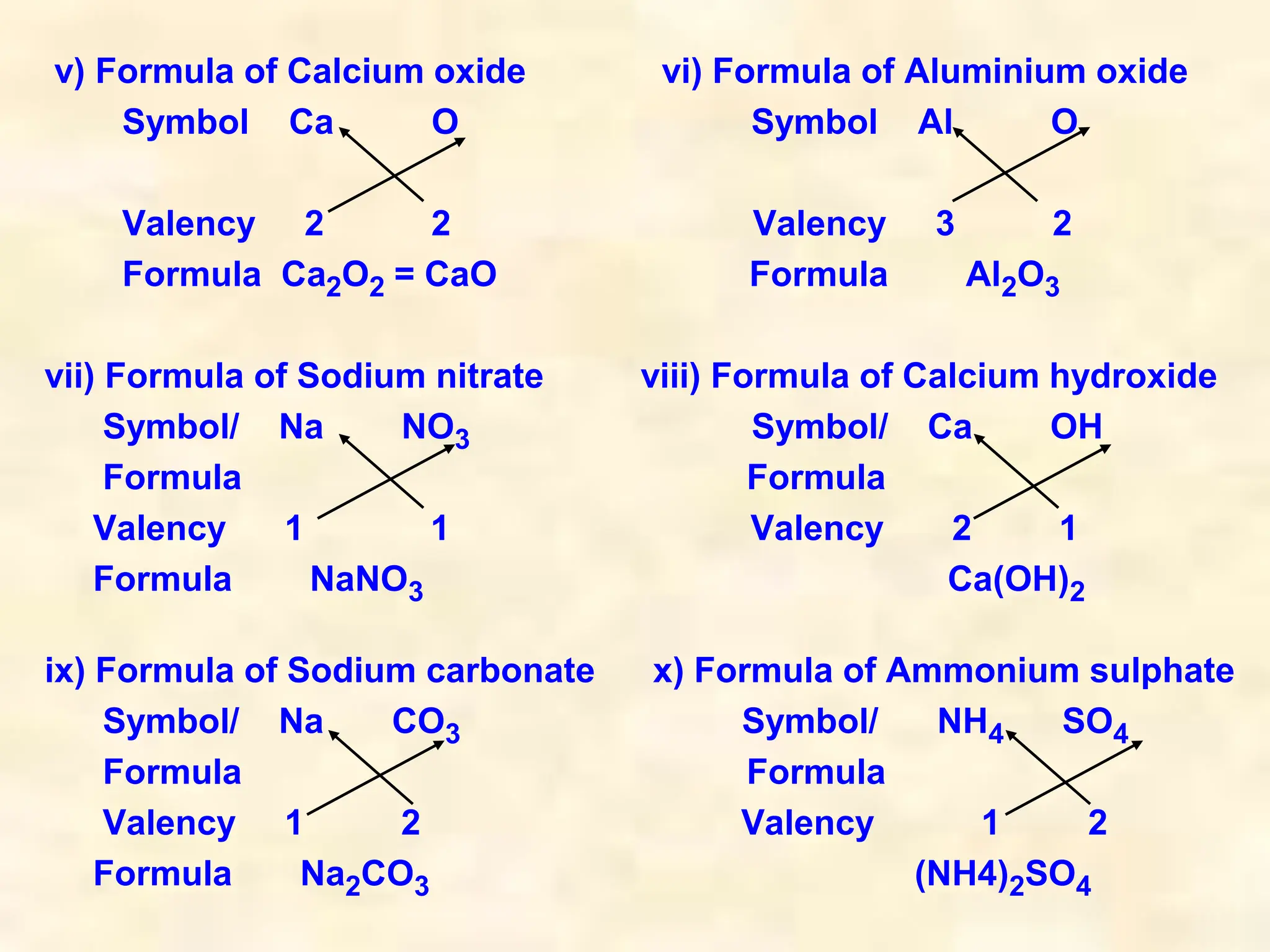
This document provides information about atoms and molecules:
1) It summarizes Dalton's atomic theory, including that atoms are indivisible and combine in small whole number ratios to form compounds.
2) It describes what atoms and molecules are, noting atoms are the smallest particle of an element and molecules are groups of atoms held together.
3) Key concepts like atomic mass, molecular mass, ions, and the mole concept are explained. The mole represents 6.022 x 1023 particles and relates mass, moles, and molar mass.