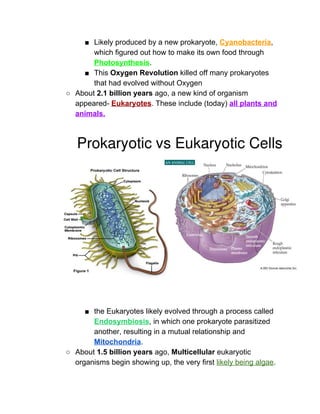
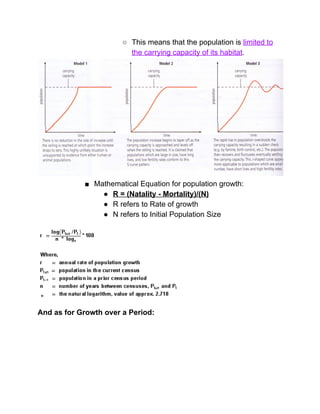
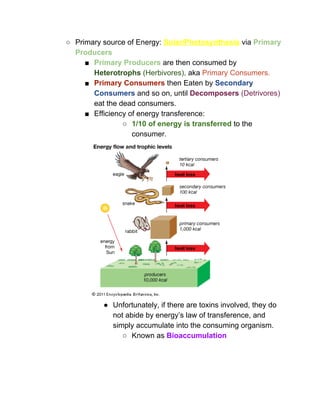
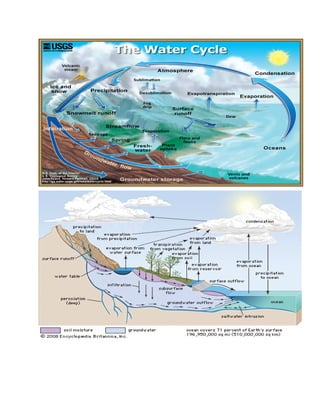
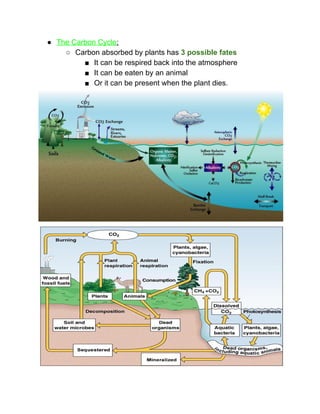
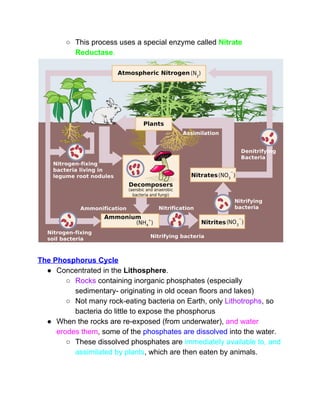
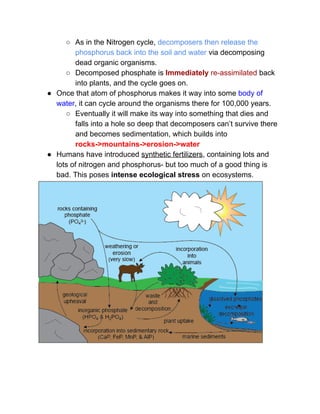
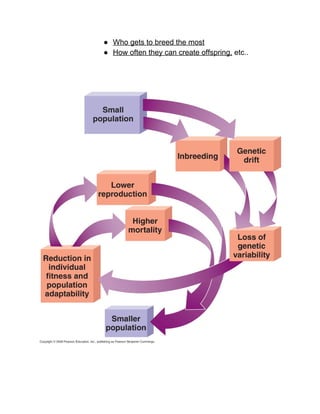
The document outlines the history of life on Earth, from the emergence of prokaryotes around 3.9 billion years ago to the evolution of complex ecosystems and the patterns of population dynamics. It discusses significant events, such as the oxygen revolution, the Cambrian explosion, and mass extinctions, while introducing basic concepts in ecology, including population and community interactions. Additionally, it contrasts human population growth with ecological theories, emphasizing the balance between quality and quantity in reproductive strategies.