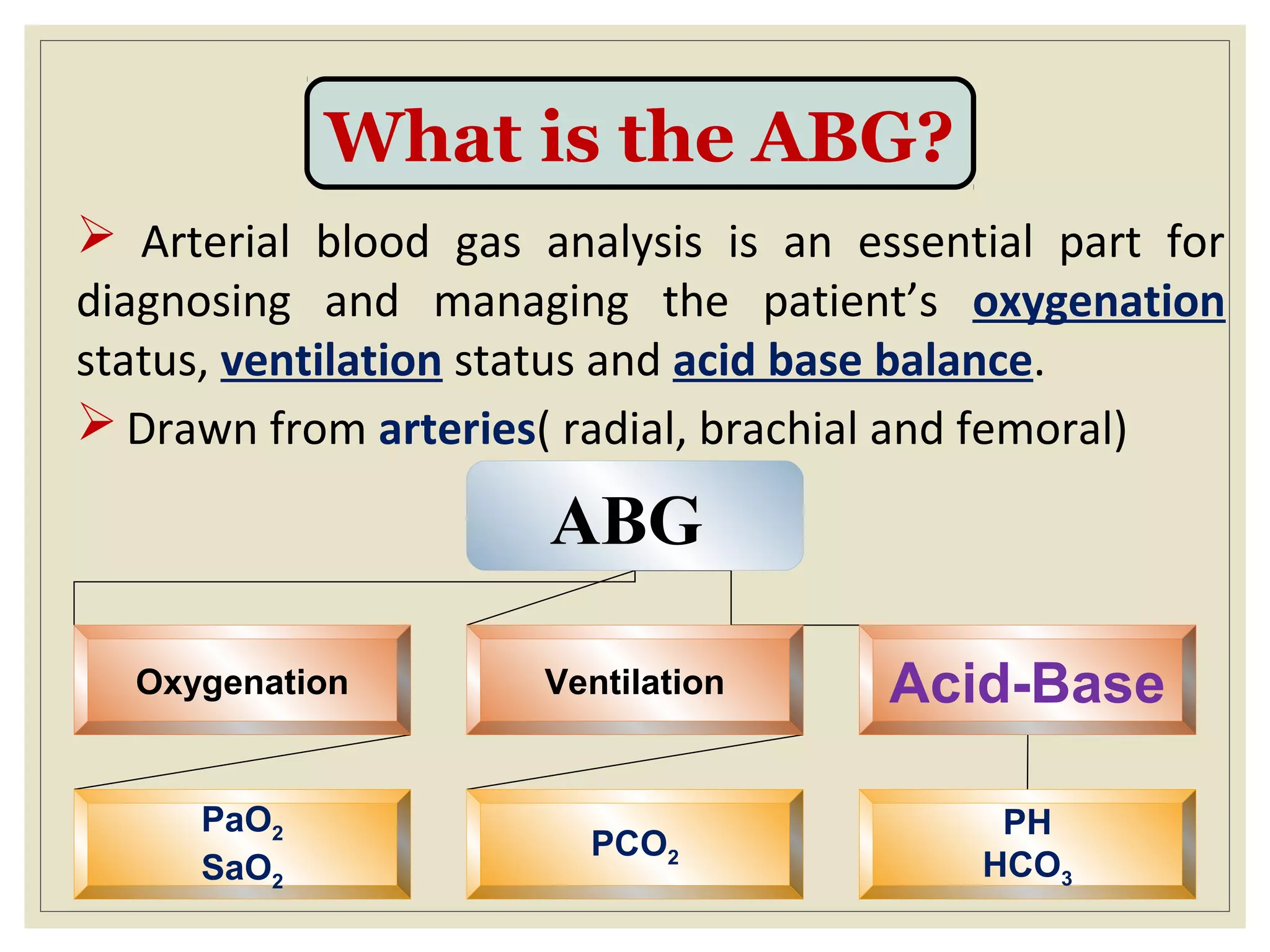
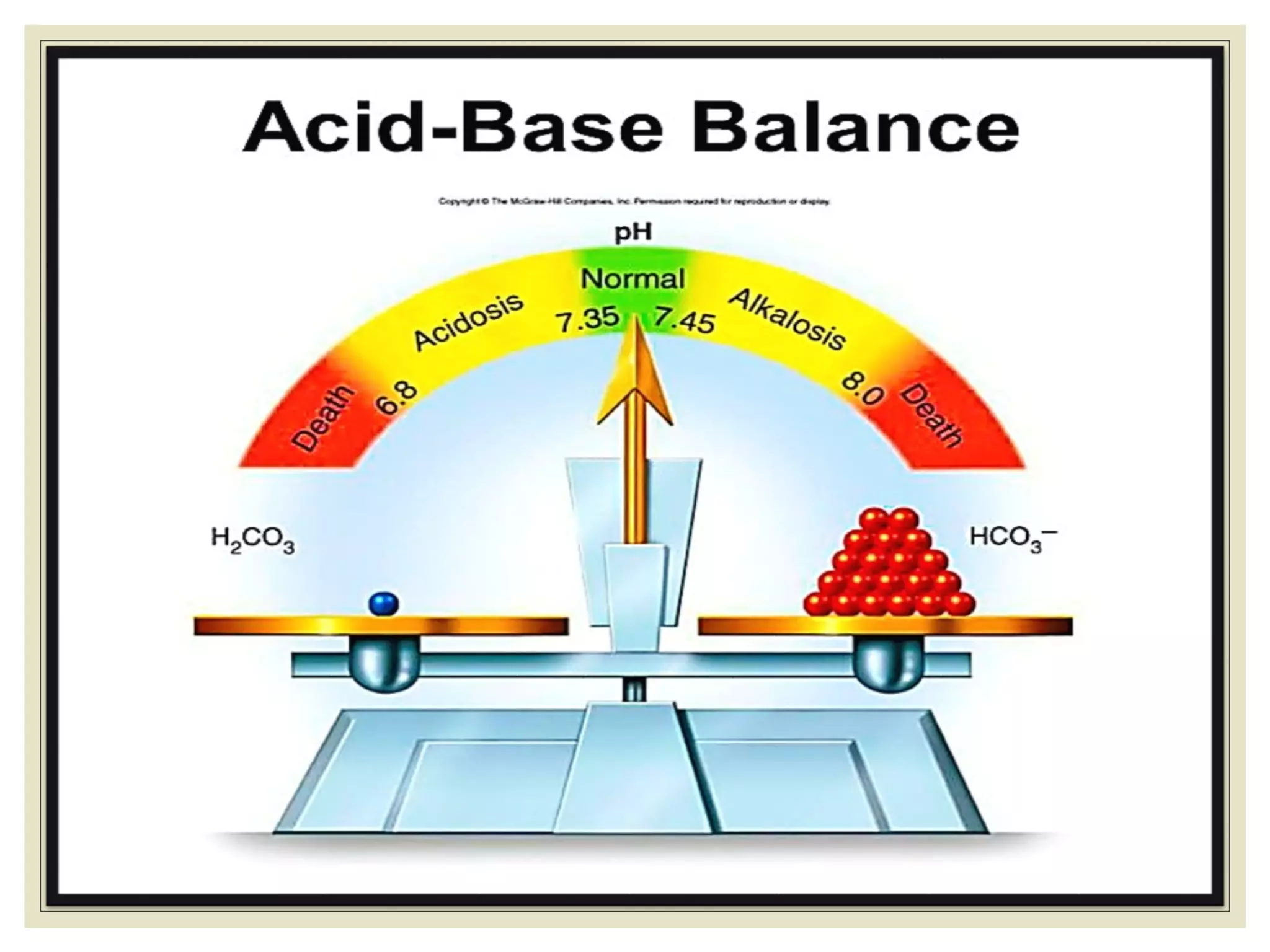
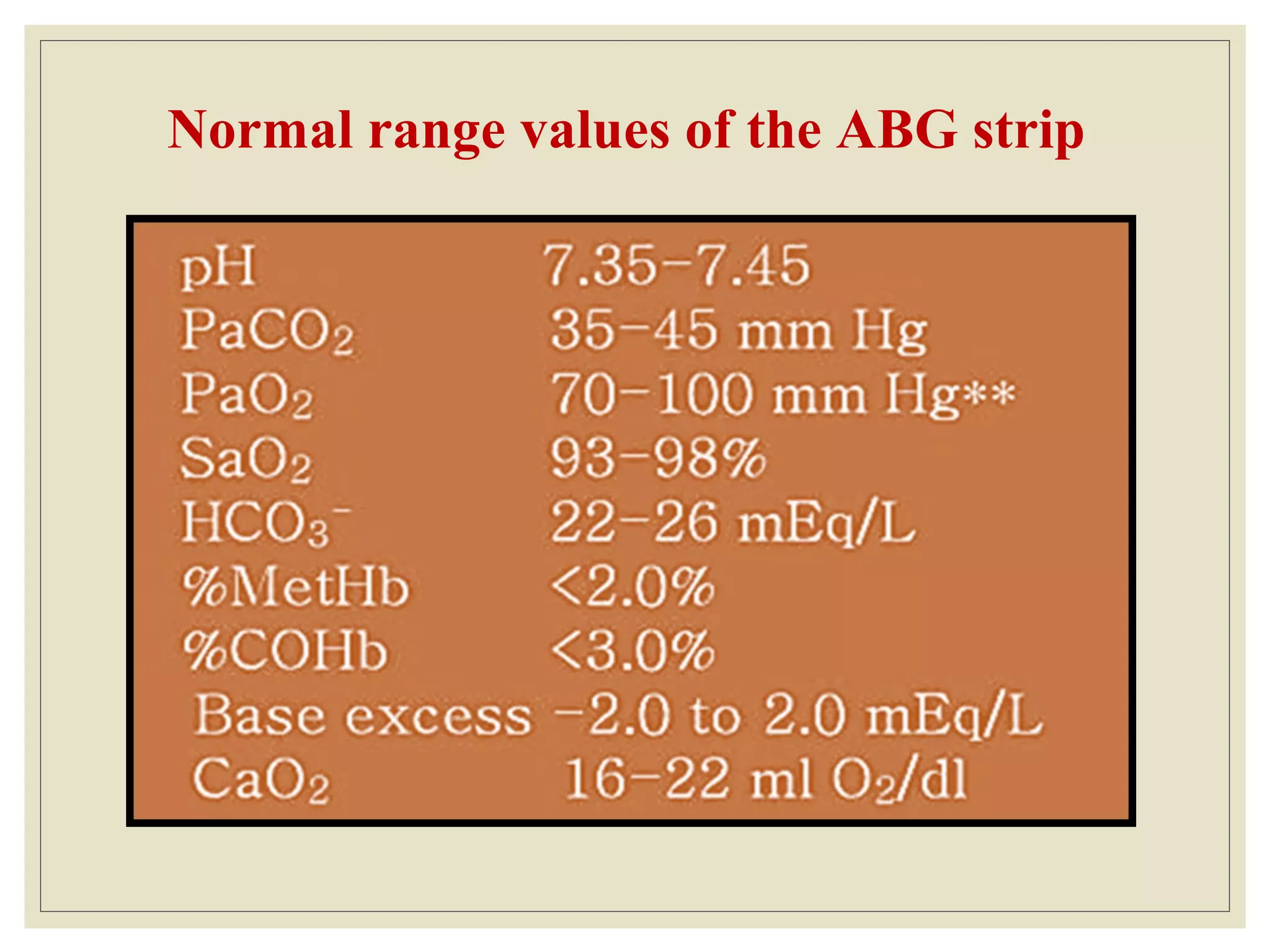
