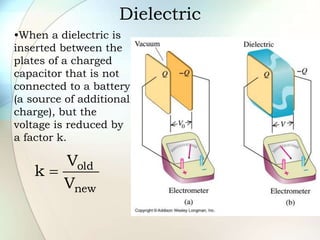
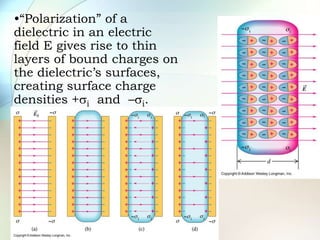
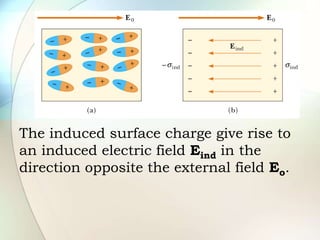
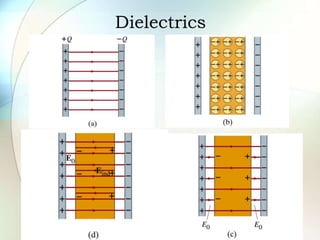
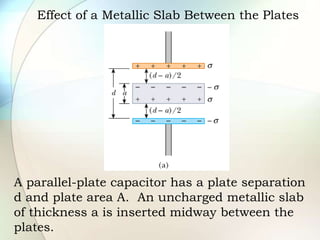
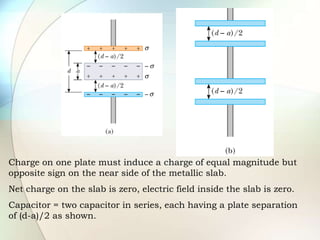
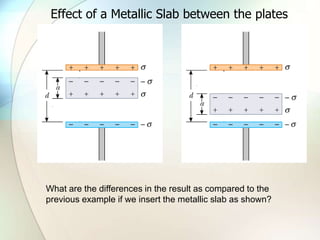
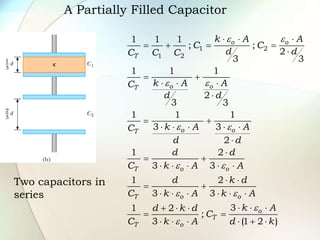
A dielectric is a nonconducting material inserted between the plates of a capacitor that increases the capacitor's ability to store an electric charge. When a dielectric is used, it increases the capacitance by a factor called the dielectric constant. The dielectric reduces the electric field strength inside itself and increases the breakdown voltage of the capacitor. Different dielectric materials have different dielectric constants depending on how easily their molecules can be polarized in an external electric field.