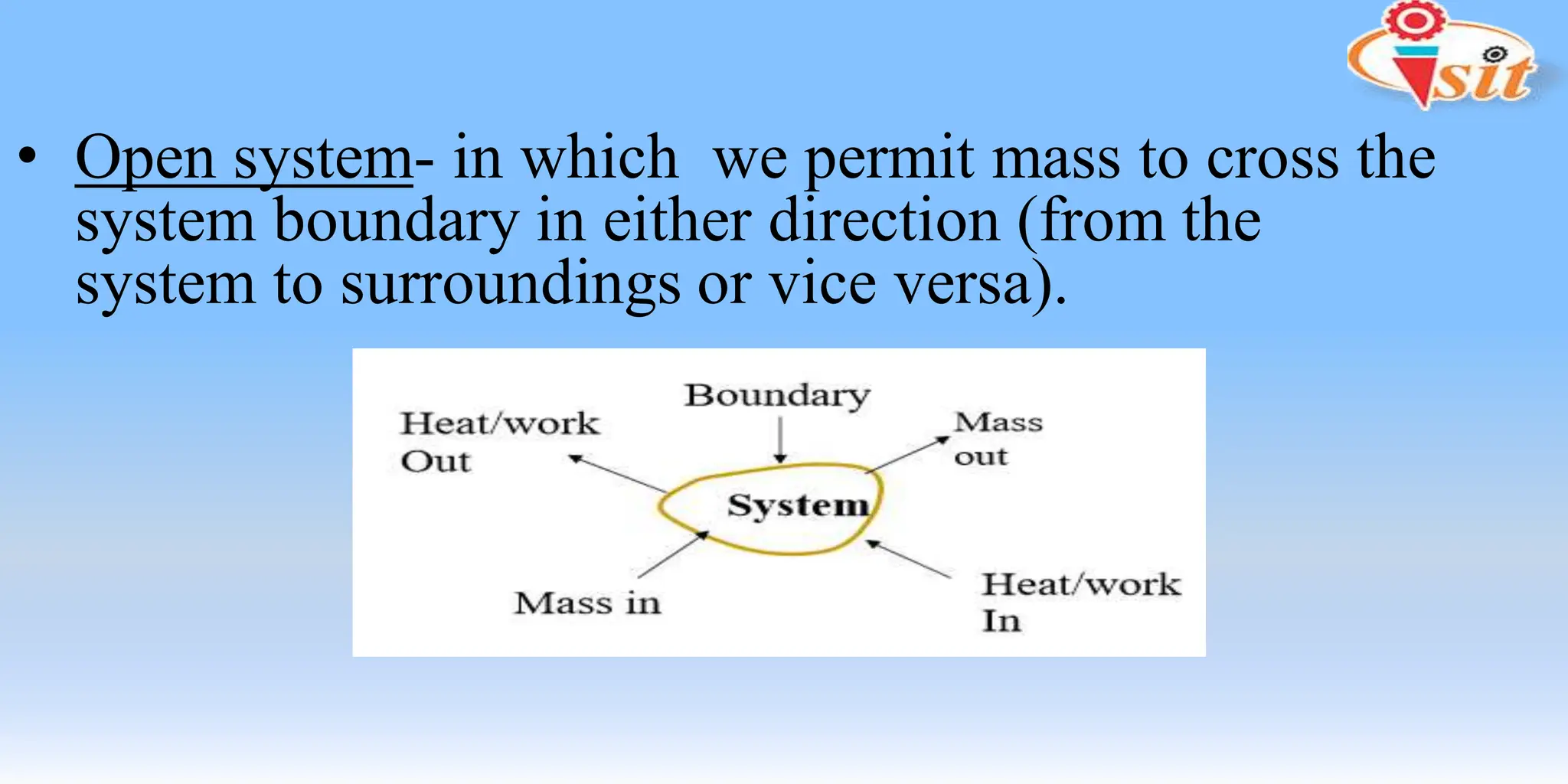
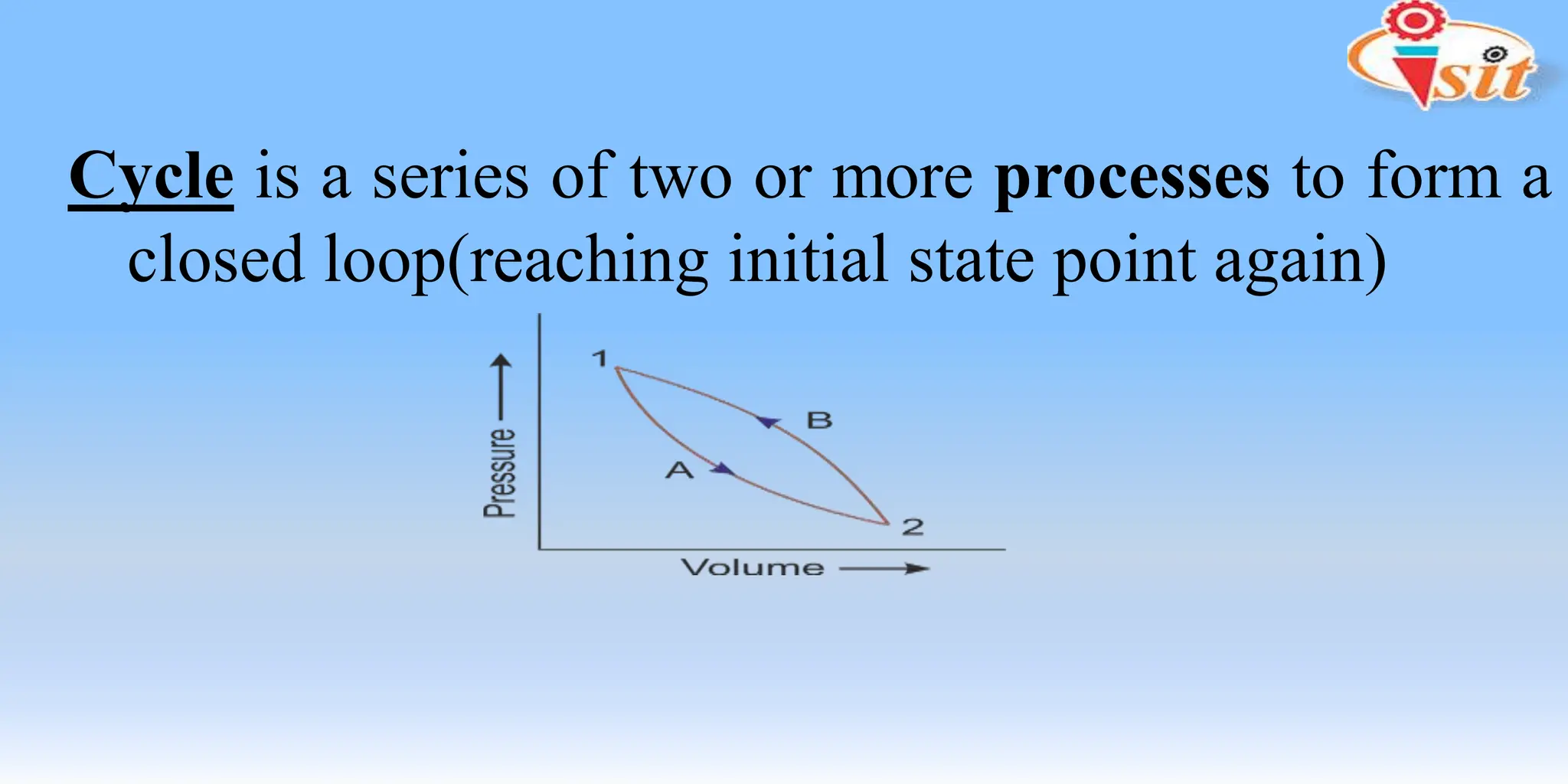
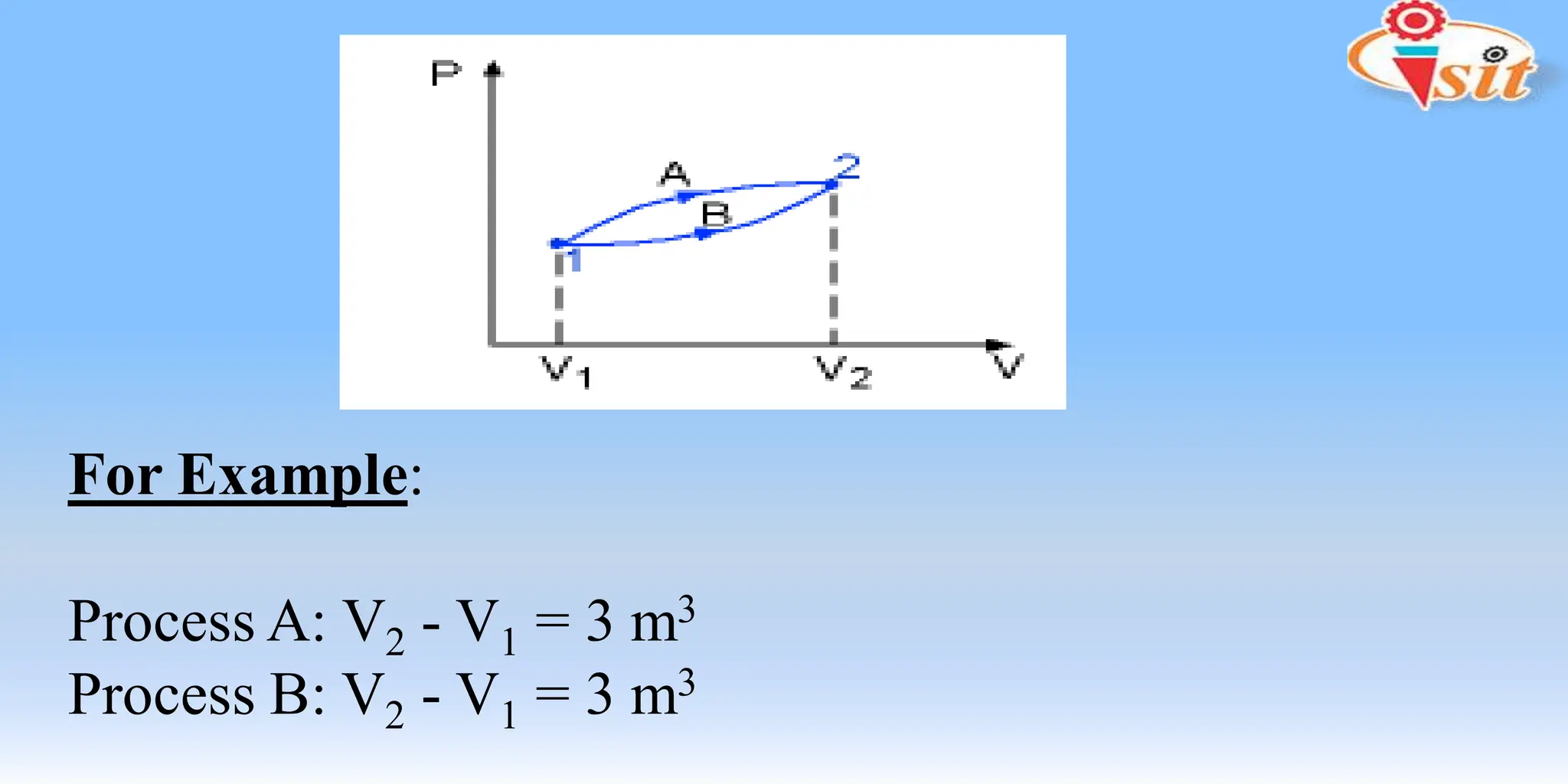