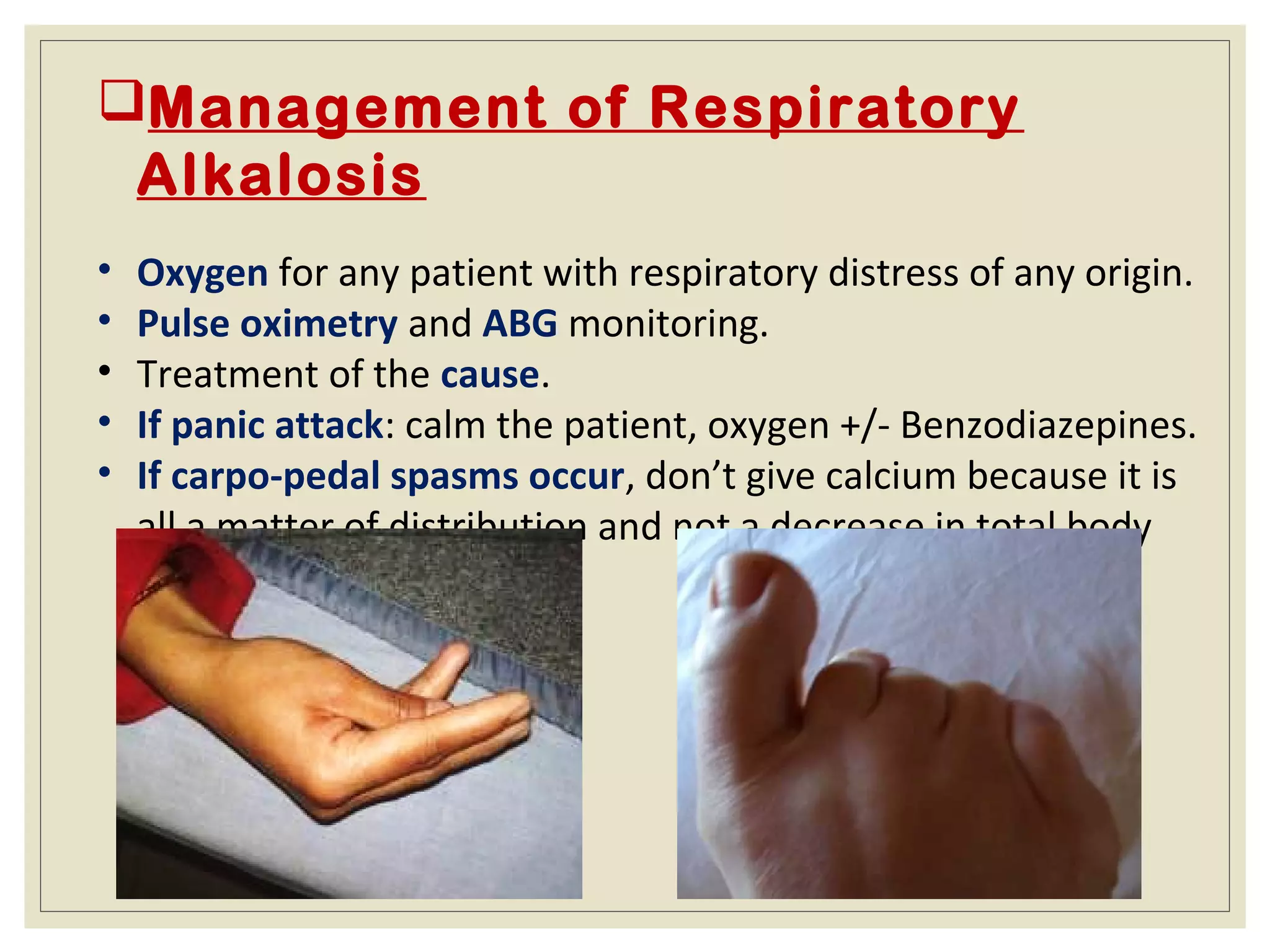
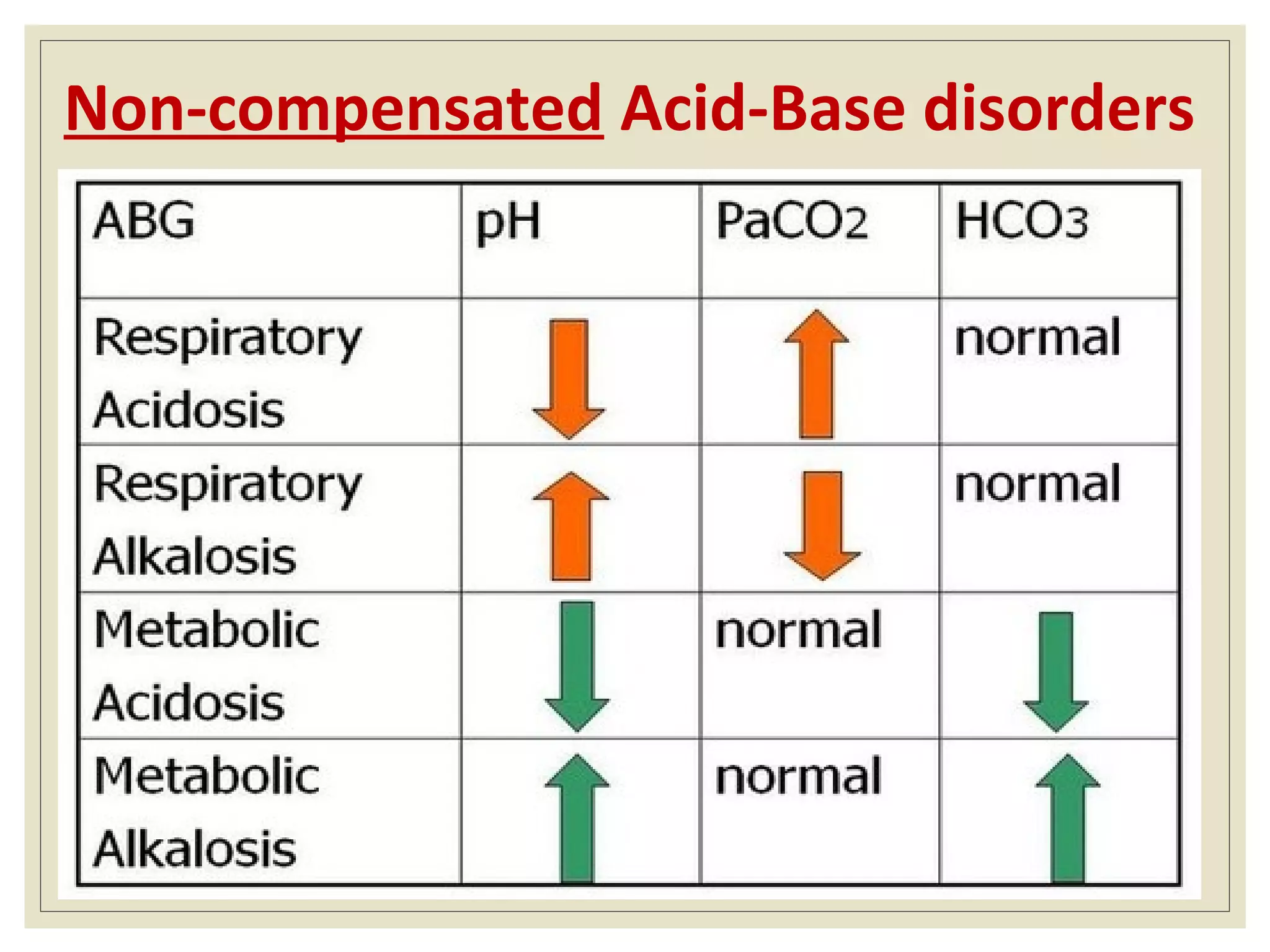
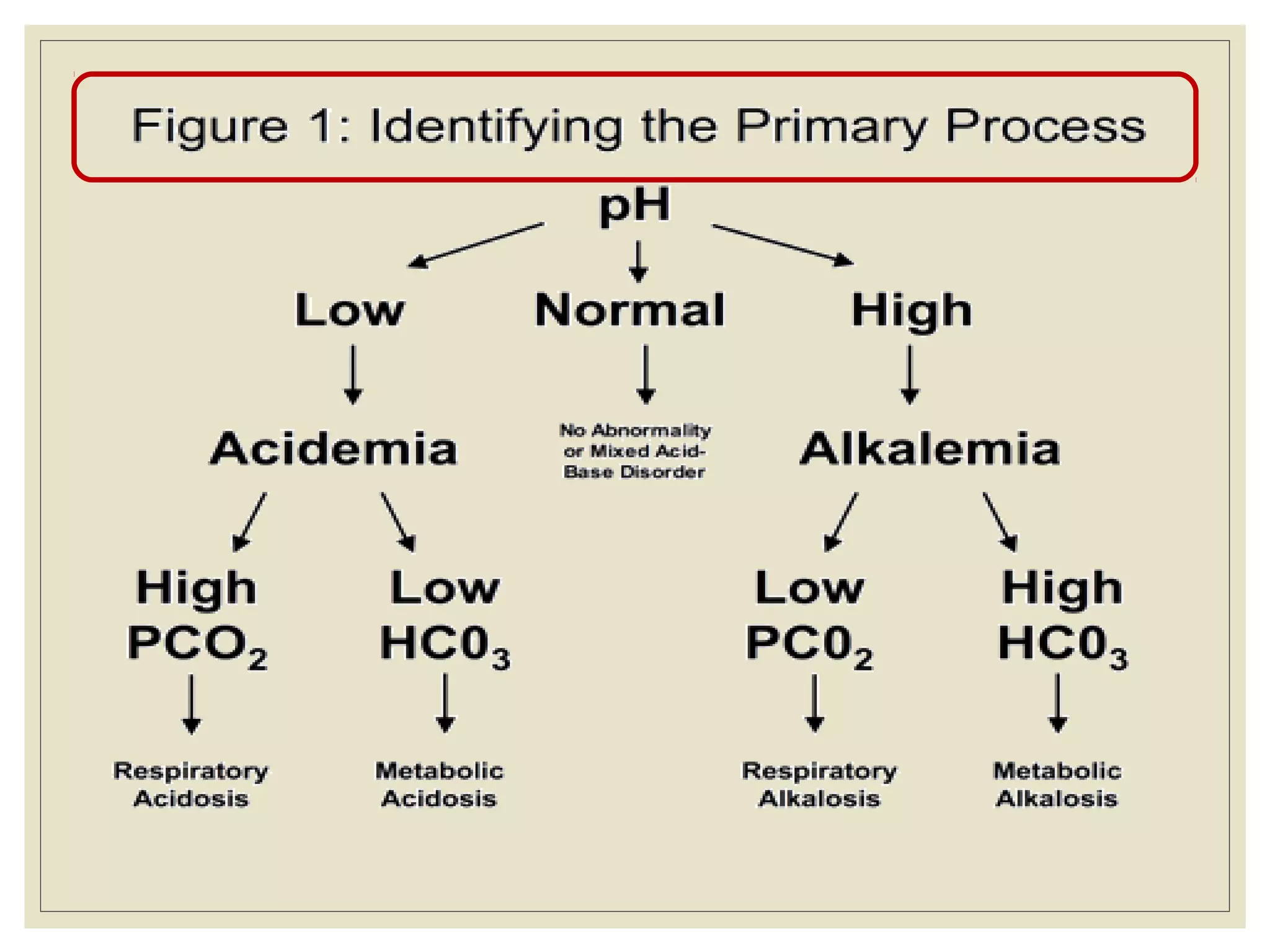
