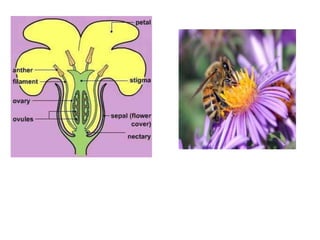
This document provides information on various carbohydrate containing drugs including honey, acacia, agar, and tragacanth. It discusses the biological sources, geographical sources, methods of collection and preparation, chemical constituents, identification tests, standards and uses of these drugs. The key drugs discussed are honey obtained from bees, acacia gum obtained from the acacia tree, and agar obtained from red algae. It provides details on the morphology, chemistry and applications of these carbohydrate drugs.