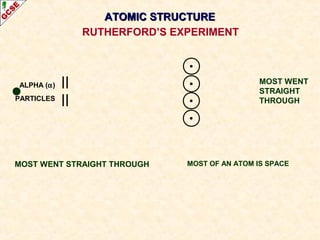
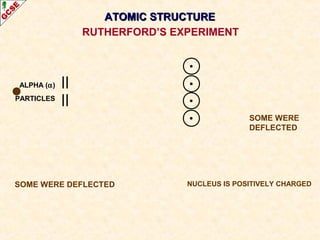
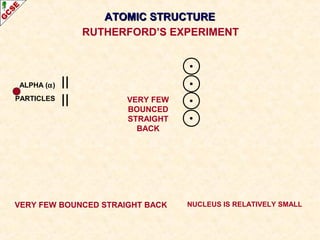
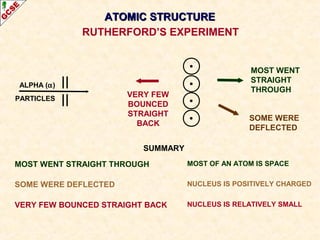
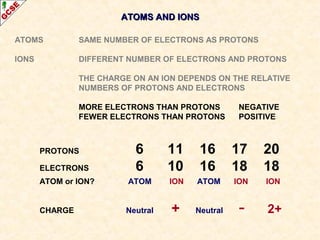
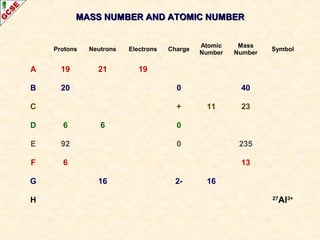
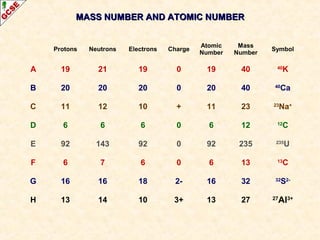
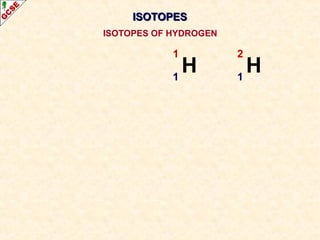
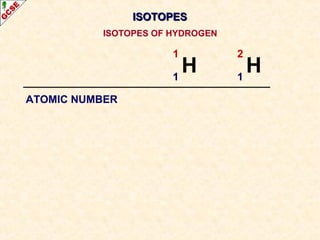
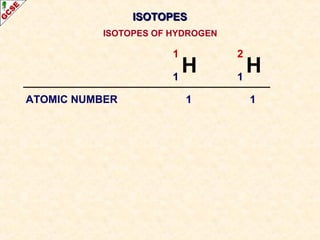
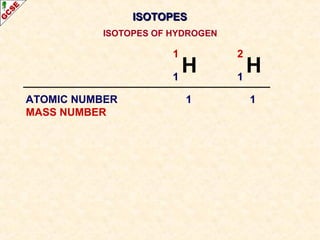
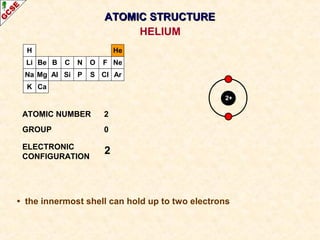
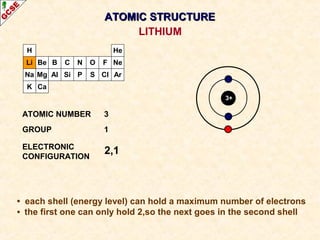
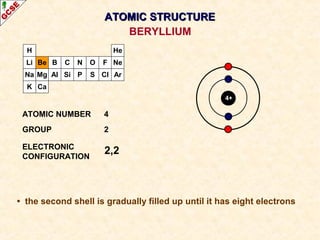
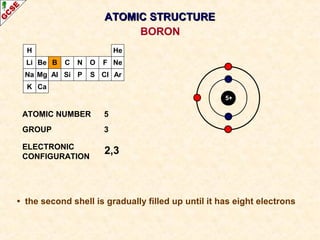
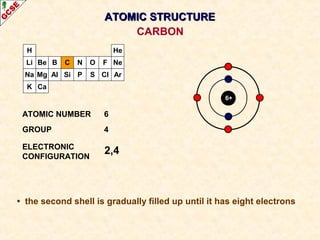
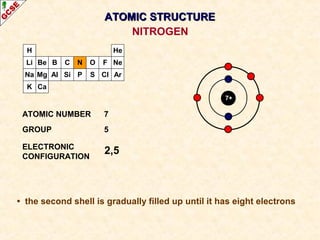
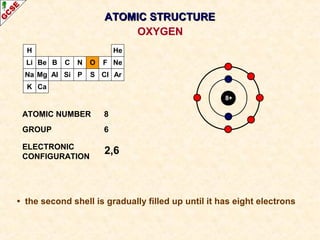
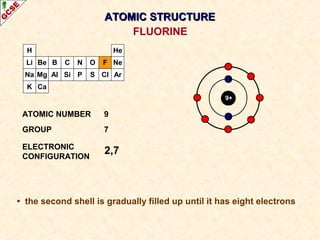
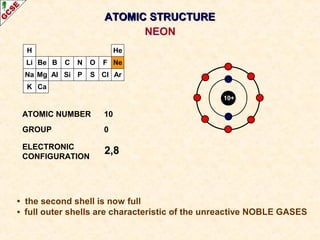
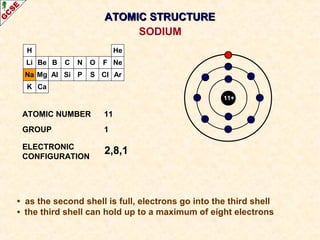
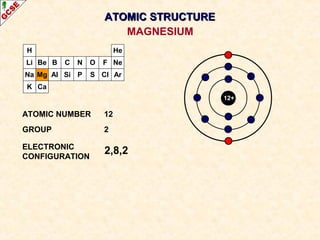
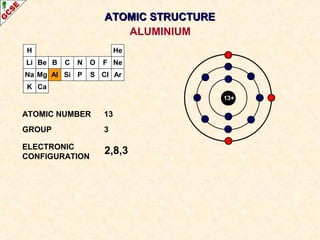
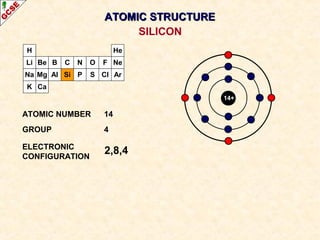
