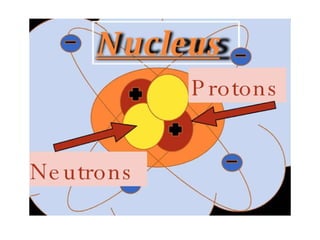
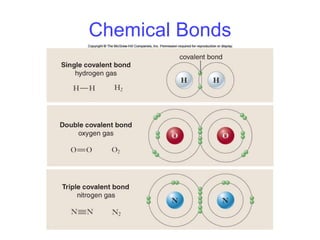
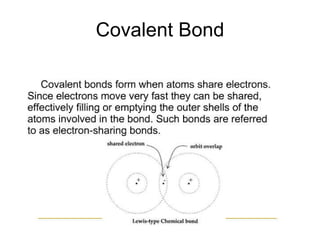
This document summarizes key concepts about atoms, molecules, and chemical bonds. It defines atoms as the smallest unit of an element, consisting of a nucleus with protons and neutrons surrounded by electrons. Molecules are formed when two or more atoms bond together, such as through covalent bonds where atoms share electrons. The document also outlines Dalton's atomic theory and the laws of chemical combination, including the law of conservation of mass and the law of constant proportions.