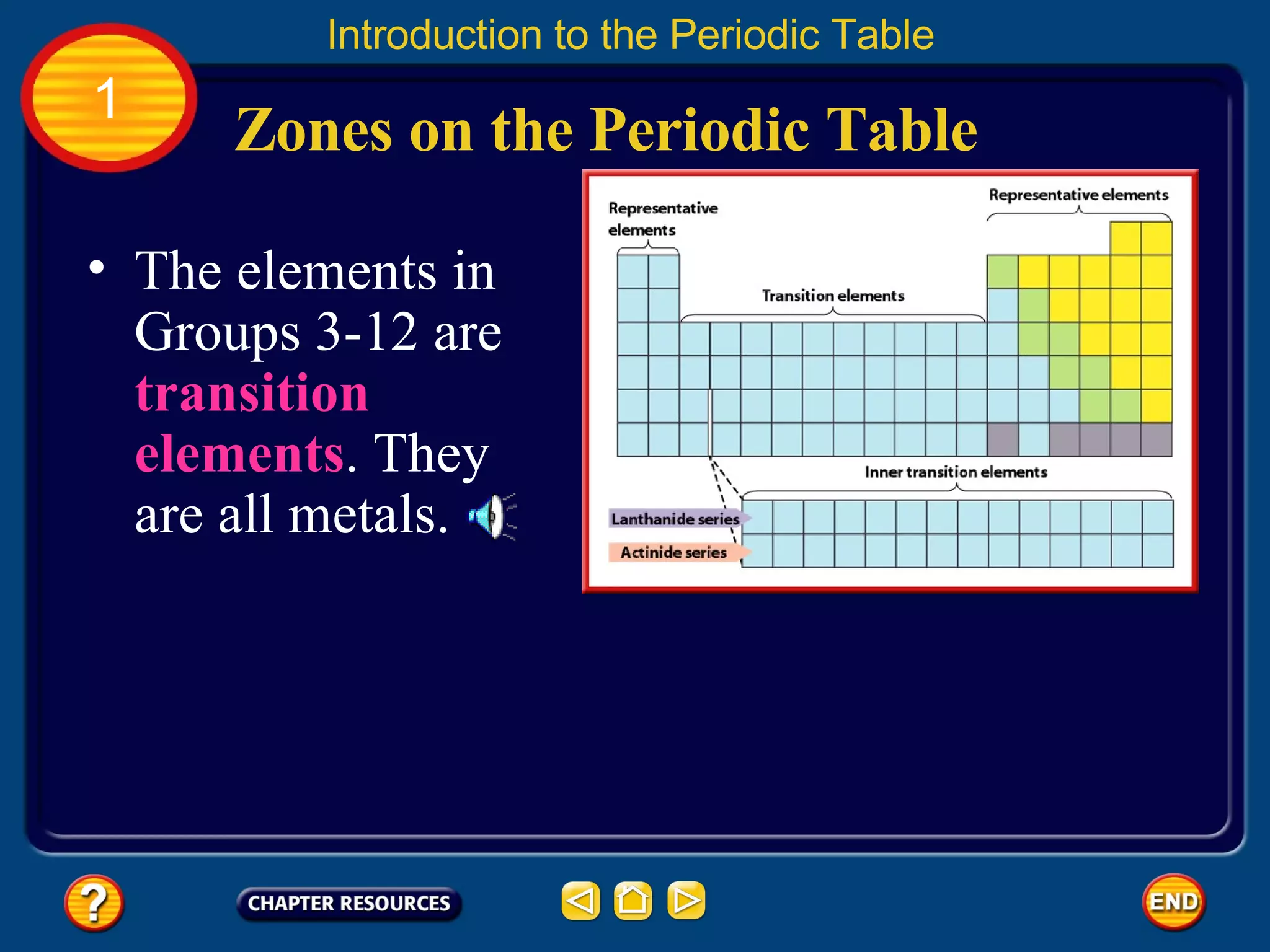
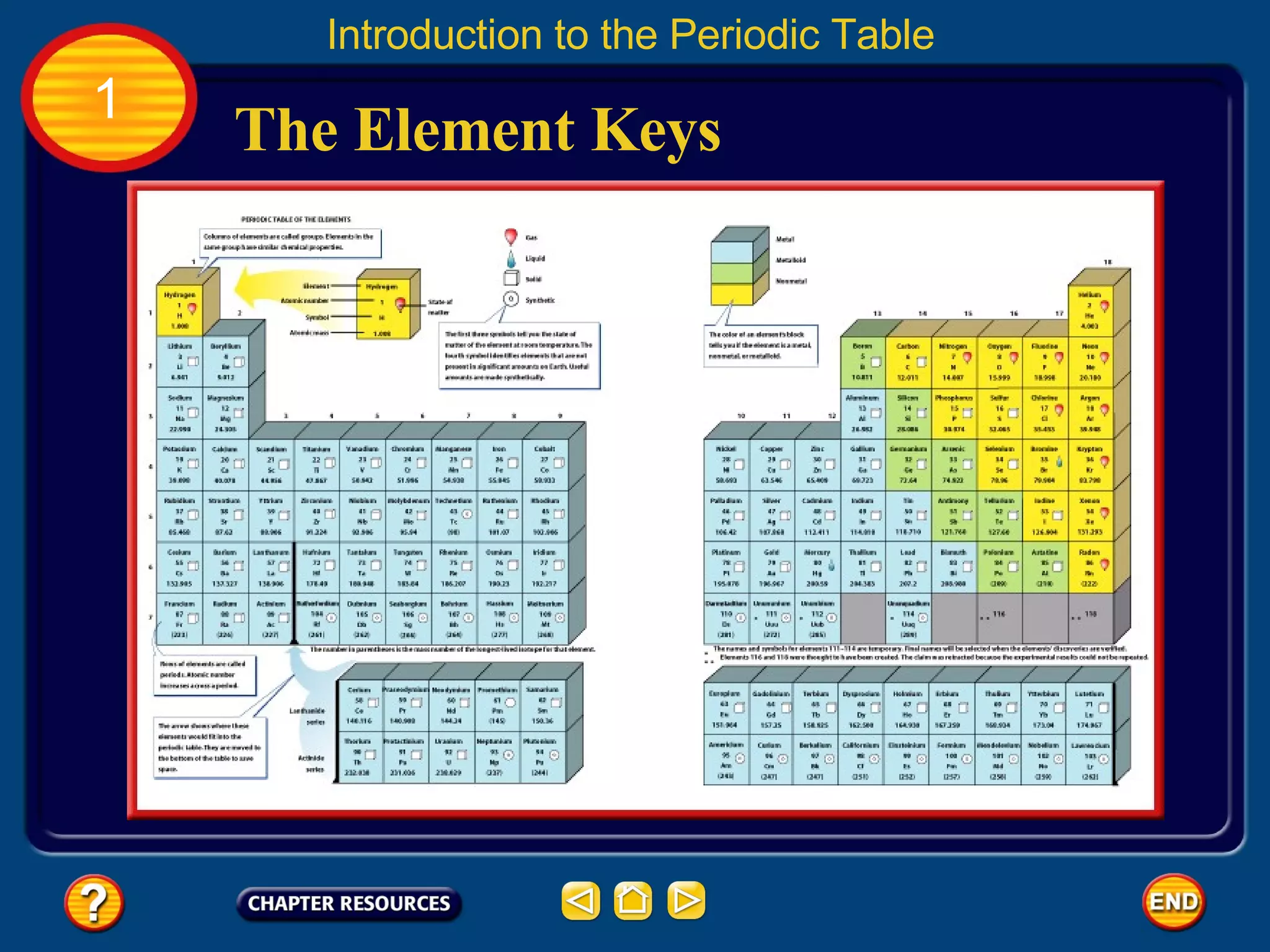
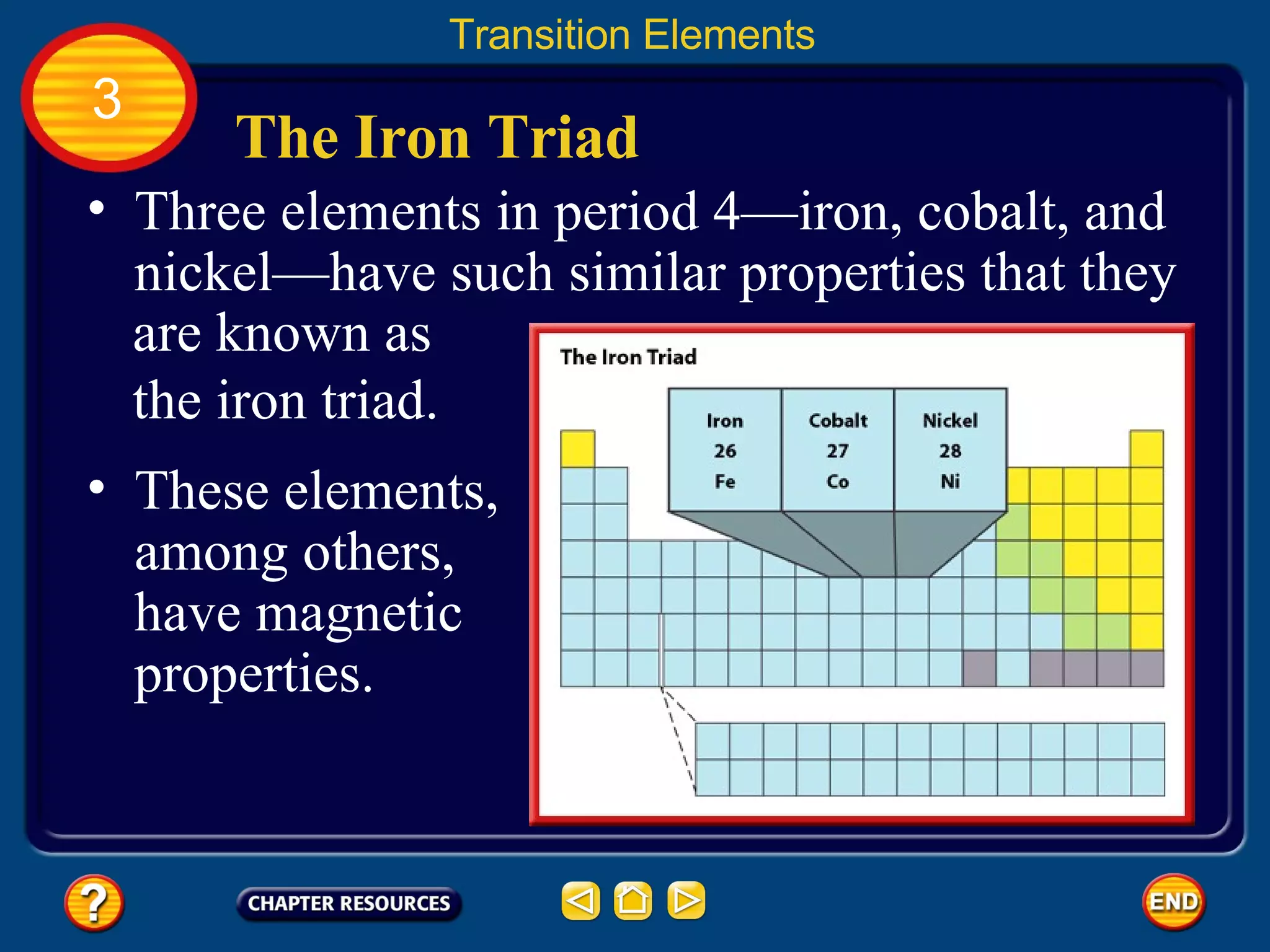
The document discusses the periodic table and its development. It describes how Dmitri Mendeleev created one of the first periodic tables in 1869 and arranged elements based on atomic mass, predicting properties of unknown elements. Later, Henry Moseley improved the table by arranging elements by atomic number. The modern periodic table still organizes elements by increasing atomic number and groups elements with similar properties together.