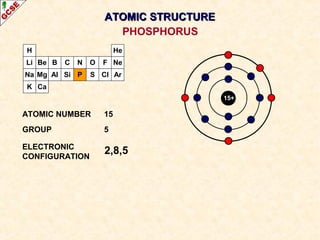
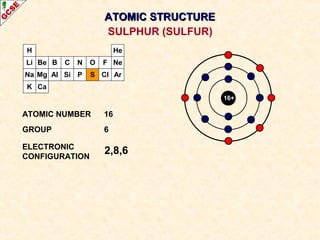
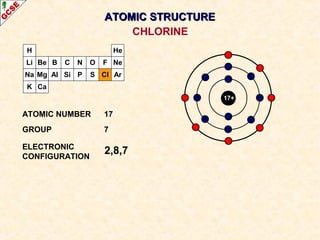
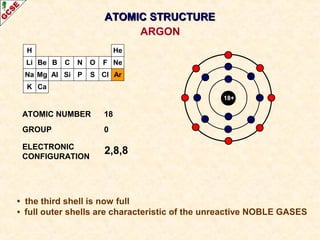
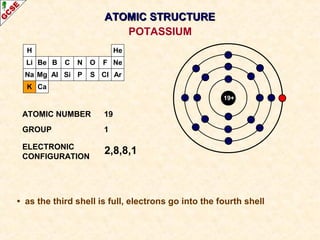
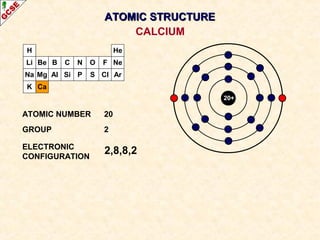
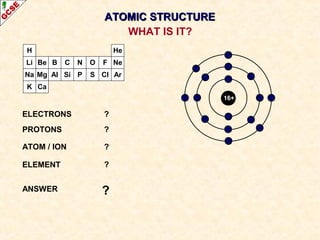
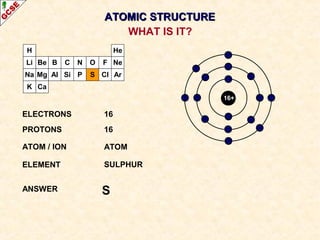
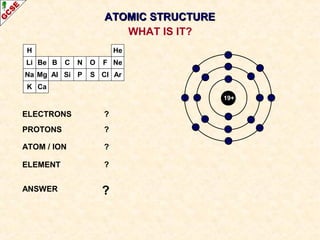
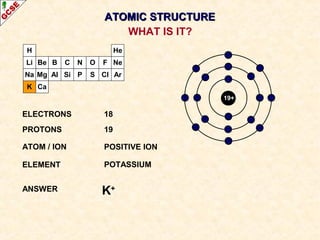
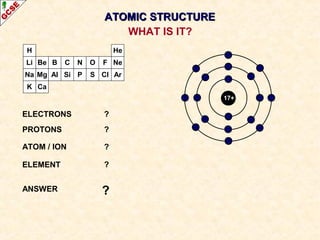
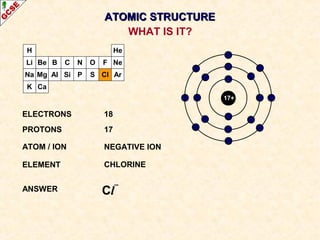
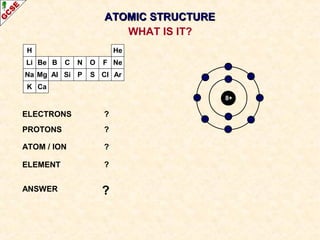
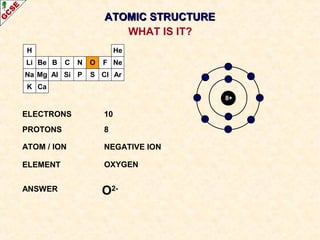
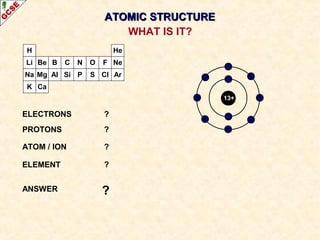
Rutherford's experiment involved firing alpha particles at a thin gold foil. Most passed straight through, showing the atom is mostly empty space. Some were deflected, showing a small, dense central nucleus. Very few bounced straight back, showing the nucleus is very small compared to the atom. This led to the discovery of the nuclear model of the atom, with a small, dense nucleus surrounded by electrons.