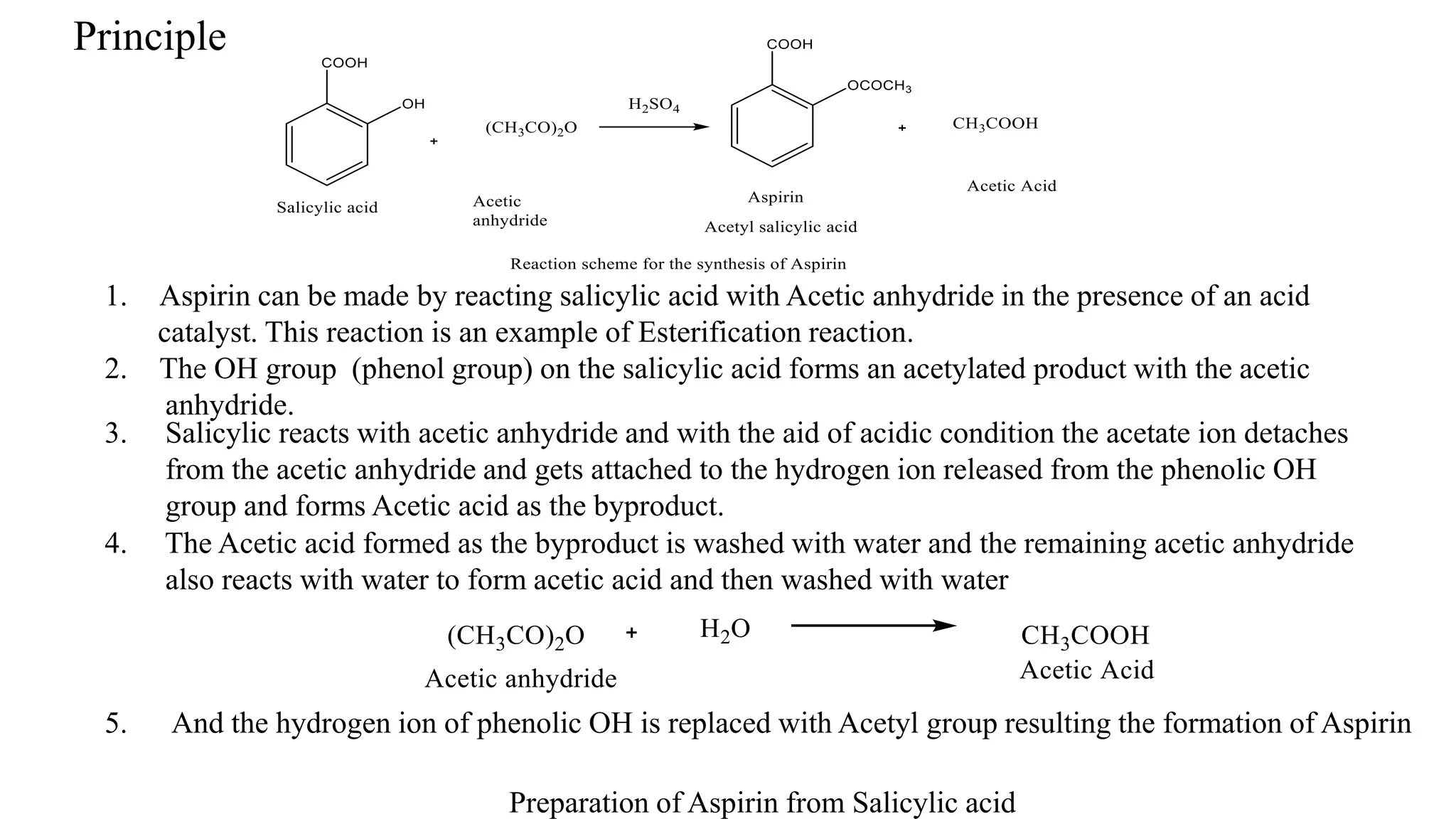
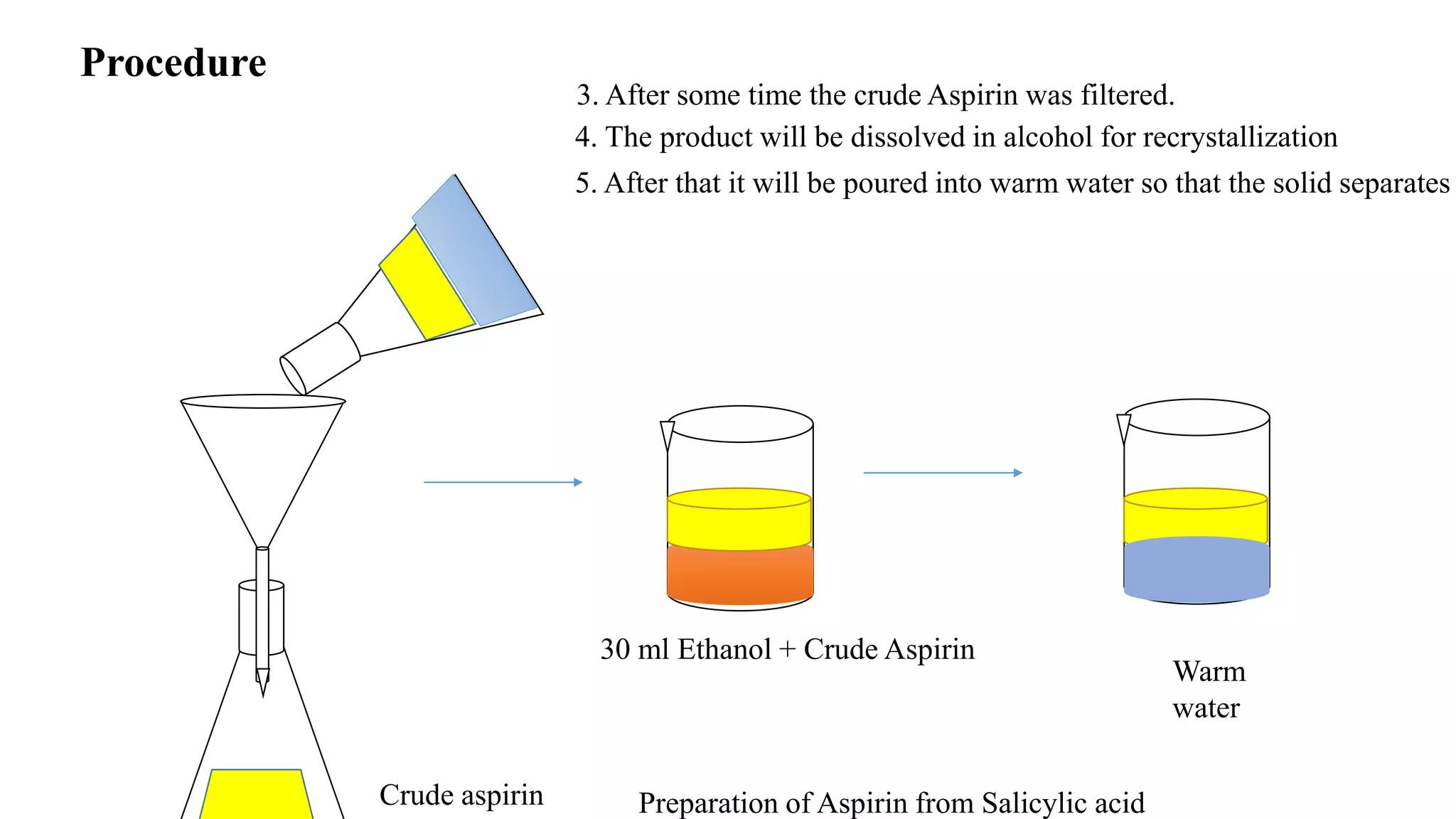
This document describes the preparation of aspirin from salicylic acid. Aspirin is synthesized through an esterification reaction, where salicylic acid reacts with acetic anhydride in the presence of sulfuric acid as a catalyst. This replaces the phenolic hydroxyl group of salicylic acid with an acetyl group, forming aspirin and acetic acid as a byproduct. Specifically, 10g of salicylic acid is reacted with 14mL of acetic anhydride and 2mL of sulfuric acid by heating on a water bath for 30 minutes. The crude aspirin product is then purified by recrystallization in ethanol. The percentage yield of aspirin is then calculated based on the theoretical