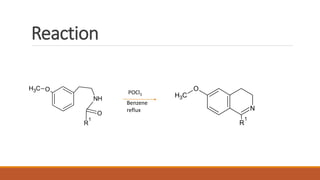
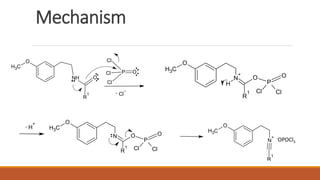
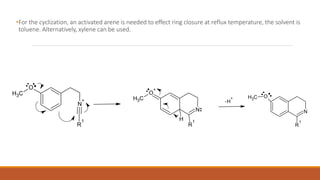
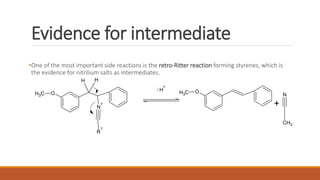
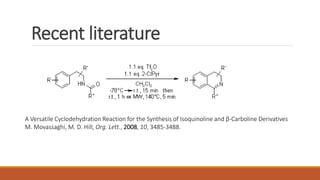
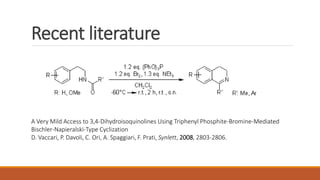
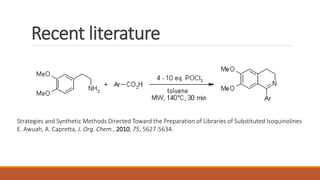
The document summarizes the Bischler-Napieralski reaction, which involves the cyclization of β-arylethylamides or β-arylethylcarbametes to form dihydroisoquinolines under acidic and dehydrating conditions. The reaction proceeds through a nitrilium ion intermediate and can be used to synthesize alkaloids containing an isoquinoline skeleton. It was first discovered in 1893 and the mechanism involves an initial dehydration followed by an intramolecular electrophilic aromatic substitution. Recent literature has explored modifying conditions and substrates to generate substituted isoquinolines and β-carbolines through this cyclodehydration reaction.