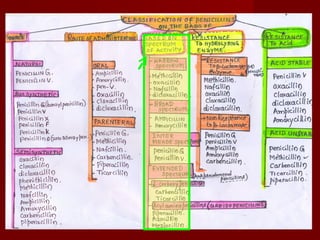
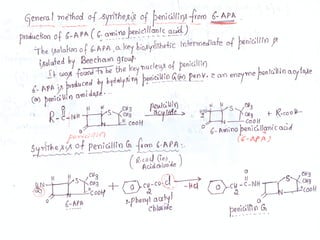
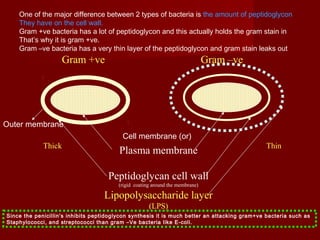
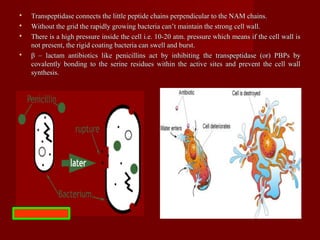
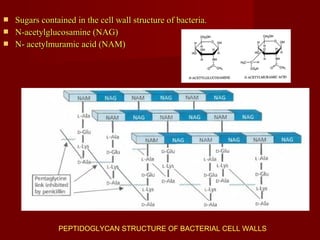
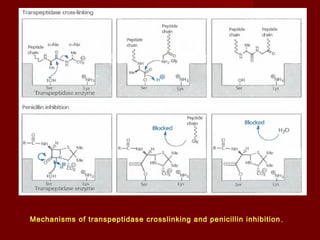
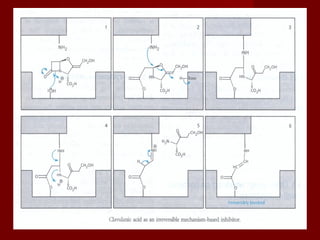
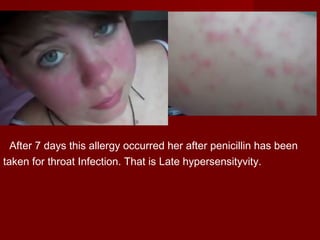
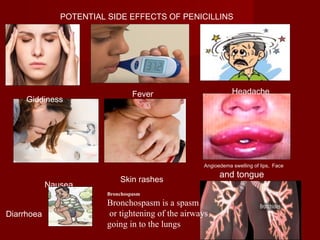
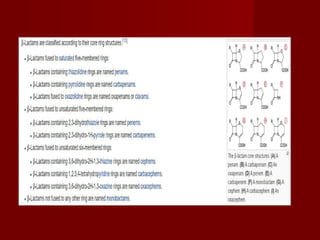
The document discusses the history, definition, and classification of antibiotics, highlighting their discovery and development over time. It details the requirements for a substance to be classified as an antibiotic and lists various types based on chemical structure and source. Key historical milestones include the discovery of penicillin by Alexander Fleming and the subsequent development of numerous antibiotics used in medicine today.







![Mechanisms of antibiotic action [bacterial cell and drug targets]](https://image.slidesharecdn.com/penicillinslatest8-8-2018-180826023506/85/Antibiotics-9-320.jpg)














![Nomenclature of Penicillins
Chemical abstract system(CAS):
According to this system penicillins are numbered starting from “S” atom.
Sulphur atom is assigned the 1st
position and the “N” atom is assigned number 4.
N
S
HH
O
N
S CH3
CH3
HH
O
COOH
1 2
3
4
56
7
(4-aza-1- thiabicyclo[3.2.0]heptane)-7-one
United States Pharmacopoeia (USP system):
The USP system of naming penicillins is the reverse of CAS.
According to this system the nitrogen atom is given the 1st
position and “S” atom is assigned the 4th
position.
N
S
HH
O 1
2
34
56
7
(4-Thia-1-azabicyclo[3.2.0]heptane)-7-one
penam(or)
As a derivatives of penicillanic acid:
According to this method of nomenclature penicillind consists of penicillanic acid ring system with
2,2-dimethyl and corboxyl groups as substutuents at positions 2 and 3 respectively.
2
3 H
As derivatives of penicillins (On the basis of Rgroup)
This is most trivalof all the naming system. In this system the entire 6-corbonylamino penicillanic acid
(6 corbonyl-APA) portion of the molecule is named as penicillin and the different penicills are
distinguished on the basis of the R group on the amino acyl side chain.
N
S CH3
CH3
HH
O
COOH
2
3 H
HNC
O
R
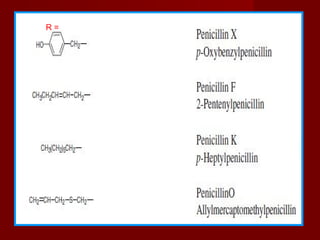
Thus when R is is a Benzyl group CH2
O
Benzyl penicillin (or) Penicillin G
CH2 Phenoxy methyl penicillin (penicillin- v)when R is Phenoxy methygroup](https://image.slidesharecdn.com/penicillinslatest8-8-2018-180826023506/85/Antibiotics-31-320.jpg)
![N
S CH3
CH3
HH
O
COOH
2
3 H
HNC
O
H2C 1
4
5
7
6
(2S,5R,6R)-3,3-dimethyl-7-oxo-6-(2-phenylacetamido)-4-thia-1-azabicylo[3.2.0]heptane-2-corboxylate.
According to CAS
Penicillin G or benzyl penicillin nomenclature:
According to USP Pencicillin G chemical name is
N
S CH3
CH3
HH
O
COOH
2
3
H
HNC
O
H2C
1
4
5
7
6
(3S,5R,6R)-2,2-dimethyl-7-oxo-6-(2-phenylacetamido)-4-aza-1-thiabicylo[3.2.0]heptane-3-corboxylate.](https://image.slidesharecdn.com/penicillinslatest8-8-2018-180826023506/85/Antibiotics-32-320.jpg)