Science 8 Quarter 3 WEEK7 Periodic Table of Elements.pptx
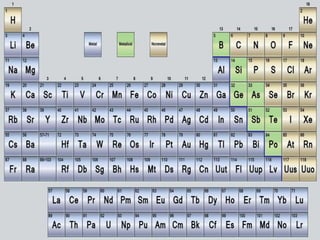
- 4. Elements • Each element on the Periodic Table has an atomic number, symbol, name and average atomic mass. • Atomic number – the number of protons found in the nucleus of an atom. • Average atomic mass – the mass of an atom including the protons, neutrons and electrons.
- 5. Dmitri Ivanovich Mendeleev - Initially made it easy for us to predict the physical and chemical behavior of an element when he published his version of the periodic table in 1869 - He meticulously arranged the elements based on increasing atomic weight and had it in a readable format. There was a predictive power in his table - based on the periodic law, Mendeleev believed that more elements would be discovered someday. He left spaces in his table where the elements would be placed once they had been discovered.
- 6. Figure 1. Mendeleev’s Periodic Table
- 7. In 1913, it was revised by Henry Gwyn Jeffreys Moseley, an English physicist who discovered that atomic number is the most fundamental property of an element and not its atomic mass (Figure 2).
- 9. How is it arranged? The red lines show the different periods and the green lines show the groups.
- 10. Metals, Nonmetals and Metalloids • The major categories of elements are the metals, non-metals, and metalloids. • Metals are lustrous, malleable, and are good conductors of heat and electricity. • Non-metals are elements that do not share the properties of metals. • Metalloids are elements that share some, but not all the properties of metals.
- 11. Metals - Lustrous, Malleable, and Good Conductors? These are physical properties of metals • Lustrous means shiny or reflective of light. – Coins and jewelry are shiny and reflective . • Malleable means capable of being shaped. – Aluminum foil is shaped or molded around food items to keep them fresh.
- 12. • Being a Good Conductor means being able to allow electricity and heat to flow through. −When you think about the wires we use for electrical devices, they are mostly made of copper and other metals.
- 13. Group of Elements and their Special Names
- 14. Alkali Metals • Elements in Group 1 (not including Hydrogen). • Very reactive metals. Always combine with something else in nature. • Salt – an Alkali Metal, Sodium, and another element, Chlorine, combined.
- 15. Alkaline Earth Metals • Elements in Group 2. • Reactive Metals that are always combined with non-metals in nature. • Several of these elements are important mineral nutrients, like Calcium.
- 16. Transition Metals • Elements in Groups 3-12. • Less reactive, harder metals. • Includes metals used in jewelry, money and construction.
- 17. Boron Family • Elements in Group 13. • Boron has properties of both metals and non-metals. • The rest of the elements in this group are metals.
- 18. Carbon Family • Elements in Group 14. • Contains elements important to life and computers. • Carbon is the basic element in all organic compounds. • Silicon and Germanium are important semiconductors.
- 19. Nitrogen Family • Elements in Group 15. • Nitrogen makes up more than ¾ of our atmosphere. • The red tip of matches is made of phosphorous.
- 20. Oxygen Family or Chalcogens • Elements in Group 16. • Oxygen is necessary for respiration. • Many things that have a bad odor contain sulfur.
- 21. Halogens • Elements in Group 17. • These elements are known to be salt-forming. • Always found combined with other elements in nature. • Chlorine is used to keep bacteria out of swimming pools.
- 22. Noble Gases • Elements in Group 18. • VERY reactive gases. • Used in lighted neon signs. • Helium is used to make party balloons float.
- 23. WHO’S WHO? Choose from the box below. Dmitri Ivanovich Mendeleev Lothar Meyer John Newlands Johann Dobereiner
- 24. Let’s do this! Activity 1: My Location! Direction: Using your periodic table, identify the element that is specified in each of the items. 1. The element in period 5, group 10 2. The element in period 4, group 15 3. The element in period 3, group 2 4. The element in period 5, group 18 5. The element in period 4, group 13 Palladium ( Pd ) Arsenic ( As ) Magnesium ( Mg ) Xenon ( Xe ) Gallium ( Ga )
- 25. Directions: The elements below and their food sources are essential nutrition our body needs. Identify each of these element’s period number (1), group number (2), and group name (3). Activity 7: I Got You! 1.Period number: _____ 2.Group number: _____ 3.Group name: __________
- 26. 1.Period number: _____ 2.Group number: _____ 3.Group name: __________
- 27. 1.Period number: _____ 2.Group number: _____ 3.Group name: __________
- 29. 1.Period number: _____ 2.Group number: _____ 3.Group name: __________ 4 IIA or 2A ALKALINE EARTH METALS
- 30. 1.Period number: _____ 2.Group number: _____ 3.Group name: __________ 4 1 ALKALI METAL
- 31. 1.Period number: _____ 2.Group number: _____ 3.Group name: __________ 4 12 TRANSITION METALSLKK’W