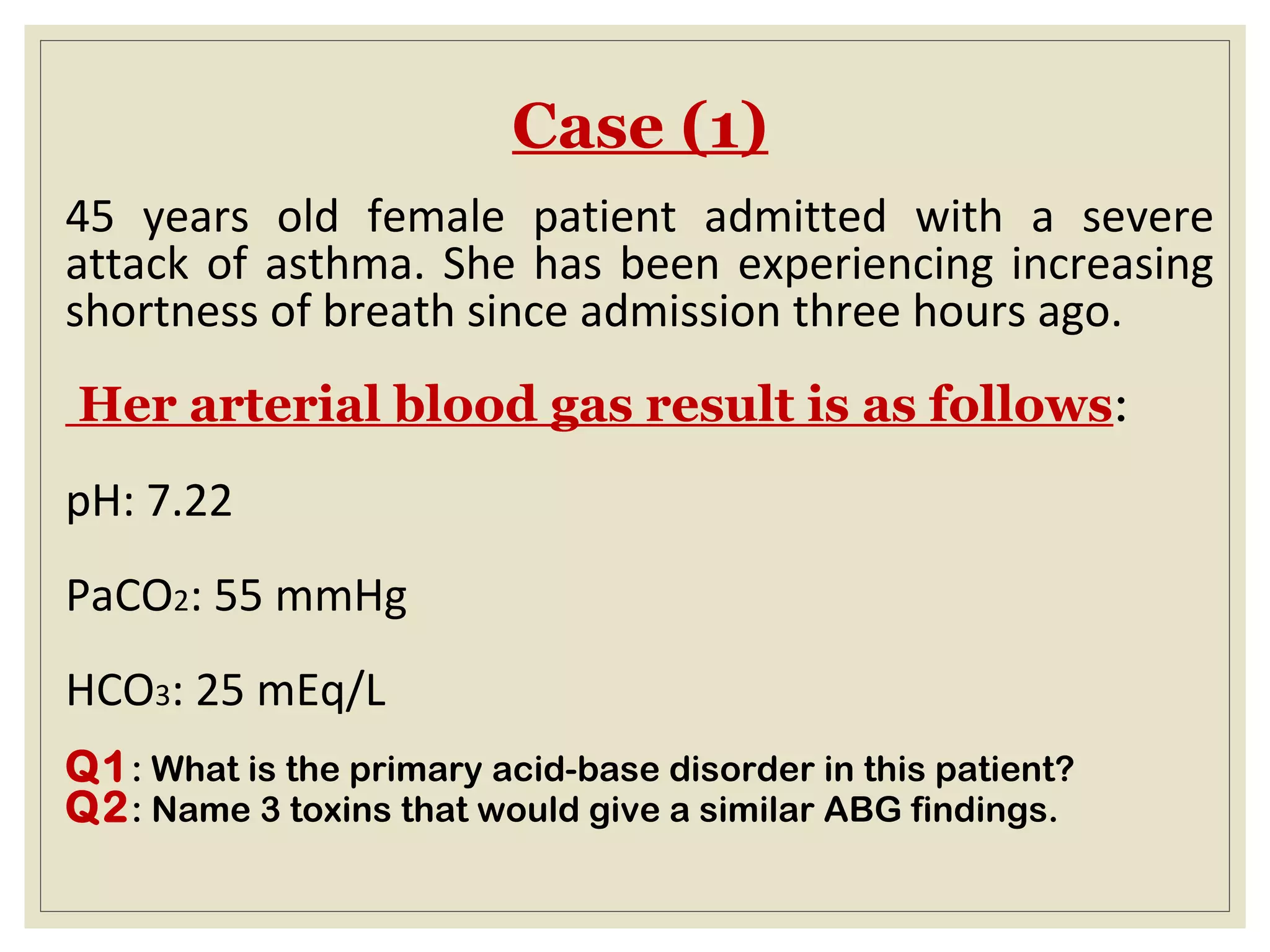
This document defines an arterial blood gas (ABG) analysis and its components. It discusses the normal acid-base balance and how acid-base imbalances present as respiratory or metabolic acidosis or alkalosis. Specific examples of each type of acid-base disorder are provided along with their typical causes, signs, symptoms, and management approaches. Common toxins that can cause acid-base disturbances are also listed. The document aims to equip readers to interpret ABG results in the clinical toxicology setting.