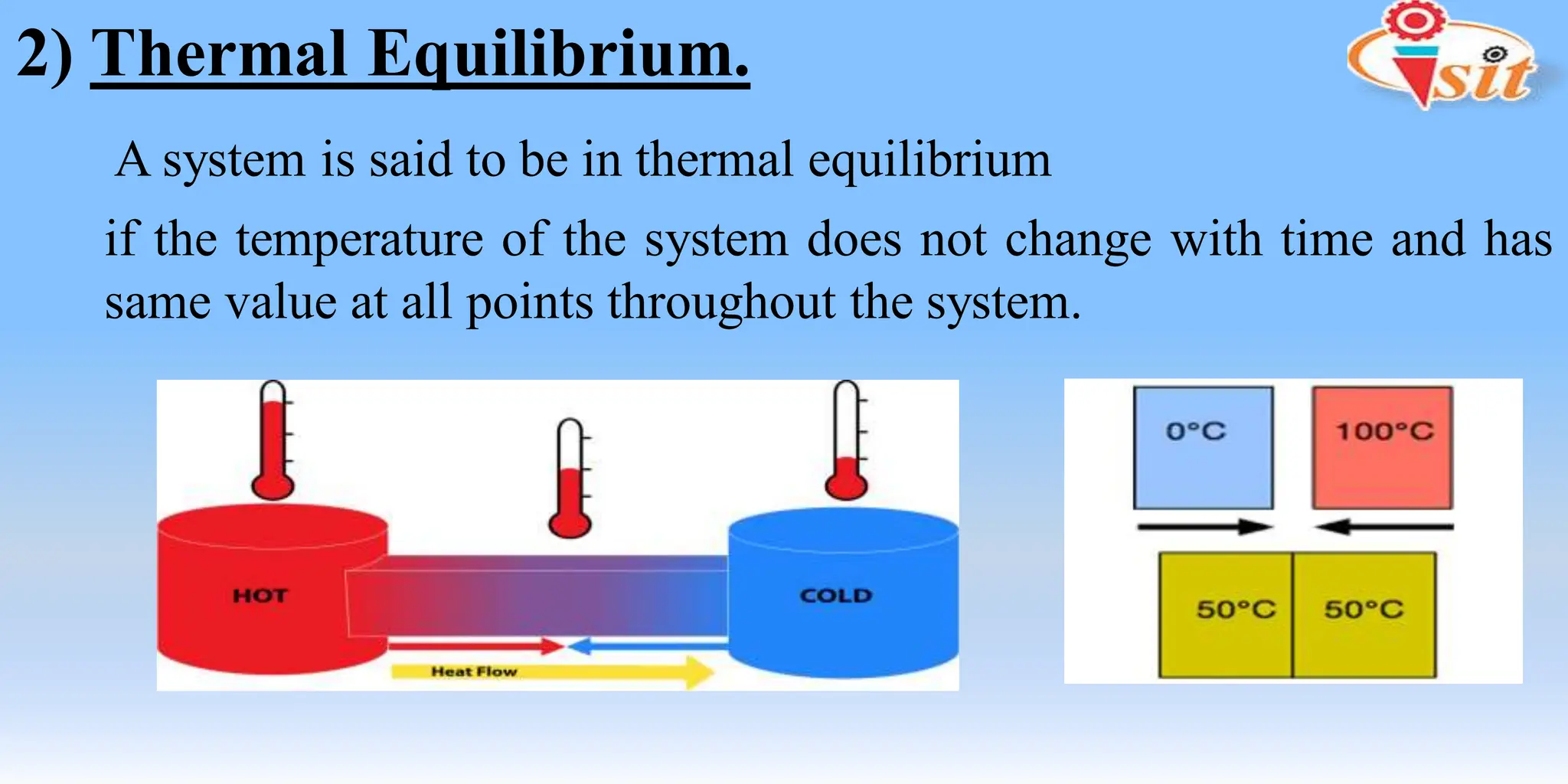
The document covers fundamental concepts of thermodynamics and heat transfer, including definitions of systems, properties, and processes. It classifies systems into closed, open, and isolated types, and introduces key terms such as specific volume, density, temperature, and pressure. The document also distinguishes between path functions and point functions, detailing concepts of thermodynamic equilibrium.